- Home
- »
- Clinical Diagnostics
- »
-
U.S. Latent Tuberculosis Infection Detection Market 2033GVR Report cover
![U.S. Latent Tuberculosis Infection Detection Market Size, Share & Trends Report]()
U.S. Latent Tuberculosis Infection Detection Market (2026 - 2033) Size, Share & Trends Analysis Report By Test (IGRA, TST), By Application (Household Contacts With Pulmonary TB, PLHIV), By End-use (Diagnostic Laboratories, Hospitals/Clinics), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-861-8
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Latent Tuberculosis Infection Detection Market Summary
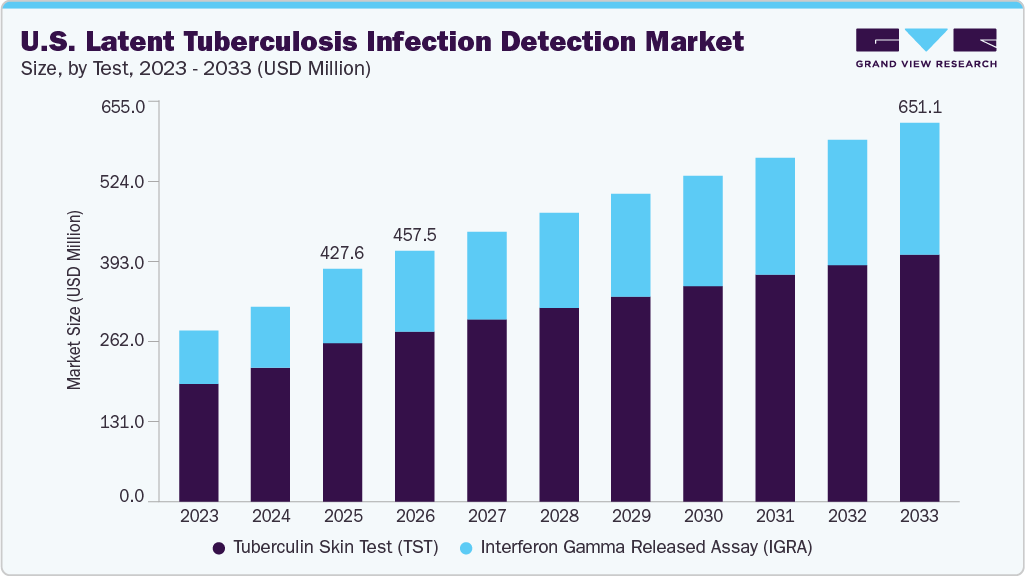
The U.S. latent tuberculosis infection detection market size was estimated at USD 427.61 million in 2025 and is projected to reach USD 651.11 million by 2033, growing at a CAGR of 5.17% from 2026 to 2033. This growth is driven by an increasing risk of developing active tuberculosis infection from latent tuberculosis infection (LTBI), growing funding programs to promote TB diagnosis, aided by technological advancements in diagnostic methods.
Key Market Trends & Insights
- By test, tuberculin skin test (TST) dominated the market and accounted for the largest revenue share of 67.48% in 2025.
