- Home
- »
- Biotechnology
- »
-
Viral Capsid Development Market, Industry Report, 2033GVR Report cover
![Viral Capsid Development Market Size, Share & Trends Report]()
Viral Capsid Development Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Adenoviral, Lentiviral), By Application (Gene Therapy, Vaccine Development), By End-use (Academic & Research Institute, CROs & CMOs), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-865-3
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
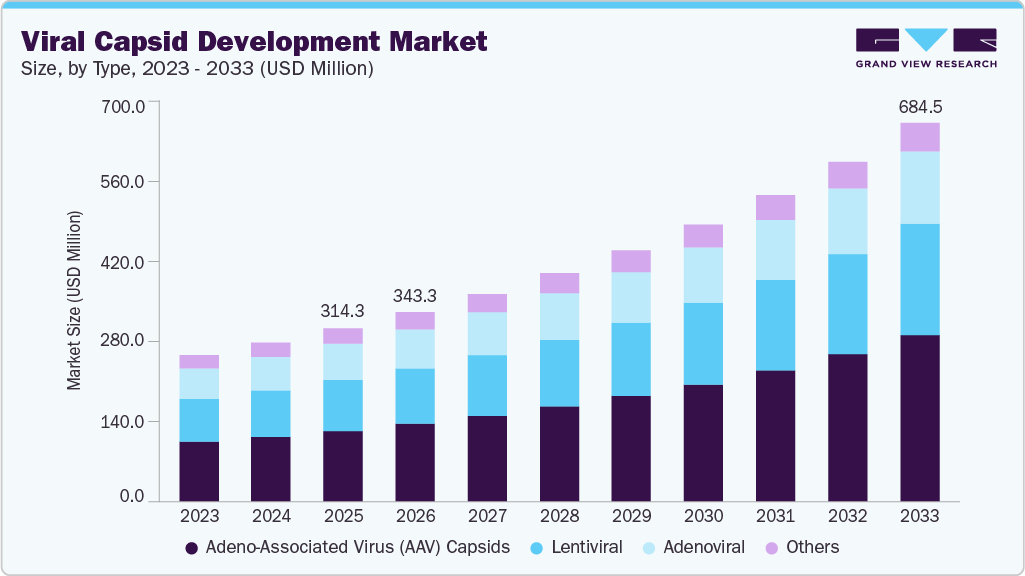
Viral Capsid Development Market Summary
The global viral capsid development market size was estimated at USD 314.3 million in 2025 and is projected to reach USD 684.5 million by 2033, growing at a CAGR of 10.36% from 2026 to 2033. Growth is driven by increasing demand for gene therapies, rising viral vector R&D, and advancements in capsid engineering, though high costs and manufacturing complexities may limit expansion.
Key Market Trends & Insights
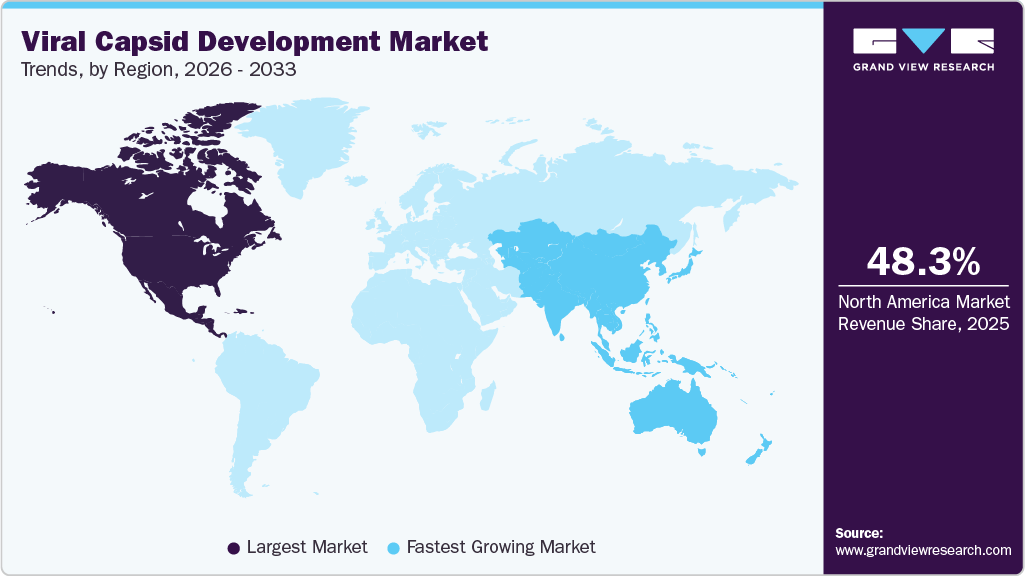
- North America dominated the global viral capsid development market with the largest revenue share of 48.31% in 2025.
