- Home
- »
- Biotechnology
- »
-
Viral Vectors And Plasmid DNA Manufacturing Market, 2033GVR Report cover
![Viral Vectors And Plasmid DNA Manufacturing Market Size, Share & Trends Report]()
Viral Vectors And Plasmid DNA Manufacturing Market (2026 - 2033) Size, Share & Trends Analysis Report By Vector Type (Lentivirus, Adenovirus, Retrovirus), By Workflow, By Application (Gene Therapy, Cell Therapy, Vaccinology), By End-use, By Disease (Cancer, Genetic Disorders), By Region, And Segment Forecasts
- Report ID: GVR-2-68038-695-0
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Viral Vectors And Plasmid DNA Manufacturing Market Summary
The global viral vector and plasmid DNA manufacturing market size was estimated at USD 6.68 billion in 2025 and is projected to reach USD 19.52 billion by 2033, growing at a CAGR of 14.5% from 2026 to 2033. One of the primary factors driving the market is the advancement in gene therapy, as the development of new gene therapies is heavily reliant on high-quality viral vectors and plasmid DNA.
Key Market Trends & Insights
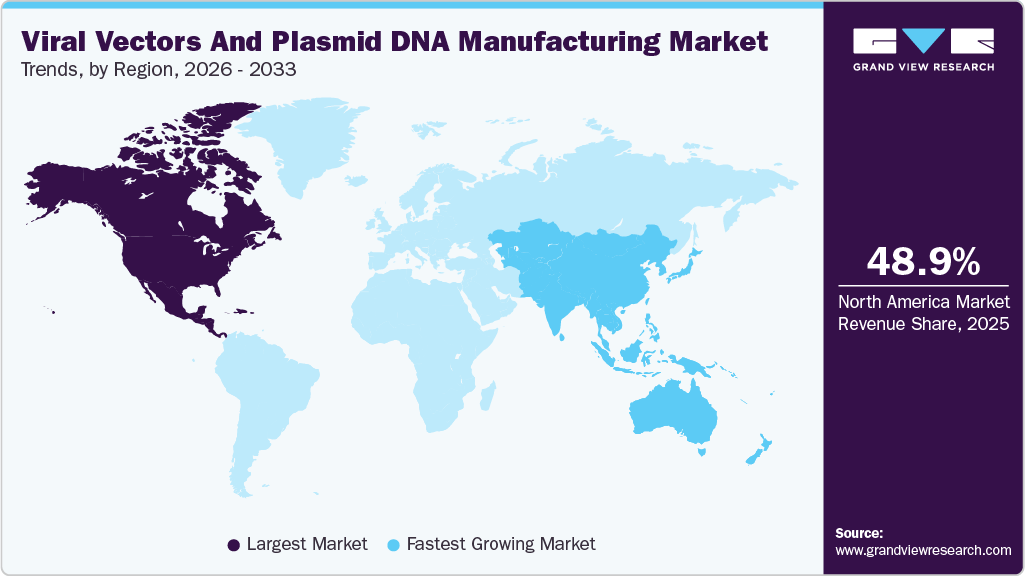
- North America viral vector and plasmid DNA manufacturing market held the largest share of 48.98% of the global market in 2025.
- The viral vector and plasmid DNA manufacturing industry in the U.S. is expected to grow significantly over the forecast period.
- By vector type, the adeno-associated virus (AAV) segment held the largest market share in 2025.
- By application, the vaccinology segment held the highest share of 22.08% in 2025.