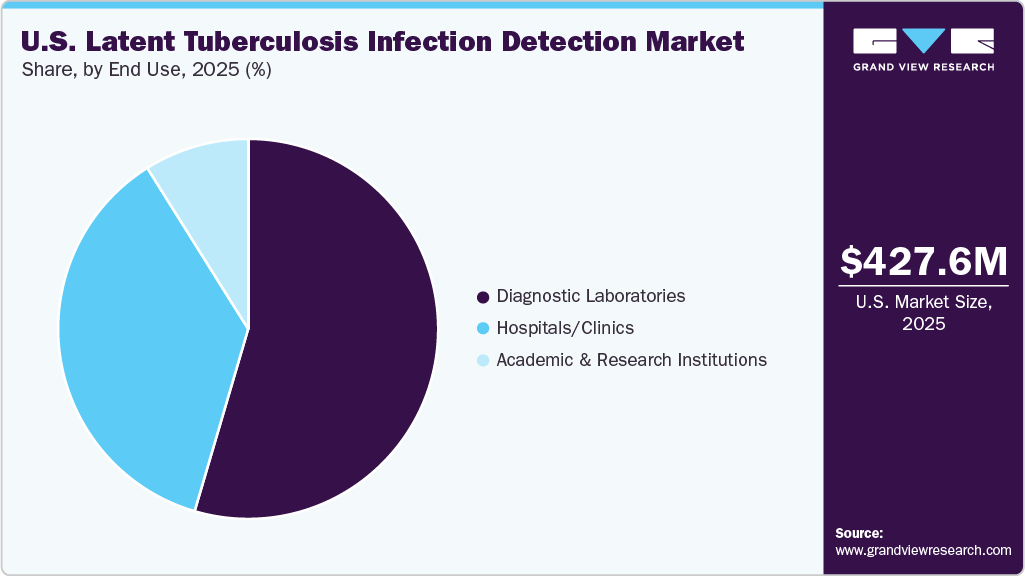
- By end use, diagnostic laboratories led the market, accounting for 54.55% of the revenue share in 2025
Market Size & Forecast
- 2025 Market Size: USD 427.61 Million
- 2033 Projected Market Size: USD 651.11 Million
- CAGR (2026-2033): 5.17%
According to estimates from the Centers for Disease Control and Prevention (CDC), up to 13 million people in the U.S. are believed to have LTBI, and without preventive treatment, about 5 %-10 % of those infected will develop active TB at some point in their lives.
Moreover, the growing focus on tuberculosis (TB) elimination and preventive care is creating a strong growth opportunity for the LTBI detection market. Public health authorities increasingly recognize that most active TB cases in the U.S. result from reactivation of latent infection rather than recent transmission. As a result, national TB elimination strategies have shifted toward identifying and treating LTBI before it progresses to active disease, placing greater emphasis on early and accurate diagnostic testing.
The risk of incident TB is influenced by various factors, such as age, underlying health conditions, and exposure settings, with older individuals and those in certain environments facing higher risks. Hence, managing LTBI through testing and treatment is crucial in reducing the risk of developing active TB and preventing the spread of the disease. Factors contributing to increased susceptibility to active TB from LTBI include:
-
Immunocompromising conditions, particularly HIV infection, significantly magnify the risk.
-
Concurrent ailments such as chronic renal failure, organ transplant history, patients under immunosuppressive therapy, and individuals afflicted with silicosis.
-
Lifestyle elements include tobacco use, alcohol dependency, inadequate nutrition, and being underweight.
-
Proximity to individuals with active TB, travel or residency in regions of high TB prevalence, and inhabiting congested or high-risk settings, such as medical facilities, correctional facilities, or refugee settlements.
Certain groups, such as people with weakened immune systems (including those with HIV or on immunosuppressive therapies), recent contacts of active TB cases, individuals born in countries with high TB prevalence, and people experiencing homelessness or incarceration, face elevated risks for LTBI progression. These risk factors increase both the urgency and frequency of screening efforts, driving uptake of more accurate and accessible testing technologies across clinical settings. Since most LTBI cases are asymptomatic and individuals with LTBI do not feel sick or spread TB, routine screening of at-risk groups is essential for early intervention.
Technological advancements in diagnostic methods are a major driver of LTBI detection market growth, as they significantly enhance diagnostic accuracy, laboratory efficiency, and patient compliance. Conventional tuberculin skin tests (TST) have notable limitations, including lower specificity in BCG-vaccinated populations and the requirement for multiple patient visits. In contrast, modern diagnostic technologies provide faster, more reliable, and scalable solutions, accelerating their adoption across public health programs, hospitals, and reference laboratories in the U.S.
Comparison of LTBI diagnostic methods in the U.S.
