- Home
- »
- Clinical Diagnostics
- »
-
Bilirubin Testing Market Size & Share, Industry Report, 2033GVR Report cover
![Bilirubin Testing Market Size, Share & Trends Report]()
Bilirubin Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Test Cartridges & Strips, Dedicated Blood Bilirubin Analyzers), By Testing , By Patient (Neonates), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-871-8
- Number of Report Pages: 320
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Bilirubin Testing Market Summary
The global bilirubin testing market size was estimated at USD 2.16 billion in 2025 and is projected to reach USD 2.94 billion by 2033, growing at a CAGR of 4.04% from 2026 to 2033. The market is experiencing a transformative phase, driven by an urgent need for early diagnostic tools in neonatal care and the rising prevalence of liver-related diseases.
Key Market Trends & Insights
- The U.S. led the North American market and held the largest revenue share in 2025
- By product, the bilirubin reagent kits and assay consumables segment held the largest market share of 63.65% in 2025.
- By testing, the laboratory-based testing segment held the largest market share of 89.36% in 2025.
- By application, the Liver Function Testing (LFT) segment held the largest market share of 55.44% in 2025.
- By patients, the adults segment held the largest market share of 63.25% in 2025.
- By end use, the hospitals & NICUs segment held the largest market share of 45.11% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.16 Billion
- 2033 Projected Market Size: USD 2.94 Billion
- CAGR (2026-2033): 4.04%
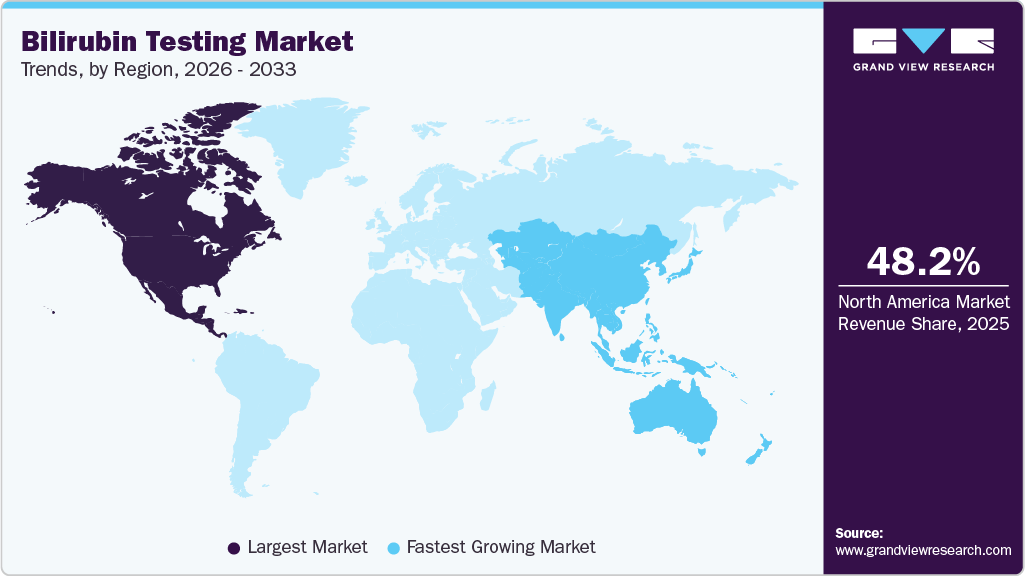
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Recently, there is a significant pivot toward point-of-care (POC) testing, with new microfluidic devices achieving over 90% sensitivity in clinical trials. These innovations are largely propelled by the increasing incidence of neonatal jaundice, which affects nearly 60% of full-term infants, and the global surge in chronic liver conditions such as Nonalcoholic Fatty Liver Disease (NAFLD).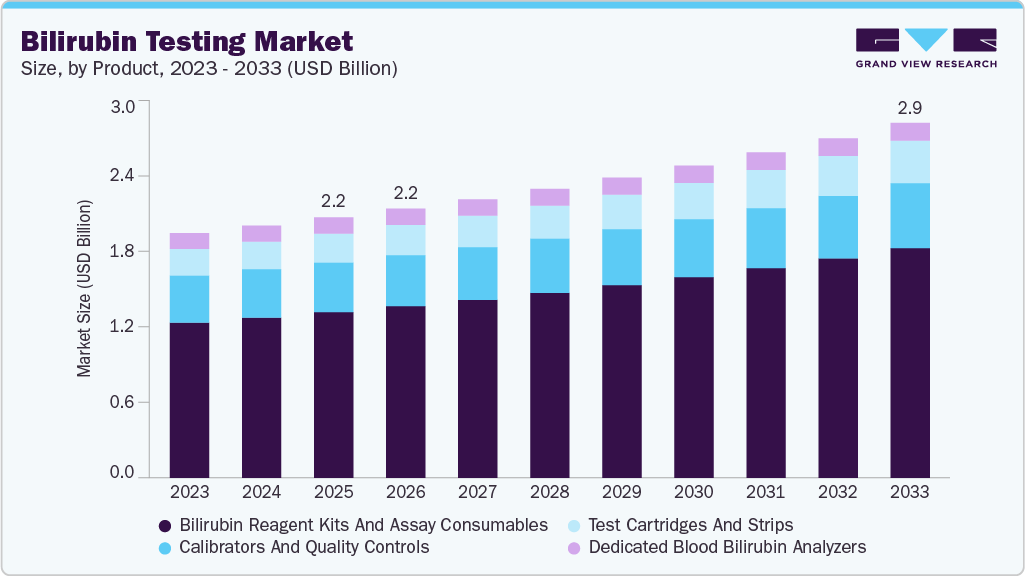
Strategic moves by industry leaders, including the integration of automated high-throughput analyzers in diagnostic labs, are streamlining workflows and reducing the time-to-result from hours to minutes. This evolution is further supported by updated clinical guidelines from organizations like the American Academy of Pediatrics (AAP), which advocate for more rigorous screening protocols to prevent kernicterus, thereby cementing bilirubin testing as a non-negotiable standard in modern pediatric healthcare.
