- Home
- »
- Healthcare IT
- »
-
U.S. AI-driven Diabetic Retinopathy Screening Market Report, 2033GVR Report cover
![U.S. AI-driven Diabetic Retinopathy Screening Market Size, Share & Trends Report]()
U.S. AI-driven Diabetic Retinopathy Screening Market (2026 - 2033) Size, Share & Trends Analysis Report By Component (Software, Hardware, Services), By Screening (Autonomous AI Screening, AI-Assisted Screening), By Deployment Mode, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-849-6
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. AI-driven Diabetic Retinopathy Screening Market Summary
The U.S. AI-driven diabetic retinopathy screening market size was estimated at USD 190.01 million in 2025 and is projected to reach USD 881.74 million by 2033, growing at a CAGR of 21.18% from 2026 to 2033. Rising prevalence of diabetes, favorable reimbursement pathways, and shortage of ophthalmologists and access gaps are significant factors contributing to market growth.
Key Market Trends & Insights
- By component, the software segment held the largest market share of 45.97% in 2025.
- By deployment mode, the cloud-based segment held the largest market share of over 57% in 2025.
- By screening, the autonomous AI screening segment held the highest market share of over 47% in 2025.
- By end use, the primary care settings segment held the largest market share of over 40% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 190.01 Million
- 2033 Projected Market Size: USD 881.74 Million
- CAGR (2026-2033): 21.18%
The country faces a growing public health challenge from diabetes, thereby increasing the population at risk of diabetic retinopathy. For instance, according to the data published by the U.S. Centers for Disease Control and Prevention in May 2024, around 38.4 million people were affected by diabetes, accounting for 11.6% of the total U.S. population. Furthermore, the American Academy of Ophthalmology reports that nearly 60 percent of individuals with diabetes do not attend their recommended annual dilated eye examinations, despite established clinical guidelines. This discrepancy between recommended care and actual adherence substantially elevates the risk of undiagnosed disease progression and preventable vision loss.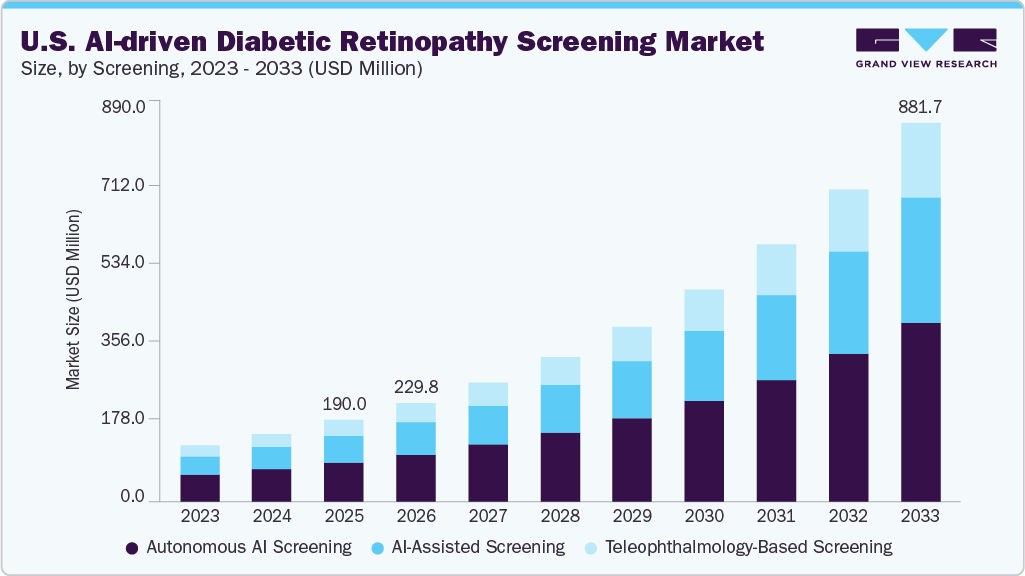
Diabetic patients are commonly managed in primary care or endocrinology settings, where retinal screening is frequently unavailable. As the prevalence of diabetes increases, the demand for annual eye examinations surpasses the capacity of available specialists. This results in a screening burden that conventional healthcare systems cannot address efficiently. Artificial intelligence-enabled diabetic retinopathy screening systems provide scalable, point-of-care solutions that do not require immediate specialist intervention. AI addresses detection gaps through autonomous and rapid diagnostics. Moreover, primary care integration expands access beyond specialists, enabling early intervention to prevent vision loss and comorbidities. For instance, in July 2023, Mount Sinai launched the Center for Ophthalmic Artificial Intelligence and Human Health, the first in New York, to advance AI in ophthalmology for timely diagnosis of macular degeneration, diabetic retinopathy, glaucoma, hypertensive retinopathy, and retinal tumors. Partnering with the Windreich Department of AI and Human Health, it targets tele-retina, tele-ophthalmology, and eye stroke services using validated AI models.