Parameter
Tuberculin Skin Test (TST)
Interferon-Gamma Release Assay (IGRA)
Test Type
Intradermal skin test
Blood-based laboratory test
Patient Visits Required
Two (administration + reading)
One visit (blood draw)
Result Interpretation
Subjective measurement of induration
Objective laboratory measurement
Impact of BCG Vaccination
May cause false positives
No cross-reactivity
Sensitivity in Immunocompromised Patients
Reduced
Moderately improved vs TST
Turnaround Time
48-72 hours
~24 hours (lab dependent)
Infrastructure Needs
Minimal
CLIA-certified lab & instrumentation
Cost
Low
Higher than TST
Use Settings
Public health, clinics, occupational screening
Hospitals, reference labs, targeted screening
Key Advantage
Low cost & accessibility
Higher specificity & single-visit workflow
Source: National Society of Tuberculosis Clinicians, Testing and Treatment of Latent Tuberculosis Infection in the U.S.: Clinical Recommendations, 2024
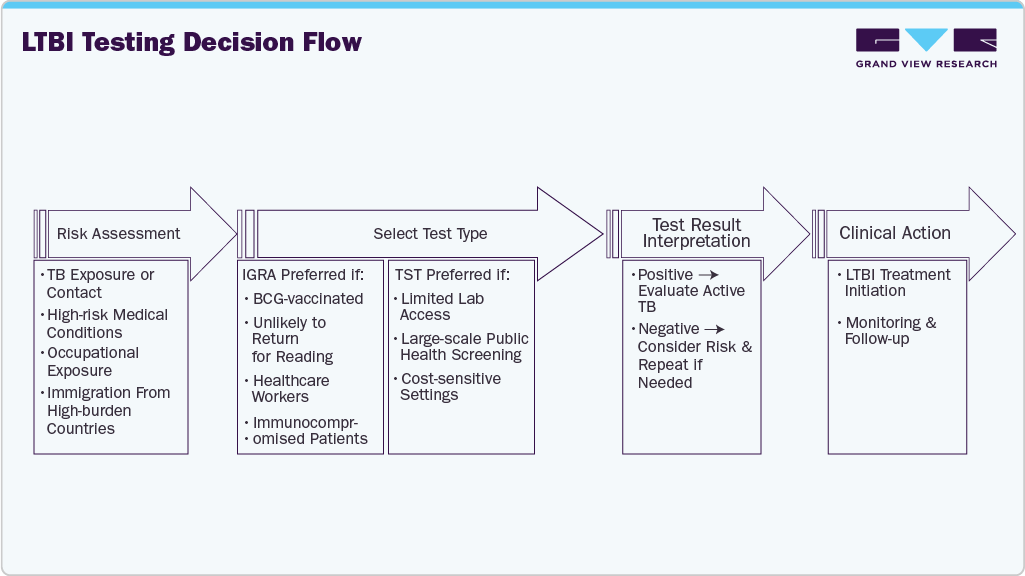
Moreover, the growing focus on tuberculosis (TB) elimination and preventive care is creating a strong market growth opportunity for the U.S. Public health authorities increasingly recognize that most active TB cases in the U.S. result from reactivation of latent infection rather than recent transmission. As a result, national TB elimination strategies have shifted toward identifying and treating LTBI before it progresses to active disease, placing greater emphasis on early and accurate diagnostic testing. This preventive focus is reflected in updated screening guidelines and targeted testing initiatives. For instance, the Centers for Disease Control and Prevention (CDC) and the U.S. Preventive Services Task Force (USPSTF) recommend LTBI screening for individuals at increased risk, including immigrants from high TB-burden countries, healthcare workers, people living with HIV, and patients receiving immunosuppressive therapies such as TNF-alpha inhibitors. These recommendations have driven routine LTBI testing in primary care, occupational health, and specialty clinics, directly increasing demand for latent TB diagnostic tests across healthcare settings.
LTBI Testing Decision Flow
However, limited awareness and the resulting underdiagnosis of latent tuberculosis infection (LTBI) represent a significant restraint on the U.S. LTBI detection market. Unlike active TB, latent TB is asymptomatic and does not cause immediate illness, which leads many individuals to underestimate the importance of screening. As a result, testing is often pursued only when required for employment, immigration, or specific medical conditions, rather than as a routine preventive measure. This low level of public awareness reduces voluntary testing rates and limits the overall demand for LTBI diagnostic services.