In July 2025, a pilot study reported the development of a new microfluidic-based point-of-care device for bilirubin testing that demonstrated strong correlation (R² = 0.986) with standard laboratory methods, along with high sensitivity (90%) and specificity (97%) in classifying clinically relevant bilirubin levels. Designed with portable optoelectronic sensing technology, the device aims to provide accurate, rapid testing outside traditional laboratory settings, particularly benefiting neonatal jaundice screening and liver function assessment in decentralized or resource-limited environments. Although validated on a small sample size and requiring broader clinical studies, the innovation supports the expansion of point-of-care diagnostics, increases accessibility to bilirubin testing, and is likely to drive growth in the decentralized and neonatal diagnostics segments of the market.
One of the most significant developments in the market scope is the rapid adoption of transcutaneous bilirubinometry (TcB) and smartphone-based diagnostic applications. Traditionally, bilirubin levels were measured via invasive "heel stick" blood draws, which cause significant distress to neonates and carry risks of infection. However, the market is now shifting toward light-based, non-invasive sensors that provide instantaneous readings through the skin. Emerging research into Artificial Intelligence (AI) and machine learning has introduced 1D Convolutional Neural Networks capable of analyzing smartphone images of an infant's skin or eyes to predict bilirubin levels with near-clinical accuracy. This technological leap not only enhances patient comfort but also expands the market’s reach into home-based monitoring and remote healthcare settings, allowing parents and clinicians to track jaundice levels without frequent hospital visits.
A defining trend in 2026 is the rapid shift toward non-invasive transcutaneous bilirubinometers and AI-integrated point-of-care (PoC) devices, which significantly reduce the need for painful blood draws in newborns. Geographically, North America remains the largest market due to its advanced neonatal care infrastructure, while the Asia-Pacific region is the fastest-growing segment, fueled by massive healthcare investments in countries like India and China. Recent industry developments include Mindray's strategic acquisition of DiaSys Diagnostic Systems to bolster its international supply chain and the launch of innovative reagent kits, such as Athenese-Dx's TRUEchemie, which offers an industry-leading 30-month shelf life.
While neonatal screening remains the primary revenue generator, the market scope is broadening to include comprehensive adult hepatology. Bilirubin is a critical biomarker for assessing liver function, bile duct patency, and hemolytic anemia. As the global population ages and lifestyle-related metabolic disorders rise, the demand for Total Serum Bilirubin (TSB) and direct/indirect bilirubin fractionation in adults is surging. Diagnostic labs are increasingly investing in sophisticated chemistry analyzers that can handle high volumes of tests with extreme precision. Furthermore, the integration of bilirubin testing into routine metabolic panels during annual physicals is becoming more common, especially as healthcare systems emphasize preventive care. This dual demand-life-saving screening for newborns and chronic disease management for adults-creates a robust and resilient market environment.