- By end-use, the research institute segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 6.68 Billion
- 2033 Projected Market Size: USD 19.52 Billion
- CAGR (2026-2033): 14.5%
- North America: Largest market in 2025
- Asia Pacific: Fastest growth market
Furthermore, improvements in manufacturing processes have made it possible to produce viral vectors and plasmid DNA more efficiently at larger scales, while also reducing costs and improving overall quality. The growth of the viral vectors and plasmid DNA manufacturing market has been strongly driven by rising demand for gene and cell therapies, particularly across North America and Europe. The U.S. plasmid DNA manufacturing market leads due to high clinical trial activity, strong CDMO presence, and increasing outsourcing trends within the plasmid DNA contract manufacturing market. Meanwhile, the UK viral vectors plasmid DNA manufacturing market continues to expand, supported by government-backed life sciences initiatives and advanced research infrastructure. Across regions, the broader viral vector and plasmid DNA manufacturing market and the DNA plasmid manufacturing market are benefiting from growing biotech investments, capacity expansion by manufacturers, and the need for scalable, GMP-compliant production to support research, clinical development, and commercialization.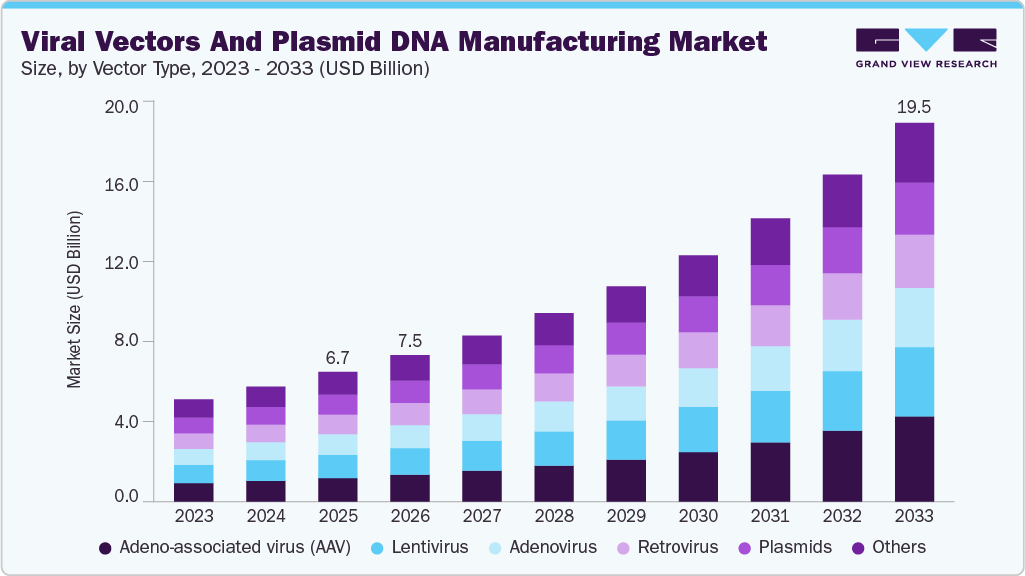
The rapid growth of cell and gene therapies, particularly in oncology, rare diseases, and genetic disorders. Increasing numbers of clinical trials, along with rising regulatory approvals for advanced therapies such as CAR-T, AAV-based gene therapies, and mRNA platforms, have significantly boosted demand for high-quality viral vectors and plasmid DNA. As biopharmaceutical companies move programs from early research into late-stage development and commercialization, the need for scalable, GMP-compliant manufacturing capabilities has continued to expand.
In addition, sustained investments in academic research, biotechnology innovation, and personalized medicine have further supported market growth. Research institutes and emerging biotech companies increasingly rely on outsourced manufacturing partners to overcome technical complexity, high capital costs, and stringent regulatory requirements. Capacity expansions by CDMOs, advancements in vector production technologies, and growing government support for life sciences infrastructure have collectively strengthened demand for reliable and efficient viral vector and plasmid DNA manufacturing solutions worldwide.
Market Concentration & Characteristics
The viral vectors and plasmid DNA manufacturing industry has witnessed a significant degree of innovation in recent years. With the increasing demand for advanced gene therapy and vaccine development, manufacturers are constantly exploring new and efficient ways to produce viral vectors and plasmid DNA. One of the key innovations in this market is the use of transient transfection technology for the large-scale production of viral vectors. Another area of innovation is the use of suspension cell culture systems for viral vector production.