Market Concentration & Characteristics
The degree of innovation in the U.S. latent tuberculosis infection (LTBI) detection market is moderate and has accelerated in response to the need for faster, more scalable, and highly accurate detection to support TB elimination goals. Molecular diagnostics, interferon-gamma release assays (IGRAs), and next-generation sequencing (NGS) are increasingly shaping the future of LTBI detection. These innovations support earlier identification, better surveillance, and more informed clinical decision-making.
Mergers and acquisitions in the LTBI detection market have increasingly been focused on expanding laboratory networks, strengthening specialty testing capabilities, and improving access to high-quality screening services. Strategic transactions are enabling major laboratory service providers to broaden their reach, integrate advanced testing platforms, and enhance service delivery for TB screening programs across hospital systems, public health departments, and employer-based screening initiatives.
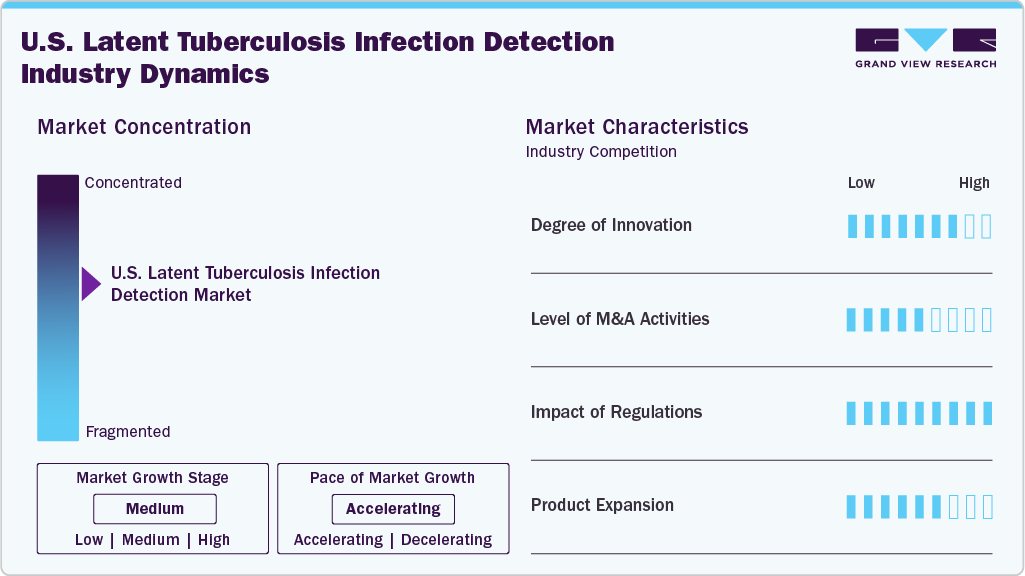
Regulatory oversight plays a critical role in shaping the adoption of LTBI diagnostics in the United States. FDA clearance requirements ensure high standards of clinical performance, reliability, and reproducibility for TB diagnostic assays and automated platforms. In parallel, guidance from the Centers for Disease Control and Prevention (CDC) and the U.S. Preventive Services Task Force (USPSTF) supports targeted LTBI screening among high-risk populations, influencing testing volumes and reimbursement coverage.
Diagnostic manufacturers continue to expand TB testing portfolios through enhanced IGRA assays, automation solutions, and workflow optimization tools designed to improve throughput and turnaround time. Product enhancements emphasize improved sensitivity, simplified laboratory workflows, and compatibility with automated liquid handling and laboratory information systems.
Test Insights
Tuberculin skin test (TST) dominated the market and accounted for the largest revenue share of 67.48% in 2025. The dominance of the segment is attributed to its widespread use as a reliable and cost-effective method for screening latent tuberculosis infection (LTBI). The test involves intradermal injection of tuberculin purified protein derivative (PPD) into the inner surface of the skin using the Mantoux method, after which the injection site is evaluated 48-72 hours later for induration. In individuals with no known risk factors, an induration of 15 mm or greater is typically considered positive. The TST remains highly penetrated across public health programs, occupational screening, and routine clinical practice due to its affordability, established clinical acceptance, and ease of deployment.