Market Concentration & Characteristics
The bilirubin testing industry demonstrates a high degree of innovation, driven by advancements in point-of-care (POC) diagnostics, non-invasive transcutaneous bilirubinometers, and integration with digital health platforms. Companies such as F. Hoffmann-La Roche and Abbott Laboratories have expanded automated clinical chemistry platforms with enhanced sensitivity and multi-analyte capabilities, enabling faster turnaround times in hospital laboratories. In parallel, portable bilirubin meters are increasingly adopted in neonatal intensive care units (NICUs) to reduce invasive blood sampling. Recent product enhancements focus on connectivity features that allow seamless integration with laboratory information systems (LIS), supporting real-time data sharing and remote monitoring.
The bilirubin testing industry experiences a high level of M&A activity as global diagnostics leaders pursue acquisitions to broaden their assay menus and strengthen technological capabilities. For instance, Danaher Corporation has strategically expanded its diagnostics portfolio through acquisitions under subsidiaries such as Beckman Coulter, reinforcing its clinical chemistry presence. Similarly, Siemens Healthineers has actively invested in diagnostic automation and digital solutions to enhance integrated testing platforms. These transactions enable companies to consolidate their market share, expand into emerging economies, and integrate novel assay technologies, including advanced photometric and enzymatic bilirubin testing methods.
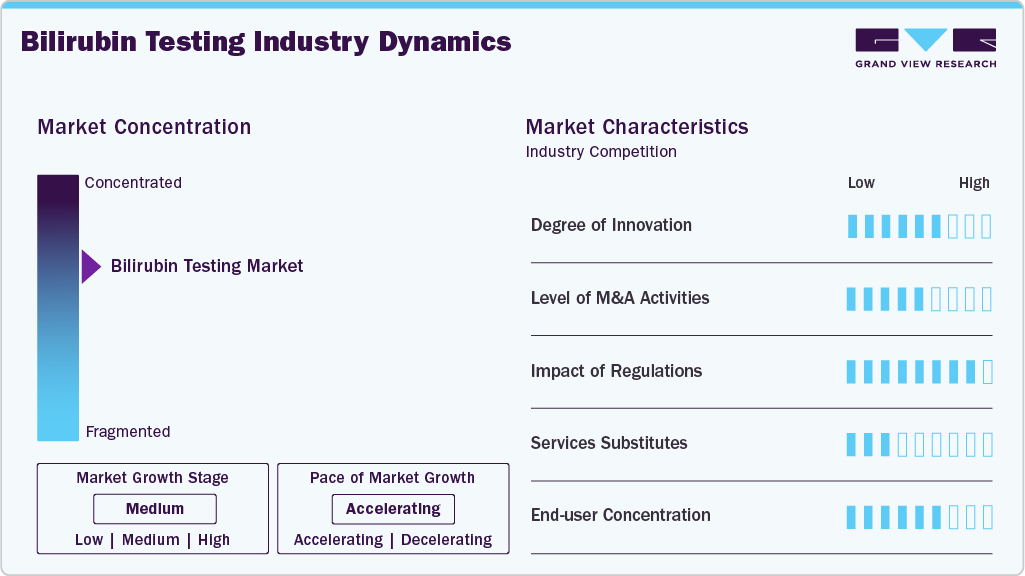
The bilirubin testing industry operates under high regulatory scrutiny due to its clinical significance in diagnosing neonatal hyperbilirubinemia and liver disorders. Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency impose stringent quality, safety, and performance requirements for in vitro diagnostic (IVD) devices. The implementation of the European Union’s In Vitro Diagnostic Regulation (IVDR) has further increased compliance obligations for manufacturers, requiring robust clinical performance data and post-market surveillance.
Product expansion within the bilirubin testing industry remains high, driven by rising neonatal birth rates in developing countries and increasing prevalence of liver diseases globally. Companies such as Drägerwerk AG have expanded their neonatal care portfolios with advanced transcutaneous bilirubin monitoring devices, while Konica Minolta continues to enhance handheld bilirubinometers with improved calibration and multi-parameter capabilities.