- The viral capsid development industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- Based on type, the adeno-associated virus (AAV) capsids segment led the market with the largest revenue share of 40.91% in 2025.
- Based on application, the gene therapy segment accounted for the largest market revenue share in 2025.
- Based on end use, the pharmaceutical and biotechnology companies segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 314.3 Million
- 2033 Projected Market Size: USD 684.5 Million
- CAGR (2026-2033): 10.36%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Advancements in Synthetic Biology & AI-Driven Design
Advancements in synthetic biology, along with AI-powered design advancements, are accelerating the development process of viral capsid systems through enhanced development accuracy and increased developmental capacity. Scientists use directed evolution together with rational protein engineering to create custom vectors that deliver gene therapies with improved safety and better ability to reach specific body tissues.
The process of analyzing extensive databases enables artificial intelligence to discover the best sequences, which decreases the need for experiments while driving clinical development forward and increasing the requirements for superior capsid engineering methods. For instance, in November 2025, in the U.S., Latus Bio expanded its AI/ML strategy for AAV gene therapy, leveraging extensive in vivo capsid datasets and adding leading experts to accelerate capsid discovery, payload design, and targeted delivery in viral capsid development.
Need for Targeted Delivery & Tissue Specificity
Researchers develop new viral capsids to meet the growing need for targeted delivery systems, which transport genetic material to specific cells in the liver, neural, retinal, and muscle tissues. The rising number of complex disease pipelines has created demand for engineered capsids that enhance cell-specific binding, transduction efficiency, and controlled biodistribution. For instance, in May 2021, Biogen partnered with Capsigen in the U.S. and Canada to engineer novel AAV capsids for targeted CNS and neuromuscular disorders, leveraging Capsigen’s TRADE platform to accelerate gene therapy development and delivery. Such collaborations highlight how advanced capsid engineering and strategic partnerships are driving innovation and expansion in the viral capsid development industry.
The development of customized capsids is advancing as scientists seek to minimize off-target effects toward safer outcomes. The designed capsids offer three advantages: they reduce immune responses, enhance receptor binding, and enable lower drug dosing without compromising treatment effectiveness. The demand for next-generation viral capsids, which offer precise targeting capabilities, is increasing because regulatory authorities require higher safety standards and gene therapies reach wider patient populations.
Market Concentration & Characteristics
The degree of innovation in the viral capsid development industry is high, driven by advances in gene delivery technologies and the growing need for precise, efficient vectors. Key innovations focus on engineered and synthetic capsids developed through directed evolution, rational design, and AI-based optimization, enabling improved tissue targeting, higher transduction efficiency, and lower immunogenicity. Emerging developments, such as immune-evasive and programmable capsids, further support the development of safer therapies and repeat dosing, making innovation a critical factor in market growth. The viral capsid development industry saw significant growth. In September 2025, VectorY Therapeutics secured an option and license agreement with Shape Therapeutics, granting rights to the AAV5-derived SHP-DB1 capsid for the development of vectorized antibodies targeting neurodegenerative diseases.
The level of M&A activity in the viral capsid development industry is moderate to high, driven by strong demand for advanced gene delivery technologies. Large pharmaceutical and biotech companies are acquiring specialized firms with proprietary capsid engineering platforms to strengthen their gene therapy capabilities. Strategic partnerships and licensing deals are also common, reflecting the need to access novel vector design technologies and accelerate pipeline development.

The impact of regulations on the viral capsid development industry is high, as capsids must meet strict safety, quality, and efficacy standards for gene therapy approval. While regulatory requirements increase development time and costs, they also drive innovation in safer designs, standardized manufacturing, and advanced quality control.
Product expansion in the viral capsid development industry is growing, as companies develop engineered and synthetic capsids beyond traditional serotypes. These next-generation capsids offer improved targeting, immune evasion, and delivery efficiency, supporting wider gene therapy applications.
Regional expansion in the viral capsid development industry is growing, led by North America and Europe due to strong R&D and regulatory support, while the Asia-Pacific is emerging rapidly with increasing biotech investments and manufacturing capabilities.
Type Insights
The adeno-associated virus (AAV) capsid segment led the market with the largest revenue share of 40.91% in 2025, and is expected to grow at the fastest CAGR throughout the forecast period. This dominance is driven by the widespread use of AAV vectors in gene therapy due to their strong safety profile, low immunogenicity, and ability to enable long-term gene expression. Advancements in capsid engineering, rising approvals for therapies, increasing investments in rare disease treatments, and expanding manufacturing capabilities are further driving segment growth.