Large pharmaceutical and biotechnology companies are increasingly acquiring smaller vector and plasmid DNA manufacturers to access their proprietary technologies, expertise, and production capacity. This leads to consolidation within the market, giving established players greater control over pricing and market share. Smaller companies benefit from the resources and infrastructure of larger acquirers, enabling them to scale up their operations and reach a wider audience.
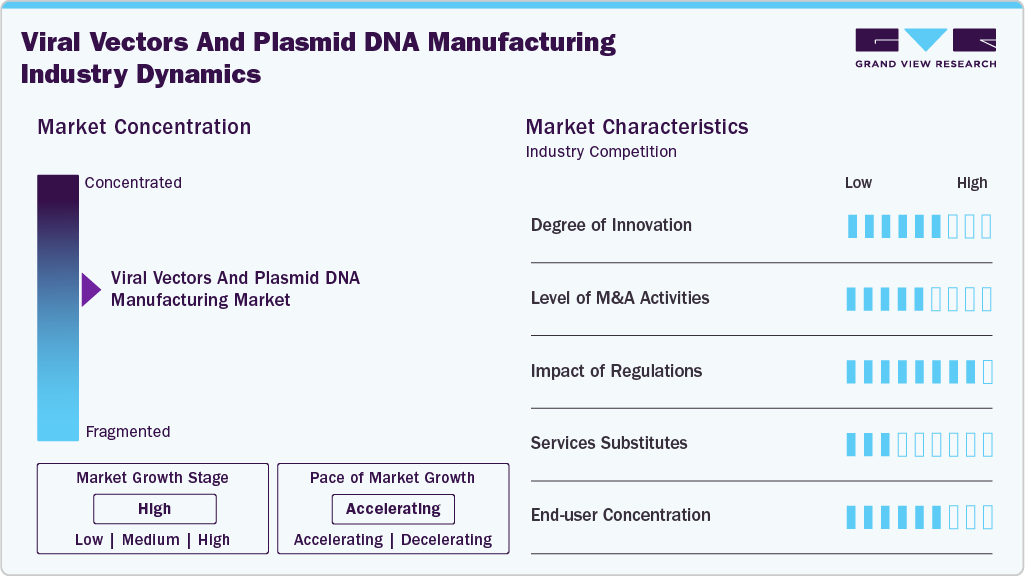
The impact of regulations on the viral vectors and plasmid DNA manufacturing market is significant, influencing product development, manufacturing processes, and market dynamics. Compliance with regulatory standards is essential for ensuring patient safety, maintaining product quality, and gaining market access. Manufacturers need to navigate these regulatory landscapes effectively to bring innovative gene therapies to patients while meeting the stringent requirements set by regulatory agencies.
Product offerings are expanding as demand diversifies beyond traditional vectors to include specialized production platforms, analytical services, and tailored manufacturing solutions for different therapeutic modalities. Market participants are launching enhanced vector types, broadening their capabilities across AAV, lentiviral, and adenoviral platforms, and integrating upstream and downstream services to address both early-stage research and large-scale commercial needs.
Geographically, North America continues to lead the viral vector industry with the largest share backed by robust R&D infrastructure and high clinical activity, while Europe maintains strength through collaborative innovation ecosystems and supportive policy frameworks. The Asia-Pacific region is emerging as the fastest-growing market, with substantial investments in biomanufacturing capacity and regulatory reforms that are attracting both local and multinational players.
Vector Type Insights
The Adeno-associated virus (AAV) segment held the largest market share of 18.17% in 2025. AAVs are in huge demand and their utilization in clinical trials is increasing rapidly as these viruses offer maximum precision in delivering the gene to the region of interest. The increasing adoption is due to clinical trials relating to the development of orthopedic and ocular gene therapy therapies exhibiting increased efficiency and efficacy.