Furthermore, in 2021, AI-driven eye disease diagnosis advanced significantly with the introduction of a new reimbursement code for AI-based diabetic retinopathy screening in the U.S. Medicare reimbursement accelerated the adoption of AI-based diabetic retinopathy screening in the country through CPT 92229, the first AI-specific code allowing primary care billing without specialist oversight. For instance, LumineticsCore (Digital Diagnostics), EyeArt (Eyenuk), and AEYE-DS (AEYE Health) have each received coverage as autonomous diagnostic systems. By authorizing reimbursement without direct physician interpretation, the Centers for Medicare & Medicaid Services (CMS) has recognized AI as a reimbursable clinical service rather than an experimental adjunct. These policy changes support workflow decentralization and enable screening at the point of care during routine diabetes visits. As a result, providers are more willing to invest in AI-enabled retinal imaging systems, since these services generate predictable revenue and advance quality care objectives.
Market Concentration & Characteristics
The chart below illustrates the relationship between industry concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, level of partnerships & collaboration activities, degree of innovation, impact of regulations, and regional expansion. The U.S. AI-driven diabetic retinopathy screening industry is slightly consolidated. However, several emerging players are entering the market, thereby contributing to market fragmentation. The degree of innovation and the impact of regulations is high. However, the level of partnerships & collaborations and the regional expansion of industry is moderate.
The U.S. AI-driven diabetic retinopathy screening industry is characterized by constant innovation, with a strong focus on launching new software and devices. For instance, in June 2024, AEYE Health and Optomed launched Aurora AEYE, the first FDA-cleared portable handheld fundus camera with autonomous AI for on-the-spot diabetic retinopathy screening.
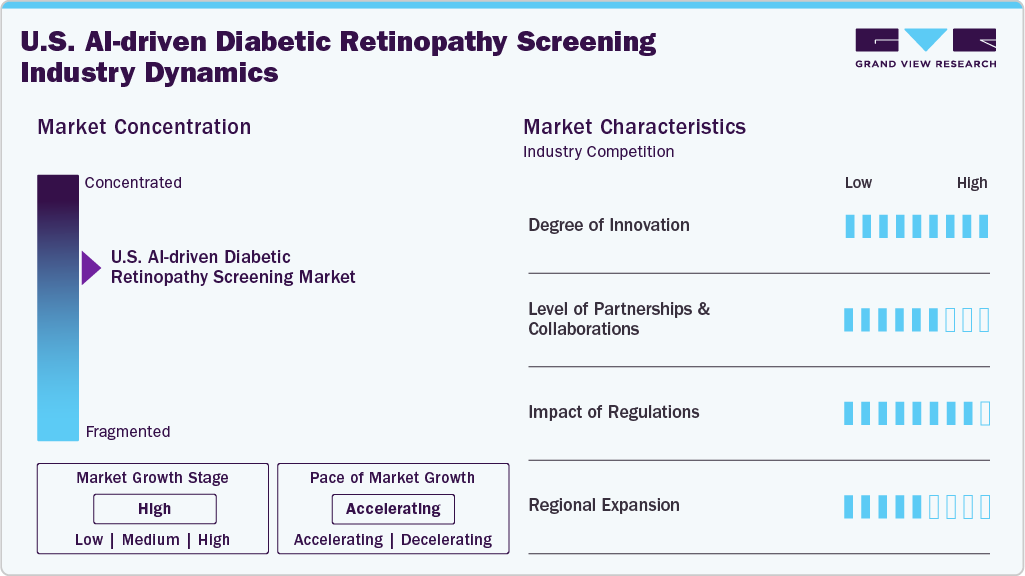
The industry is experiencing a high level of partnerships & collaborations undertaken by several key players. This is due to the desire to gain a competitive advantage in the industry, enhance technological capabilities, and consolidate in a rapidly growing market.