The lentiviral segment is rapidly growing due to its strong ability to deliver genes into both dividing and non-dividing cells, making it highly suitable for cell and gene therapies. Moreover, increasing use in CAR-T cell therapy, rising clinical research activities, and advancements in vector safety and production technologies are further accelerating segment growth.
Application Insights
The gene therapy segment led the market with the largest revenue share of 44.87% in 2025 and is expected to grow at the fastest CAGR throughout the forecast period, driven by the increasing adoption of viral vectors for treating genetic and rare diseases. The rising number of clinical trials, growing approvals of gene therapies, advancements in capsid engineering, and strong investments in gene-based treatment development are further driving segment growth.
The vaccine development segment is experiencing significant growth due to the increasing use of viral vectors in next-generation vaccines, particularly for infectious diseases and emerging pathogens. Rising vaccine R&D investments, advancements in capsid engineering, and increasing demand for rapid, scalable vaccine platforms are driving segment growth.
End-use Insights
The pharmaceutical and biotechnology companies segment led the market with the largest revenue share of 50.83% in 2025, and is expected to grow at the fastest CAGR from 2026 to 2033, due to increasing investments in gene and cell therapy research, strong involvement in viral vector development, expanding clinical pipelines, and growing collaborations with contract development and manufacturing organizations (CDMOs).
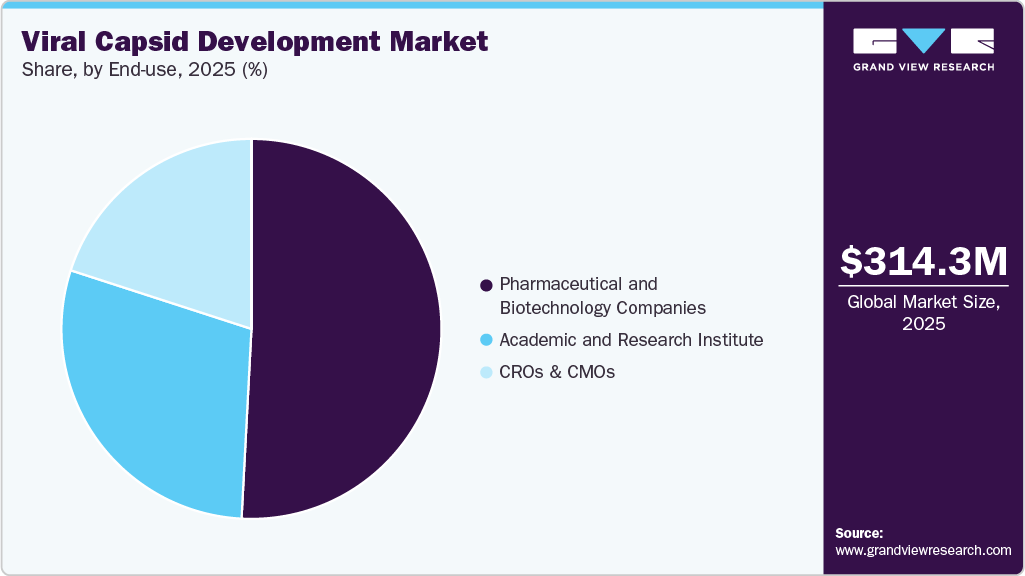
The academic and research institute segment is expected to register at a significant CAGR over the forecast period, driven by increasing research on viral vectors, rising funding for genetic studies, growing collaborations with biotech and pharmaceutical companies, and the adoption of advanced capsid engineering technologies.
Regional Insights
North America dominated the global viral capsid development market with the largest revenue share of 48.31% in 2025. The region hosts a high concentration of biotechnology firms, academic research centers, and CDMOs specializing in AAV and lentiviral capsid design. Strong venture capital investment and public funding support rapid progress from discovery to clinical optimization.
U.S Viral Capsid Development Market Trends
The viral capsid development market in the U.S. accounted for the largest market revenue share in North America in 2025. The market is driven by strong gene therapy pipelines, advanced vector engineering companies, and extensive clinical research activity. Significant public funding, venture capital investment, and large-scale GMP manufacturing capabilities drive continuous innovation in capsid optimization and commercialization.
Europe Viral Capsid Development Market Trends
The viral capsid development market in Europe is shaped by strong academic leadership and collaborative research networks. The region establishes itself as a primary center for early-stage capsid development through its funding partnerships and international research programs, which pursue precision bioengineering to enhance tropism and reduce immunogenicity in rare disease therapies.
The UK viral capsid development market is anticipated to grow at a significant CAGR during the forecast period. The UK is a major center for early-stage capsid innovation, driven by strong academic research and collaborative gene therapy programs. Government funding and university-industry partnerships support advancements in capsid engineering technologies and translational research.
The viral capsid development market in Germany plays a key role through its strong biomanufacturing expertise and engineering-focused research. The country emphasizes scalable viral vector production and precision capsid optimization for commercial applications.