The bilirubin testing industry is witnessing medium to high regional expansion, particularly across Asia-Pacific, Latin America, and the Middle East, where improving healthcare infrastructure and rising awareness of neonatal jaundice screening are accelerating demand. Multinational players are strengthening distribution networks and forming local partnerships in countries such as India and Brazil to capture untapped growth potential. Meanwhile, established markets in North America and Europe continue to upgrade laboratory automation systems, sustaining steady demand. Government-led maternal and child health programs in emerging economies further support market penetration, while increased healthcare expenditure and expansion of diagnostic laboratory chains contribute to broader geographic footprint and long-term revenue growth.
Product Insights
The bilirubin reagent kits and assay consumables segment dominated the market and held the largest revenue share of 63.65% in 2025, driven by the majority of total revenue due to its recurring demand model and integration into routine diagnostic workflows. Unlike capital equipment, reagent kits generate continuous revenue streams driven by high test volumes across hospitals, reference laboratories, and diagnostic chains. Bilirubin testing is a standard component of liver function panels, metabolic assessments, and neonatal screening protocols, ensuring sustained consumption across both developed and emerging healthcare systems. The expansion of automated chemistry analyzers has further strengthened reagent utilization, as higher throughput systems increase daily testing capacity. In addition, laboratories increasingly prefer standardized, ready-to-use liquid-stable reagents that improve workflow efficiency and reduce operational variability
Test cartridges and strips segment is anticipated to grow at the fastest CAGR over the forecast period, due to the decentralization of diagnostics and the expansion of near-patient testing environments. Cartridge-based systems reduce manual handling, improve turnaround time, and require minimal technical expertise, making them highly suitable for emergency rooms, ambulatory settings, and rural healthcare centers. In September 2023, the Indian Institute of Technology Kanpur (IIT-K) signed a memorandum of understanding with Sensa Core Medical Instrumentation Pvt. Ltd. to mass-manufacture and sell a novel point-of-care electrochemical strip for bilirubin analysis in human blood/serum. Developed at IIT Kanpur’s National Centre for Flexible Electronics, the non-enzymatic sensor strip can simultaneously detect direct and total bilirubin from a single drop of blood and deliver results within a minute, offering a portable and affordable alternative to conventional methods. The agreement aims to include bilirubin test strips in point-of-care screening portfolios, supporting bedside testing, diagnostic laboratories, and health screening centres, with potential benefits for conditions such as neonatal jaundice due to its rapid, accurate detection capabilities
Testing Insights
The laboratory-based testing segment dominated the market and held the largest revenue share of 89.36% in 2025. High-throughput automated analyzers in hospital laboratories and independent diagnostic centers process large volumes of liver function tests daily, making centralized testing economically efficient and operationally scalable. Laboratory-based systems offer superior analytical precision, multi-parameter capability, and strong compliance with regulatory standards, making them the preferred method for confirmatory testing and chronic disease monitoring. In addition, large healthcare institutions continue investing in automation, connectivity solutions, and laboratory information system integration, further reinforcing centralized testing dominance.
Point-of-Care (POC) testing is anticipated to grow at the fastest CAGR over the forecast period. This growth is driven by the increasing need for rapid diagnosis in emergency care, neonatal units, and outpatient settings. POC bilirubin testing significantly reduces turnaround time, enabling immediate clinical decision-making. The rising emphasis on early detection of neonatal hyperbilirubinemia and the reduction of hospital stay durations further supports adoption. Technological advancements in portable analyzers, improved accuracy of compact devices, and wireless data integration are enhancing reliability and clinician confidence in decentralized settings. As healthcare systems focus on efficiency and accessibility, POC testing is expected to gradually expand its share within the overall bilirubin diagnostics landscape.
Application Insights
The Liver Function Testing (LFT) segment dominated the market in 2025, due to the widespread prevalence of liver diseases, metabolic disorders, and lifestyle-related health conditions. Bilirubin is a fundamental biomarker within LFT panels, routinely used to evaluate hepatic performance, bile duct obstruction, and hemolytic disorders. Preventive healthcare trends, including annual health check-ups and corporate wellness programs, further contribute to high test volumes. For instance, the NHS in the UK has implemented "iLFT," an automated system that analyzes a standard liver blood panel (including bilirubin, ALT, ALP, and albumin) and automatically triggers follow-up tests (like liver fibrosis tests) if liver damage is detected. This approach is highly cost-effective and improves diagnostic speed. As liver disorders increasingly correlate with rising obesity and diabetes rates, bilirubin testing remains integral to chronic disease management strategies.