The lentivirus segment is expected to grow at a significant CAGR over the forecast period. Increasing use of lentiviral vectors in ongoing research areas, the research industry is focusing on advancements in these vectors. For instance, scientists are studying the potential of non-integrating lentiviral vectors (NILVs) as a tool to avoid insertional mutagenesis. NILVs are able to transduce both non-dividing and dividing cells. These vectors have potential applications in CAR-T cell therapy research.
Workflow Insights
In 2025, the downstream processing segment dominated the market, accounting for the largest revenue share owing to highly complex procedures carried out for polishing and purification of clinical grade final products. Increase in demand for clinical grade viral vectors, manufacturers are involved in development of novel economic downstream processes to address the challenges associated with conventional lab-scale manufacturing of vectors and it expect to support market growth. For example, combining a fed-batch fermentation technique and genetically optimized cell systems with conventional and novel technologies for the purification of vectors.
The upstream processing segment is expected to grow at a significant CAGR over the forecast period. Upstream processing involves infecting cells with virus, cultivating of cells, and harvesting the virus from cells. Advanced product development, such as ambr 15 microbioreactor system for high-throughput upstream process development, is expected to drive the segment. ambr 15 microbioreactor system allows efficient cell culture processing with automated experimental set up and sampling, which requires less labor and laboratory space and time taken for cleaning and sterilization is also very less.
Application Insights
The vaccinology segment held the highest share of 22.08% in 2025. The increasing demand for vaccines for various diseases, such as cancer and infectious diseases such as COVID-19, primarily drives this growth. Viral vectors and plasmid DNA are widely used in the development of vaccines, and the increasing emphasis on research and development of new vaccines is expected to drive the growth of this market segment. In addition, the availability of government funding for vaccine development programs is also contributing to the growth of the vaccinology application in the viral vectors and plasmid DNA manufacturing market.
The cell therapy segment is expected to grow at the fastest CAGR over the forecast period, owing to increase in personalized cancer treatments. Moreover, the success of Chimeric Antigen Receptor (CAR)-based cell therapies for cancer treatment is expected to further fuel the market growth.
End-use Insights
The research institutes segment dominated the viral vector and plasmid DNA manufacturing industry with market share in 2025. Expanding genetic and molecular biology research in academic and government research institutes is driving demand for viral vectors and plasmid DNA manufacturing. Increased funding for gene editing, functional genomics, and translational research is pushing institutes to source reliable, high-quality vectors and plasmids to support preclinical studies and early-stage innovation.
The pharmaceutical and biotechnology companies segment is expected to grow significantly over the forecast period. This can be attributed to continuous introduction of advanced therapies coupled with subsequent increase in the number of gene therapy-based research programs by pharmaceutical firms. The number of biotech companies that are employing vectors for therapeutics production continues to increase over time.
Disease Insights
In 2025, the cancer segment dominated the market, accounting for the largest share of 38.03%. The growing use of gene and cell therapies in oncology is a major driver for viral vectors and plasmid DNA manufacturing, as many next-generation cancer treatments rely on these platforms for gene delivery. Rising cancer prevalence, along with increased clinical trials and regulatory approvals for CAR-T and other gene-based therapies, is steadily boosting demand for scalable, high-quality manufacturing solutions.