Asia Pacific Viral Capsid Development Market Trends
The viral capsid development market in the Asia Pacific is expected to grow at the fastest CAGR of 13.59% throughout the forecast period. The region is rapidly emerging as a hub for viral capsid development, supported by growing biotechnology investments and expanding gene therapy pipelines. Government initiatives, rising clinical trial activity, and global partnerships are strengthening regional capabilities. Local firms are prioritizing scalable production and cost-efficient capsid engineering to meet increasing therapeutic demand.
The China viral capsid development market is rapidly expanding due to strong government investment, growing gene therapy pipelines, and increasing domestic vector manufacturing capabilities. The market is driven by rising clinical trials and biotech innovation.
The viral capsid development market in Japan is driven by government support for regenerative medicine and advanced molecular engineering capabilities. The country focuses on developing highly targeted capsid platforms for rare and neurological diseases.
Middle East & Africa Viral Capsid Development Market Trends
The viral capsid development market in the Middle East & Africa is still nascent but is gradually advancing as biomedical research and interest in advanced therapeutics expand. Growth is concentrated in select countries investing in genomic infrastructure and biotech ecosystems, supported by international collaborations and technology transfer.
The Kuwait viral capsid development market remains at an early stage, with activity focused on academic research and healthcare infrastructure development. Growth is supported mainly by international collaborations and gradual investment in biotechnology capabilities.
Key Viral Capsid Development Company Insights
The viral capsid development industry features a mix of global biopharmaceutical firms and specialized biotech companies competing through proprietary vector engineering, strong clinical pipelines, and application-specific viral capsid solutions.
Key players such as PackGene Biotech Inc., Sartorius AG, Revvity, VectorBuilder, and AAVnerGene maintain strong positions due to advanced capsid platforms, extensive R&D capabilities, and scalable manufacturing solutions, while companies like AskBio Inc., Voyager Therapeutics, Capsigen, and uniQure N.V. are expanding through strategic collaborations, clinical-stage pipeline diversification, and innovative gene and cell therapy applications.

Market growth is increasingly driven by the expansion of gene therapy and vaccine applications, regional market penetration, and strategic partnerships across the biotech and pharmaceutical sectors.
Key Viral Capsid Development Companies:
The following key companies have been profiled for this study on the viral capsid development market.
- PackGene Biotech Inc.
- Sartorius AG
- Revvity
- VectorBuilder
- AAVnerGene
- AskBio Inc.
- Voyager Therapeutics
- uniQure N.V.
- Vigene Biosciences
- Capsigen
Recent Developments
-
In July 2025, in Japan, JCR Pharmaceuticals signed a license agreement with Alexion, granting rights to its JUST-AAV capsid platform for up to five genomic medicine programs, including milestone and royalty provisions.
-
In September 2024, in the U.S., Asimov launched the AAV Edge System, an end-to-end AI-driven platform combining capsid engineering, host cells, and genetic tools to enhance gene therapy design, production, and manufacturability.
Viral Capsid Development Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 343.3 million
Revenue forecast in 2033
USD 684.5 million
Growth rate
CAGR of 10.36% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
PackGene Biotech Inc.; Sartorius AG; Revvity; VectorBuilder; AAVnerGene; AskBio Inc.; Voyager Therapeutics; uniQure N.V.; Vigene Biosciences; Capsigen.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Viral Capsid Development Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global viral capsid development market report based on the type, application, end-use, and region.
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Adeno-associated Virus (AAV) Capsids
-
Adenoviral
-
Lentiviral
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Gene Therapy
-
Vaccine Development
-
Cancer Therapy
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biotechnology Companies
-
Academic and Research Institute
-
CROs & CMOs
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global viral capsid development market size was estimated at USD 314.3 million in 2025 and is expected to reach USD 343.3 million in 2026
b. The global viral capsid development market is expected to grow at a compound annual growth rate of 10.36% from 2026 to 2033 to reach USD 684.5 million by 2033.
b. North America led the viral capsid development industry in 2025 with a share of 48.31%. The region hosts a high concentration of biotechnology firms, academic research centers, and CDMOs specializing in AAV and lentiviral capsid design. Strong venture capital investment and public funding support rapid progress from discovery to clinical optimization.
b. Some key players operating in the viral capsid development market include PackGene Biotech lnc., Sartorius AG, Revvity, VectorBuilder, AAVnerGene, AskBio Inc., Voyager Therapeutics, uniQure N.V., Vigene Biosciences, Capsigen.
b. Growth is driven by increasing demand for gene therapies, rising viral vector R&D, and advancements in capsid engineering, though high costs and manufacturing complexities may limit expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