Neonatal jaundice screening and monitoring segments are anticipated to grow at a considerable CAGR over the forecast period. The rising rate of institutional deliveries and expanding newborn screening initiatives are primary growth catalysts. Early detection of hyperbilirubinemia is critical to preventing neurological complications, prompting hospitals to implement systematic screening protocols. In addition, increased awareness among pediatricians and parents regarding early-stage jaundice management has elevated testing rates. Technological advancements in non-invasive monitoring devices further enhance adoption in maternity wards and neonatal intensive care units. With global healthcare policies increasingly prioritizing maternal and child health, neonatal screening applications are expected to drive sustained growth in the forecast period.
Patient Insights
The adults segment accounted for the largest revenue share of market in 2025, largely due to the high burden of chronic liver conditions and systemic diseases in the adult population. Adults undergo routine liver function testing as part of diagnostic evaluation for metabolic disorders, cardiovascular diseases, infectious diseases, and medication monitoring. Long-term therapeutic regimens often require periodic liver enzyme and bilirubin assessments to detect hepatotoxicity. Moreover, lifestyle-related risk factors such as alcohol consumption, obesity, and sedentary behavior contribute significantly to liver dysfunction, reinforcing diagnostic demand. The expansion of preventive screening programs and rising health awareness in middle-income populations further sustain adult testing volumes.
The neonates segment is expected to grow at the fastest CAGR during the forecast period. Neonatal jaundice remains one of the most common clinical conditions in newborns worldwide, necessitating early and repeated bilirubin measurements. Growing investments in neonatal care infrastructure, particularly in developing countries, are enhancing screening accessibility. In February 2026, A systematic review published in Healthcare (Volume 14, Issue 3) evaluated the performance of point-of-care (PoC) bilirubin testing systems for neonates, highlighting that neonatal jaundice remains a significant clinical condition with potential complications such as bilirubin-induced neurological dysfunction if not detected early. The review compared available PoC bilirubin devices with standard laboratory methods and discussed their accuracy, clinical utility, and operational benefits in settings where rapid decision-making is critical, such as maternity and neonatal care units. It emphasized the importance of reliable, bedside bilirubin measurement to support early diagnosis and treatment of hyperbilirubinemia in newborns, suggesting that advances in portable testing technologies may improve clinical workflows and broaden access to effective neonatal care
End Use
The hospitals & NICUs and NICU’s segment held the largest market share in 2025. Hospitals & NICUs conduct high volumes of liver function tests for inpatient and outpatient populations, ensuring consistent reagent and analyzer utilization. NICUs specifically drive demand for both laboratory-based and bedside bilirubin monitoring solutions to manage neonatal jaundice efficiently. Integration of automated analyzers, electronic medical records, and standardized screening pathways further reinforces hospital dominance. In addition, tertiary care centers often serve as referral hubs, increasing testing intensity and complexity.