Interferon Gamma Released Assay (IGRA) is projected to experience the fastest CAGR of 6.39% during the forecast period. This growth is driven by the increasing adoption of blood-based testing for latent tuberculosis infection (LTBI), supported by its clinical and operational advantages over the tuberculin skin test (TST). IGRAs detect infection by measuring interferon-gamma released by sensitized T cells in response to TB-specific antigens, enabling objective, laboratory-based interpretation. In the U.S., these tests are favored because they require only a single patient visit for blood collection, improving compliance in outpatient care, occupational health screening, and public health contact investigations where follow-up visits can be challenging.
Application Insights
People living with HIV (PLHIV) segment accounted for the largest market share of 42.85% in 2025, attributed to their substantially elevated risk of progression from latent infection to active TB disease. HIV infection is recognized by the CDC as one of the strongest risk factors for TB activation, and U.S. clinical guidelines recommend routine TB screening as part of comprehensive HIV care. Even in low-incidence settings, TB remains an important opportunistic infection among PLHIV, particularly in individuals with additional risk factors such as foreign birth or immunosuppressive therapy. Both the tuberculin skin test (TST) and interferon-gamma release assays (IGRAs) are used for LTBI screening; however, immune suppression may reduce test sensitivity. For PLHIV, a TST induration of ≥5 mm is considered positive. IGRAs are often preferred in HIV care settings due to higher specificity and operational convenience. Ongoing risk-based screening and integration of LTBI management into HIV programs continue to sustain testing demand in this high-risk population.
Household contacts with pulmonary TB is likely to grow at the fastest CAGR of 6.19% over the forecast period. In the U.S., household contacts of individuals with infectious pulmonary tuberculosis represent one of the highest-priority populations for TB contact investigations and latent tuberculosis infection (LTBI) screening. Close and prolonged exposure significantly increases the risk of TB transmission, and U.S. public health authorities consistently prioritize this group for early identification and preventive treatment. According to the U.S. Centers for Disease Control and Prevention (CDC), targeted testing of close contacts is a cornerstone of TB control efforts, as untreated LTBI in this population carries a measurable risk of progression to active disease, particularly within the first two years following exposure.
End Use Insights
Diagnostic laboratories led the LTBI detection market, accounting for 54.55% of the revenue share in 2025, owing to efforts to improve patient outcomes by providing diagnostic facilities at the retail level. Moreover, the ability of laboratories to handle a large volume of tests at an expedited rate is expected to further fuel the segment over the forecast period. Furthermore, diagnostic laboratories also deal with samples received from clinics and hospitals. Diagnostic laboratories are providing newer blood tests for the screening or detection of LTBIs. For instance, independent laboratories such as Dynacare and Life Labs provide tests, such as QuantiFERON-TB blood tests and T-SPOT.TB tests, for the detection of LTBI, are available at comparatively lower prices. In addition, in March 2023, the U.S. Food and Drug Administration approved two additional cell isolation instruments from PerkinElmer’s Oxford Immunotec for use with the T-Cell Select™ reagent kit, aiding in the diagnosis of tuberculosis (TB) in vitro. This approval enhances the automation of TB detection, reducing time and labor for laboratories and providing faster and more reliable results for patients.
The hospitals/clinics segment is expected to grow at a lucrative rate over the forecast period. Individuals with medical conditions, such as HIV/AIDS, chronic renal failure, and other diseases that weaken the immune system, are at increased risk of LTBI and its development into an active TB case. The increasing prevalence of diseases that affect the immune system is likely to boost the testing rate for LTBIs in hospitals and clinics. The penetration of TST is higher in hospitals and clinics, as it requires standardization of procedures and trained professionals to conduct tests & read the skin test reaction. Moreover, TST does not require specific equipment and reagent kits to detect latent tuberculosis infection. In addition, LTBI detection market players, such as bioMérieux, are working to equip hospital laboratories with the newer blood test for the detection of LTBI.