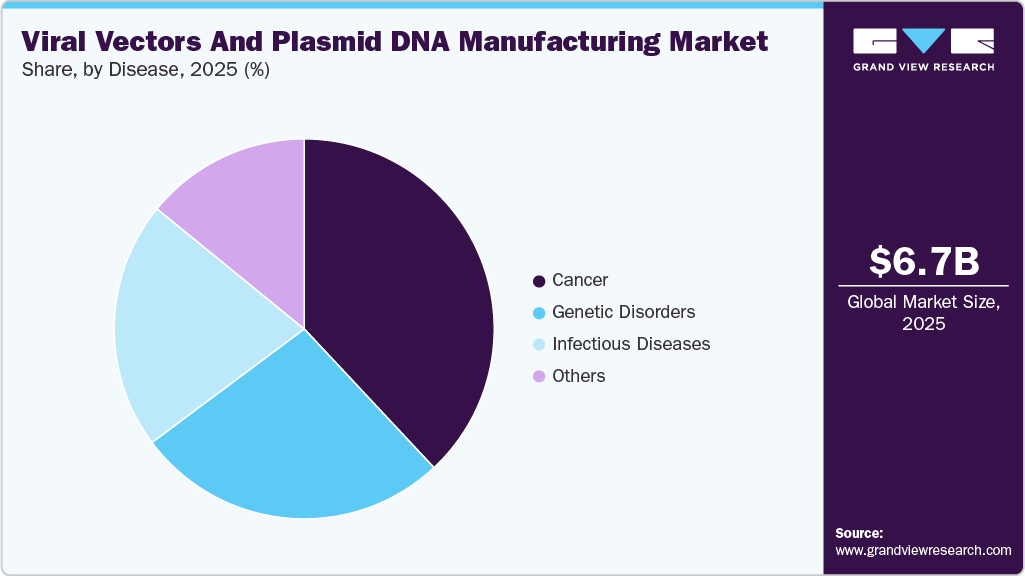
Genetic disorders are expected to register a significant CAGR during the forecast period. genetic disorders are the most focused area of application for gene therapy, with more than 10% of ongoing clinical trials directed toward the same. This makes gene therapy for genetic disorders one of the most crucial markets in the forecast years and it expected to support growth of viral vector and plasmid DNA manufacturing industry.
Regional Insights
North America viral vectors And plasmid DNA manufacturing marketdominated the market and accounted for 48.98% share in 2025. This can be attributed to the growing engagement of companies in research and product development in gene & cell therapy coupled with a substantial number of contract development organizations in the region. In addition, homegrown companies are expanding their manufacturing facilities in the region.The U.S. held the highest revenue share in North America viral vectors and plasmid DNA manufacturing market owing to the presence of key market players, including CDMOs offering GMP manufacturing services, and the adoption of highly innovative manufacturing technologies for production.
U.S. Viral Vectors And Plasmid DNA Manufacturing Vector Market Trends
Viral vector and plasmid DNA manufacturing industry in the U.S. is driven by strong biotech and pharmaceutical activity, high investment in gene and cell therapy R&D, and a large number of ongoing clinical trials. Supportive regulatory pathways and the presence of leading CDMOs further accelerate demand for viral vectors and plasmid DNA manufacturing.
Europe Viral Vectors And Plasmid DNA Manufacturing Vector Market Trends
Europe’s growth is supported by rising adoption of advanced therapies, increasing public-private research collaborations, and expanding academic research infrastructure. EU-backed funding programs for rare diseases and oncology continue to fuel demand for reliable vector and plasmid production.
Viral vectors And plasmid DNA manufacturing market in the UK benefits from a strong life sciences ecosystem, government-backed genomics initiatives, and active clinical research networks. Growing focus on cell and gene therapies, supported by institutions like the NHS and academic centers, is boosting manufacturing demand.
France viral vectors And plasmid DNA manufacturing market is witnessing increased government investment in biopharmaceutical innovation and a growing number of translational research projects are key drivers. The country’s focus on oncology and rare disease research is expanding the need for viral vectors and plasmid DNA.
Viral vectors And plasmid DNA manufacturing market in Germany is driven by its strong biomanufacturing base, advanced research institutes, and rising clinical development of gene therapies. Continuous funding for biomedical research and process innovation supports steady market growth.
Asia Pacific Viral Vectors And Plasmid DNA Manufacturing Vector Market Trends
The Asia Pacific region is witnessing rapid growth due to expanding biotech industries, increasing healthcare spending, and rising clinical trial activity. Government initiatives to strengthen local biomanufacturing capabilities are further accelerating demand.