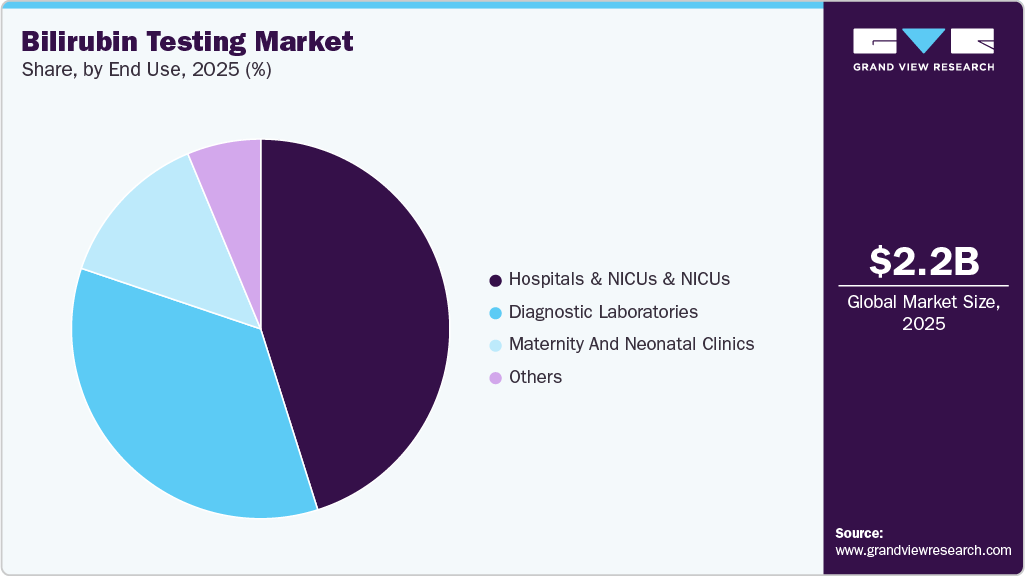
The others segments are expected to grow at the fastest CAGR over the forecast period. This growth is primarily driven by the expansion of private diagnostic chains and the decentralization of healthcare services. Increasing patient preference for outpatient diagnostics, coupled with shorter waiting times and competitive pricing, is shifting routine testing volumes toward independent laboratories. Furthermore, government initiatives aimed at expanding primary healthcare infrastructure in rural and semi-urban areas are supporting wider adoption of compact chemistry analyzers. As healthcare delivery models evolve toward accessibility and efficiency, non-hospital end use settings are expected to contribute significantly to future market expansion.
Regional Insights
North America bilirubin testing industry dominated the market and accounted for a 48.22% share in 2025. The North American market is driven by advanced healthcare infrastructure, widespread neonatal jaundice screening protocols, and the substantial burden of liver diseases affecting millions. U.S. dominance stems from high awareness, extensive insurance coverage, and major players like Abbott Laboratories and Thermo Fisher Scientific channeling heavy R&D into point-of-care innovations, transitioning from traditional lab assays to rapid handheld meters. CDC-highlighted liver conditions in nearly 4.5 million adults amplify demand for precise diagnostics, while preterm birth trends necessitate early intervention tools, propelling bedside testing adoption.
U.S. Bilirubin Testing Market Trends
The U.S. bilirubin testing industry leads the market, driven by the prevalence of neonatal jaundice, chronic liver diseases, and a mature ecosystem of leading IVD firms investing in cutting-edge solutions. Major companies such as Abbott and Thermo Fisher lead with R&D-focused portfolios targeting point-of-care analyzers, responding to CDC data on liver impacts across millions and rising preterm cases requiring immediate screening. This impetus manifests in shifts toward portable devices, enhancing clinical efficiency in hospitals and clinics. The market's resilience shines through adaptive innovations amid healthcare cost pressures, prioritizing non-invasive transcutaneous methods for newborns. Strategic player dominance ensures sustained pipeline vitality, with U.S.-specific protocols amplifying adoption rates. This positions the U.S. as a trendsetter, influencing continental dynamics through exported tech and best practices in jaundice management.
Europe Bilirubin Testing Market Trends
The Europe Bilirubin Testing (IVD) market is rapidly evolving. The region's evolution flows from standardization to innovation: mercury-free bilirubinometers address environmental concerns, while cross-border research transfers tech from academia to manufacturers. Western Europe's robust primary care pushes decentralized testing, contrasting emerging Eastern needs for disparity reduction. This regulatory-tech synergy propels growth, with 2025 trials validating commercialization potential and fostering widespread multi-panel adoption. Europe's balanced approach merging compliance, collaboration, and clinical evidence drives efficient scaling, exemplifying integrated diagnostics leadership.
The UK bilirubin testing industry is experiencing significant growth, driven by NHS-mandated universal neonatal screening, rising liver disorder cases, and integration of advanced IVD under post-Brexit frameworks. Emphasis on cost-effective solutions aligns with primary care expansions, building on EU-harmonized protocols for jaundice prevention. Neonatal jaundice monitoring remains standard practice across maternity hospitals, ensuring consistent bilirubin assay demand. Continued NHS investment in laboratory capacity expansion further supports growth.
The bilirubin testing industry in France is evolving rapidly, due to modernization of hospital laboratories and increased integration of automated clinical chemistry systems. Public healthcare reforms emphasize early diagnosis and chronic disease management, indirectly strengthening liver function test volumes. Rising awareness regarding metabolic disorders and hepatitis screening contributes to higher diagnostic rates. Neonatal screening coverage remains strong, supporting stable demand for bilirubin monitoring solutions. Laboratory digital transformation and regional hospital upgrades further enhance efficiency and testing capacity.