Key U.S. Latent Tuberculosis Infection Detection Company Insights
The competitive scenario in the LTBI detection market is high, with key players such as QIAGEN, Euroimmun US, bioMérieux, and Oxford Immunotec holding significant positions. Prominent market participants are focusing on increasing their customer base using acquisition strategies.
Key U.S. Latent Tuberculosis Infection Detection Companies:
- QIAGEN
- Euroimmun US
- bioMérieux
- Oxford Immunotec
- SD Biosensor, INC.
- Quest Diagnostics
- LabCorp
- ARUP Laboratories
- Mayo Clinic Laboratories
- BioReference Laboratories
- Par Pharmaceutical, Inc.
Recent Developments
-
In April 2025, Revvity announced the U.S. FDA approval of the Auto-Pure 2400 liquid handling platform with the T-SPOT.TB test. The product was initially introduced outside the U.S. in 2024. This integrated solution enables laboratories to enhance productivity while maintaining high clinical performance in latent tuberculosis (TB) detection. The approval represents an advancement in TB control, offering a faster, high-throughput testing solution that delivers accurate results to support timely treatment and disease containment in the U.S.
-
In March 2025, BioReference/OPKO Health entered into an agreement for Labcorp to acquire selected assets of BioReference’s clinical testing businesses, expanding clinical laboratory access and service footprint. As part of the agreement, Labcorp will take over BioReference Health’s U.S.-based oncology and oncology-related laboratory testing operations, along with associated customer relationships and select operational assets.
-
In January 2025, SD Biosensor’s latent TB diagnostic product STANDARD E TB-Feron ELISA was officially included in the WHO’s list of recommended TB diagnostics following a Pathway B evaluation, affirming performance comparable to existing WHO-recommended tests.
U.S. Latent Tuberculosis Infection Detection Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 457.51 million
Revenue forecast in 2033
USD 651.11 million
Growth rate
CAGR of 5.17% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, volume (number of tests) in thousands, and CAGR from 2026 to 2033
Report coverage
Revenue & volume forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test, application, end use
Country scope
U.S.
Key companies profiled
QIAGEN; Euroimmun US; bioMérieux; Oxford Immunotec; SD Biosensor, INC.; Quest Diagnostics; LabCorp; ARUP Laboratories; Mayo Clinic Laboratories; BioReference Laboratories; Par Pharmaceutical, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Latent Tuberculosis Infection Detection Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. latent tuberculosis infection detection market report based on test, application, and end use:
-
Test Outlook (Revenue, USD Million, 2021 - 2033) (Volume, Number of Tests in Thousands)
-
Interferon Gamma Released Assay (IGRA)
-
QFT
-
T.SPOT
-
-
Tuberculin Skin Test (TST)
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033) (Volume, Number of Tests in Thousands)
-
Household Contacts With Pulmonary TB
-
PLHIV
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033) (Volume, Number of Tests in Thousands)
-
Diagnostic Laboratories
-
Hospitals/Clinics
-
Academic & Research Institutions
-
Frequently Asked Questions About This Report
b. The global U.S. latent tuberculosis infection detection market size was estimated at USD 427.61 million in 2025 and is expected to reach USD 457.51 million in 2026.
b. The global U.S. latent tuberculosis infection detection market is expected to grow at a compound annual growth rate of 5.17% from 2026 to 2033 to reach USD 651.11 million by 2033.
b. People living with HIV (PLHIV) segment accounted for largest market share of 42.85% in 2025. This is attributed to their substantially elevated risk of progression from latent infection to active TB disease.
b. Some key players operating in the U.S. latent tuberculosis infection detection market include QIAGEN, Euroimmun US, bioMérieux, Oxford Immunotec, SD Biosensor, INC., Quest Diagnostics , LabCorp, ARUP Laboratories, Mayo Clinic Laboratories, BioReference Laboratories, Par Pharmaceutical, Inc.
b. Key factors that are driving the market growth include an increasing risk of developing active tuberculosis infection from LTBI, growing funding programs to promote TB diagnosis aided with technological advancements in diagnostic methods.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