Regulations, such as the HIPAA in the U.S. establish standards for safeguarding patient data privacy and security. Compliance with these regulations is crucial for AI applications in healthcare to ensure the safe and secure handling of patient information, reducing the risk of data breaches and unauthorized access.
The industry is witnessing moderate geographical expansion. Companies within the AI-driven diabetic retinopathy screening industry seek geographic expansion strategies to maintain their foothold in the market. For instance, in August 2024, Eyenuk partnered with the American Academy of Ophthalmology's EyeCare America to deploy EyeArt AI screening in Delaware Federally Qualified Health Centers (FQHCs) for underserved patients with diabetes. Primary care staff operate camera-integrated AI for autonomous detection without specialists; positive cases are referred to volunteer ophthalmologists.
List of FDA Clearances in the U.S. AI-driven Diabetic Retinopathy Screening Industry
Company
Year
Month
Description
AEYE Health
2024
April
AEYE Health received first FDA clearance for fully autonomous AI (AEYE-DS) diagnosing referable diabetic retinopathy using Optomed Aurora handheld camera images.
AEYE Health
2022
November
AEye Health received FDA 510(k) clearance for AEYE-DS, an AI system detecting more-than-mild diabetic retinopathy in adults with diabetes (no prior retinopathy diagnosis).
Eyenuk, Inc.
2020
August
Eyenuk's EyeArt received FDA 510(k) clearance as the first autonomous AI system detecting more-than-mild (mtmDR) and vision-threatening diabetic retinopathy (vtDR) in adults with diabetes (no prior mtmDR diagnosis).
Digital Diagnostics Inc.
2018
April
FDA approved IDx-DR (now called as LumineticsCore) as the first autonomous AI diagnostic system for detecting diabetic retinopathy (including macular edema) in primary care settings.
Component Insights
Based on component, the software segment held the largest revenue share of 45.97% in 2025, enabling automated image analysis, disease classification, and referral decision support. FDA-cleared autonomous AI software detects referable diabetic retinopathy from retinal images without specialist interpretation, supporting scalable deployment in primary care and outpatient settings. These platforms integrate with fundus cameras, EHR systems, and practice management software to streamline workflows. Moreover, growth in this segment is driven by continuous algorithm improvement, expanding regulatory approvals, and favorable reimbursement frameworks. For instance, in August 2024, Thirona divested its spinout, Thirona Retina BV, to Revenio Group's subsidiary, Icare Finland Oy. With this devistuiture, Revenio Group's integrated RetCAD AI software for screening fundus images for diabetic retinopathy, AMD, and glaucoma is being added to iCare's diagnostics portfolio.
The services segment is expected to grow at the fastest CAGR during the forecast period. This segment supports implementation, optimization, and sustained clinical performance of AI solutions. It includes installation, system integration, staff training, workflow redesign, and compliance support for FDA-cleared screening programs. Service providers assist healthcare organizations in embedding AI screening into primary care, endocrinology, and teleophthalmology settings.
Deployment Mode Insights
Based on deployment mode, the cloud-based segment accounted for the largest revenue share of 57.31% in 2025 due to its scalability, rapid deployment, and centralized model management. Cloud platforms enable real-time image upload, automated AI analysis, and immediate clinical outputs across distributed care settings. They support seamless integration with EHRs, retinal cameras, and teleophthalmology networks.