Germany bilirubin testing industry is expected to grow over the forecast period. The presence of leading players such as Siemens Healthineers AG reinforces innovation in clinical chemistry platforms. High diagnostic accuracy standards and strong reimbursement systems sustain centralized testing dominance. The increasing incidence of chronic liver disorders and preventive health screening programs contribute to steady reagent consumption. Continued investment in hospital laboratory modernization ensures stable market expansion
Asia Pacific Bilirubin Testing Market Trends
The Asia Pacific bilirubin testing industry is emerging as the fastest-growing market, with a CAGR of 6.18% over the forecast period. The growth is driven as countries are investing in laboratory automation to improve diagnostic accessibility in secondary and tertiary hospitals. Government-led maternal and child health initiatives are significantly boosting neonatal bilirubin screening volumes. Expansion of private diagnostic chains and improved insurance coverage further support testing growth. The region’s large patient population and rising healthcare expenditure position it as the fastest-growing contributor globally.
China bilirubin testing industry is expanding rapidly due to healthcare reforms emphasizing early disease detection and rural healthcare strengthening. Domestic IVD manufacturing growth improves affordability and accessibility of biochemical assays. Expansion of hospital networks and tiered diagnostic systems increases routine liver function testing volumes. Rising hepatitis prevalence and metabolic disorders further elevate demand.
Japan bilirubin testing industry is expected to expand rapidly over the forecast period. Advanced laboratory automation and strict clinical guideline implementation sustain consistent bilirubin testing demand. Chronic liver disease management programs and strong neonatal screening standards further reinforce stable utilization rates.
Latin America Bilirubin Testing Market Trends
Latin America’s bilirubin testing industry is growing due to technological advancements in the healthcare sector. Key factors contributing to this growth include increasing government expenditure on Research & Development (R&D), and governments are increasing healthcare investment to improve biochemical testing access. Rising hepatitis prevalence and maternal healthcare improvements contribute to steady demand growth.
The Brazil bilirubin testing industry is witnessing substantial growth. Brazil represents a key Latin American market, driven by expansion of tertiary hospitals and private laboratory networks. Urban healthcare modernization and increasing preventive screening initiatives are strengthening liver panel volumes. Neonatal screening improvements further support bilirubin monitoring adoption.
Middle East & Africa Bilirubin Testing Market Trends
The Middle East and Africa (MEA) bilirubin testing industry is poised for growth over the forecast period. Gulf countries are modernizing diagnostic services, increasing automated analyzer adoption. Growing prevalence of metabolic disorders and improving neonatal care facilities are key growth drivers.
Saudi Arabia bilirubin testing industry is witnessing strong growth aligned with Vision 2030 healthcare transformation initiatives. Expansion of tertiary hospitals increased chronic disease screening and strengthening maternal healthcare programs are boosting bilirubin testing demand. Improved laboratory automation and public healthcare investments continue to reinforce market expansion.
Key Bilirubin Testing Company Insights
The competitive landscape of the bilirubin testing industry is characterized by the strong presence of established multinational diagnostics companies alongside a growing base of emerging innovators. Leading players such as Abbott Laboratories, bioMérieux, F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Thermo Fisher Scientific, Inc., and Bio-Rad Laboratories, Inc. dominate the market through comprehensive clinical chemistry portfolios, advanced automation platforms, and strong global distribution networks. These mature players invest significantly in high-throughput analyzers, digital laboratory integration, and assay menu expansion to maintain technological leadership. Their strategies focus on continuous product innovation, strategic acquisitions, and global partnerships to enhance assay precision, expand liver function testing capabilities, and strengthen their installed base across hospitals and reference laboratories. Infrastructure scalability and integrated data management systems further allow these companies to efficiently process high testing volumes, reinforcing recurring reagent revenues.