Viral vectors and plasmid DNA manufacturing market in China is growing due to strong government support for biotechnology, rapid expansion of domestic gene therapy companies, and increasing clinical research activity. Investments in local manufacturing infrastructure are reducing reliance on imports and driving market expansion.
Japan viral vectors and plasmid DNA manufacturing market is supported by advanced regenerative medicine research, favorable regulatory reforms, and a strong focus on innovation in cell and gene therapies. Collaboration between academia and industry continues to boost demand for high-quality vectors and plasmids.
Viral vectors and plasmid DNA manufacturing market in India is gaining traction due to rising biotechnology research activities, expanding clinical development in gene-based therapies, and increasing investment in biomanufacturing infrastructure. Lower cost of operations and a growing skilled workforce make it an attractive location for both domestic and outsourced viral vector production.
Middle East & Africa Viral Vectors And Plasmid DNA Manufacturing Vector Market Trends
The Middle East market is driven by growing investments in healthcare infrastructure and biomedical research, particularly in genomics and personalized medicine. Governments are increasingly supporting life sciences as part of economic diversification strategies.
Viral vectors and plasmid DNA manufacturing market in Saudi Arabia is witnessing growth due to major healthcare spending and Vision 2030 initiatives that prioritize biotech and advanced therapeutics infrastructure. With strategic investments and policy reforms, the country aims to build self-sufficient high-tech manufacturing capacity and engage with global value chains.
UAE viral vectors and plasmid DNA manufacturing market is experiencing growth owing to the diversification of healthcare and biotech sectors, investments in research infrastructure, and partnerships with global pharmaceutical and biotech players are driving nascent but accelerating demand for viral vector technology and manufacturing. Efforts to establish regional hubs for advanced therapies underpin long-term market growth.
Key Viral Vectors And Plasmid DNA Manufacturing Vector Company Insights
Industry leaders such as Thermo Fisher Scientific, Lonza Group, Merck KGaA, and FUJIFILM Diosynth Biotechnologies have built expansive global footprints with capabilities that span preclinical development, GMP-compliant production, analytical testing and regulatory support, giving them an edge in serving both clinical and commercial needs. Their scale allows them to offer end-to-end solutions, which attracts large biopharma clients and helps them command premium pricing and long-term supply contracts, particularly in high-growth segments like AAV and lentiviral vector manufacturing. These companies collectively account for a significant portion of global capacity, and their ongoing investments in automation, single-use systems and flexible platforms position them well to capitalize on increasing demand for advanced therapies.
At the same time, specialist and mid-tier players carve out meaningful niches by focusing on specific technologies, vector types or regional markets. Firms such as Oxford Biomedica, Cobra Biologics, VGXI and Aldevron leverage deep technical know-how in particular vector platforms or plasmid quality control to attract academic, biotech and emerging therapy developers looking for tailored solutions.
Key Viral Vectors And Plasmid DNA Manufacturing Companies:
The following key companies have been profiled for this study on the viral vector and plasmid DNA manufacturing market.
- Merck KGaA
- Lonza
- FUJIFILM Diosynth Biotechnologies
- Thermo Fisher Scientific
- Cobra Biologics
- Catalent Inc.
- Wuxi Biologics
- Takara Bio Inc.
- Waisman Biomanufacturing
- Genezen laboratories
- Batavia Biosciences
- Miltenyi Biotec GmbH
- SIRION Biotech GmbH
- Virovek Incorporation
- BioNTech IMFS GmbH
- Audentes Therapeutics
- BioMarin Pharmaceutical
- RegenxBio, Inc.
Recent Developments
-
In January 2026, Asimov and AGC Biologics had signed a licensing agreement for Asimov’s off-the-shelf LV Edge Packaging cell line. Under the terms of the agreement, AGC Biologics’ Cell and Gene Center of Excellence in Milan began offering a lentiviral packaging system that enabled production from a single-plasmid transfection instead of the standard four-plasmid process.