The hybrid segment is anticipated to grow at the fastest CAGR over the forecast period. Hybrid architecture allows healthcare providers to maintain control over sensitive patient data while leveraging scalable AI models. Local systems enable screening during connectivity disruptions, while cloud layers support advanced analytics, model updates, and population-level reporting. Providers benefit from flexible deployment tailored to regulatory, operational, and bandwidth constraints. In addition, hybrid platforms facilitate integration with EHRs and teleophthalmology services across care networks, thereby contributing to segment growth.
Screening Insights
Based on screening, the autonomous AI screening segment accounted for the largest revenue share of 47.78% in 2025. These systems independently analyze retinal images and generate diagnostic output without clinician intervention at the point of care. This technology enables rapid screening, standardized grading, and consistent accuracy across high-volume environments, thereby directly addressing the shortage of ophthalmologists and retinal specialists. Moreover, autonomous AI screening also supports large-scale population health initiatives by enabling point-of-care screening in non-specialist settings. Integration with fundus cameras and EHR platforms allows immediate clinical decision support and automated referral pathways. The reduction in manual interpretation lowers operational costs and improves workflow efficiency, further contributing to the segment’s growth.
The teleophthalmology-based screening segment is projected to grow at the fastest CAGR from 2026 to 2033. Teleophthalmology-based screening supports collaborative care pathways by integrating AI outputs with remote ophthalmologist validation and referral management. AI triage prioritizes high-risk cases, optimizing specialist time and reducing unnecessary referrals. The model aligns with value-based care initiatives by lowering downstream treatment costs through earlier intervention. Favorable reimbursement for telehealth services further strengthens adoption.
End Use Insights
Based on end use, the primary care settings segment held the largest market share of 40.79% in 2025. Primary care practices serve as the first point of contact for most patients with diabetes, creating a healthcare setting for routine retinal screening. Adoption in primary care settings is further driven by reimbursement alignment and population health objectives. AI-driven screening supports quality measures linked to diabetes management and value-based care programs.
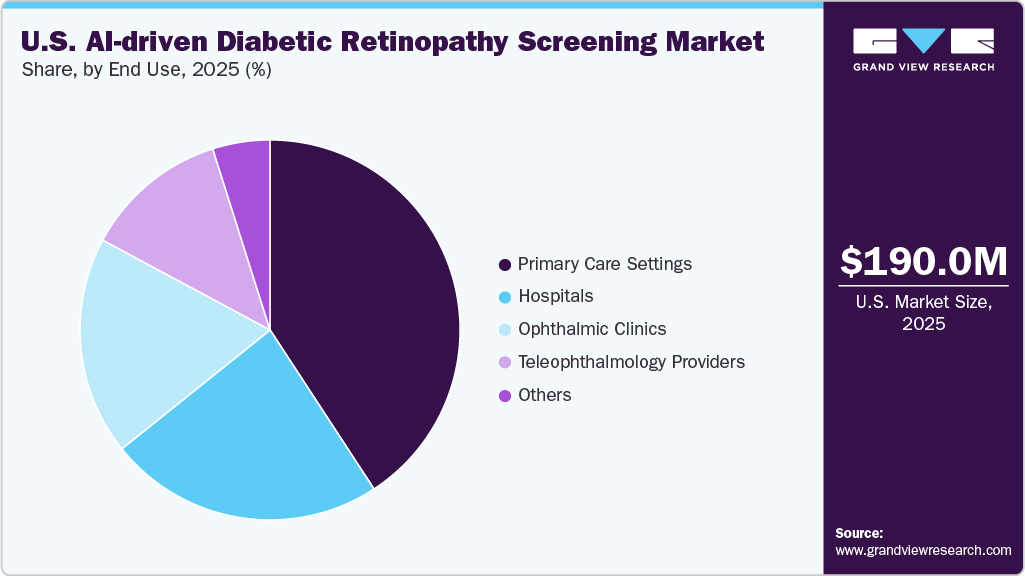
The teleophthalmology providers segment is anticipated to grow at the fastest CAGR from 2026 to 2033. These providers leverage remote retinal imaging combined with AI algorithms to deliver specialist-level screening without on-site ophthalmologists. This model is effective in rural, underserved, and resource-constrained settings. Moreover, growth in this segment is supported by the growing acceptance of telehealth and remote diagnostics for reimbursement.