Alongside established leaders, emerging participants such as ARUP Laboratories, RayBiotech, Inc., Randox Laboratories Ltd., Everlywell, Orreco, and Biostarks are strengthening market penetration through product differentiation and niche positioning. These companies emphasize innovative assay development, specialty biomarker testing, and decentralized diagnostic solutions. Many are actively collaborating with academic research institutes, public health organizations, and global diagnostic leaders to expand clinical validation studies and improve market access. Emerging players also focus on affordability, portability, and digital integration to target underserved markets and outpatient care settings. This dual dynamic of large-scale automation leadership and agile innovation from smaller firms is shaping a competitive yet growth-oriented bilirubin testing ecosystem.
Key Bilirubin Testing Companies:
The following key companies have been profiled for this study on the bilirubin testing market.
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Abbott
- Beckman Coulter, Inc.
- Randox Laboratories Ltd.
- SEKISUI Diagnostics
- Fortress Diagnostics
- Agappe Diagnostics Ltd.
- Nova Biomedical.
- Radiometer Medical ApS.
Recent Developments
-
In October 2025, ShenZhen New Industries Biomedical Engineering announced that it received a Medical Device Registration Certificate from the Guangdong Provincial Drug Administration for its Total Bilirubin Assay Kit (Diazonium Salt Method), valid through 2030. The kit quantitatively measures total bilirubin in human serum or plasma, contributing to the company’s biochemical reagent portfolio in the liver function category, though the registration is not expected to immediately impactdx production or financial performance.
-
In October 2025, Nova Biomedical announced that Advanced Instruments is now fully merged under the Nova Biomedical brand, creating a unified entity with a refreshed visual identity and combined expertise across analytical instrumentation and diagnostics. The merger aims to enhance global presence, accelerate innovation, and support customer workflows in clinical and biopharmaceutical markets, while maintaining existing operations and customer relationships under a single brand umbrella.
-
In Nov 2023, Verichem Laboratories introduced the Tru-Zero Bilirubin Standard, a liquid-stable, protein-based reference material designed for total and direct bilirubin assays, with a verified 0.0 mg/dL level for calibration verification across multiple wet chemistry systems. The ready-to-use standard requires no reconstitution, offers universal instrument compatibility, and supports clinical labs and IVD product developers by enhancing calibration accuracy and quality control processes.
Bilirubin Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.23 billion
Revenue forecast in 2033
USD 2.94 billion
Growth rate
CAGR of 4.04% from 2026 to 2033
Actual data
2021- 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion, volume (number of units installed and reagents sold) and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Products, testing, application, patient, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Spain; Italy; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd.; Siemens Healthineers AG; Abbott Laboratories; Beckman Coulter, Inc.; Randox Laboratories Ltd.; SEKISUI Diagnostics; Fortress Diagnostics; Agappe Diagnostics Ltd.; Nova Biomedical; Radiometer Medical ApS
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Bilirubin Testing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global bilirubin testing market report based on product, technology, application, patient, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Bilirubin Reagent Kits and Assay Consumables
-
Calibrators and Quality Controls
-
Test Cartridges and Strips
-
Dedicated Blood Bilirubin Analyzers
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Laboratory-Based Testing
-
POC Testing
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Neonatal Jaundice Screening and Monitoring
-
Liver Function Testing (LFT)
-
Hemolytic Disorders Assessment
-
Routine & Pre-operative Health Screening
-
-
Patient Outlook (Revenue, USD Million, 2021 - 2033)
-
Neonates
-
Pediatric Patients
-
Adults
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & NICUs
-
Diagnostic Laboratories
-
Maternity and Neonatal Clinics
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Spain
-
Italy
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global bilirubin testing market size was estimated at USD 2.16 billion in 2025 and is expected to reach USD 2.23 billion in 2026.
b. The global bilirubin testing market is expected to grow at a compound annual growth rate of 4.04% from 2026 to 2033 to reach USD 2.94 billion by 2033.
b. North America dominated the bilirubin testing market with a share of 48.22% in 2025. This is attributable to presence of advanced healthcare infrastructure, widespread neonatal jaundice screening protocols, and the substantial burden of liver diseases affecting millions.
b. Some key players operating in the bilirubin testing market include F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Abbott Laboratories, Beckman Coulter, Inc., Randox Laboratories Ltd., SEKISUI Diagnostics, Fortress Diagnostics, Agappe Diagnostics Ltd., Nova Biomedical, and Radiometer Medical ApS.
b. Key factors that are driving the market growth include urgent need for early diagnostic tools in neonatal care and the rising prevalence of liver-related diseases.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