-
In June 2025, Cell and gene therapy CDMO ProBio officially announced the opening of its flagship Cell and Gene Therapy Center of Excellence at the Princeton West Innovation Campus in Hopewell, New Jersey. The 128,000-square-foot GMP facility was set up to manufacture high-quality plasmid DNA and viral vectors, including AAV and lentiviral platforms. The enhancements were expected to enable ProBio to support a broader range of therapeutic candidates from early proof-of-concept through clinical readiness, all from a single integrated site.
-
In May 2025, CDMO 3PBIOVIAN officials reported that the company had launched the AAVion platform, a fully integrated adeno-associated virus (AAV) manufacturing solution designed to accelerate gene therapy development.
Viral Vectors And Plasmid DNA Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 7.54 billion
Revenue forecast in 2033
USD 19.52 billion
Growth rate
CAGR of 14.5% from 2026 to 2033
Actual data
2021 - 2025
Forecast Period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Vector type, workflow, application, end-use, disease, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Merck KGaA; Lonza; FUJIFILM Diosynth Biotechnologies; Thermo Fisher Scientific; Cobra Biologics; Catalent Inc.; Wuxi Biologics; Takara Bio Inc.; Waisman Biomanufacturing; Genezen laboratories; Batavia Biosciences; Miltenyi Biotec GmbH; SIRION Biotech GmbH; Virovek Incorporation; BioNTech IMFS GmbH; Audentes Therapeutics; BioMarin Pharmaceutical; RegenxBio, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Viral Vector And Plasmid DNA Manufacturing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global viral vector and plasmid DNA manufacturing market report based on vector type, workflow, application, end-use, disease, and region.
-
Vector Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Adeno-associated virus (AAV)
-
Lentivirus
-
Adenovirus
-
Retrovirus
-
Plasmids
-
Others
-
-
Workflow Outlook (Revenue, USD Billion, 2021 - 2033)
-
Upstream Manufacturing
-
Vector Amplification & Expansion
-
Vector Recovery/Harvesting
-
-
Downstream Manufacturing
-
Purification
-
Fill Finish
-
-
-
Application Outlook (Revenue, USD Billion, 2021 - 2033)
-
Antisense & RNAi Therapy
-
Gene Therapy
-
Cell Therapy
-
Vaccinology
-
Research Applications
-
-
End-use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Pharmaceutical and Biopharmaceutical Companies
-
Research Institutes
-
-
Disease Outlook (Revenue, USD Billion, 2021 - 2033)
-
Cancer
-
Genetic Disorders
-
Infectious Diseases
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
Kuwait
-
UAE
-
South Africa
-
Saudi Arabia
-
-
Frequently Asked Questions About This Report
b. Merck, Lonza, FUJIFILM Diosynth Biotechnologies, Thermo Fisher Scientific, Cobra Biologics, Catalent Inc., Wuxi Biologics, Takara Bio Inc., and Waisman Biomanufacturing are some key companies operating in the viral vector and plasmid DNA manufacturing market.
b. The viral vectors and plasmid DNA manufacturing market report scope covers segmentation by vector type, workflow, application, end-use, disease, and region.
b. The global viral vector and plasmid DNA manufacturing market size was estimated at USD 6.68 billion in 2025 and is expected to reach USD 7.54 billion in 2026.
b. The global viral vector and plasmid DNA manufacturing market are expected to witness a compound annual growth rate of 14.55% from 2026 to 2033 to reach USD 19.52 billion by 2033.
Which vector type accounted for the largest viral vector and plasmid DNA manufacturing market share?b. AAV is expected to witness a compound annual growth rate of 18.17% owing to the development of ocular and orthopedic gene therapy treatment exhibiting increased efficacy and efficiency.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