Key U.S. AI-Driven Diabetic Retinopathy Screening Company Insights
Key players operating in the U.S. AI-driven diabetic retinopathy screening industry are undertaking various initiatives to strengthen their market presence and increase the reach of their products and services. Strategies such as new product launches and partnerships play a key role in propelling market growth.
Key U.S. AI-Driven Diabetic Retinopathy Screening Companies:
- Eyenuk, Inc.
- Digital Diagnostics Inc.
- AEYE Health.
- Optomed
- IRIS (Intelligent Retinal Imaging Systems)
- RETINA-AI Health, Inc.
- iCare
- RetinaRisk (by Risk Medical Solutions)
- BeamMed Inc.
Recent Developments
-
In July 2025, AEYE Health partnered with Ford Medical to distribute FDA-cleared AEYE-DS, a fully autonomous AI for diabetic retinopathy screening in pharmacies/non-acute centers.
-
In May 2024, Optomed USA launched Aurora AEYE, an FDA-cleared handheld AI fundus camera with AEYE Health for instant detection of more than mild diabetic retinopathy in primary care.
“The Optomed Aurora AEYE gives primary care providers the opportunity to bridge the diabetic retinopathy gap in the US with immediate, on-the-spot detection of diabetic retinopathy with results in about 60 seconds.”
-
In April 2024, Visionix USA and 20/20NOW launched an ocular telehealth program for diabetic retinopathy screening.
-
In April 2023, Digital Diagnostics launched LumineticsCore, the first De Novo FDA-cleared autonomous AI for diabetic retinopathy detection, in 9 Labcorp patient service centers in Alabama.
U.S. AI-driven Diabetic Retinopathy Screening Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 229.78 million
Revenue forecast in 2033
USD 881.74 million
Growth rate
CAGR of 21.18% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Component, deployment mode, screening, end use
Country scope
U.S.
Key companies profiled
Eyenuk, Inc.; Digital Diagnostics Inc.; AEYE Health.; Optomed; IRIS (Intelligent Retinal Imaging Systems); RETINA-AI Health, Inc.; iCare; RetinaRisk (by Risk Medical Solutions); BeamMed Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. AI-driven Diabetic Retinopathy Screening Market Report Segmentation
This report forecasts, revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented U.S. AI-driven diabetic retinopathy screening market report based on component, deployment mode, screening, and end use.
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Software
-
Hardware
-
Services
-
-
Deployment Mode Outlook (Revenue, USD Million, 2021 - 2033)
-
Cloud-Based
-
On-Premise
-
Hybrid
-
-
Screening Outlook (Revenue, USD Million, 2021 - 2033)
-
Autonomous AI Screening
-
AI-Assisted Screening
-
Teleophthalmology-Based Screening
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Primary Care Settings
-
Hospitals
-
Ophthalmic Clinics
-
Teleophthalmology Providers
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. AI-driven diabetic retinopathy screening market size was estimated at USD 190.01 million in 2025 and is expected to reach USD 229.78 million in 2026.
b. The U.S. AI-driven diabetic retinopathy screening market is expected to grow at a compound annual growth rate of 21.18% from 2026 to 2033 to reach USD 881.74 million by 2033.
b. The cloud-based segment held the largest market share of over 57% in 2025.
b. Some key players operating in the U.S. AI-driven diabetic retinopathy screening market include Eyenuk, Inc.; Digital Diagnostics Inc.; AEYE Health.; Optomed; IRIS (Intelligent Retinal Imaging Systems); RETINA-AI Health, Inc.; iCare; RetinaRisk (by Risk Medical Solutions); BeamMed Inc.
b. Key factors that are driving the U.S. AI-driven diabetic retinopathy screening market are rising prevalence diabetes, favorable reimbursement pathways, and shortage of ophthalmologists and access gaps are significant factors contributing to market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









