- Home
- »
- Medical Devices
- »
-
Sterile Injectables CDMO Market Size, Industry Report, 2033GVR Report cover
![Sterile Injectables CDMO Market Size, Share & Trends Report]()
Sterile Injectables CDMO Market (2026 - 2033) Size, Share & Trends Analysis Report By Molecule Type (Small Molecule, Large Molecule), By Product (API, Finished Drug Product), By Service, By Therapeutic Area, By Route Of Administration (Subcutaneous, Intravenous), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-363-5
- Number of Report Pages: 324
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Sterile Injectables CDMO Market Summary
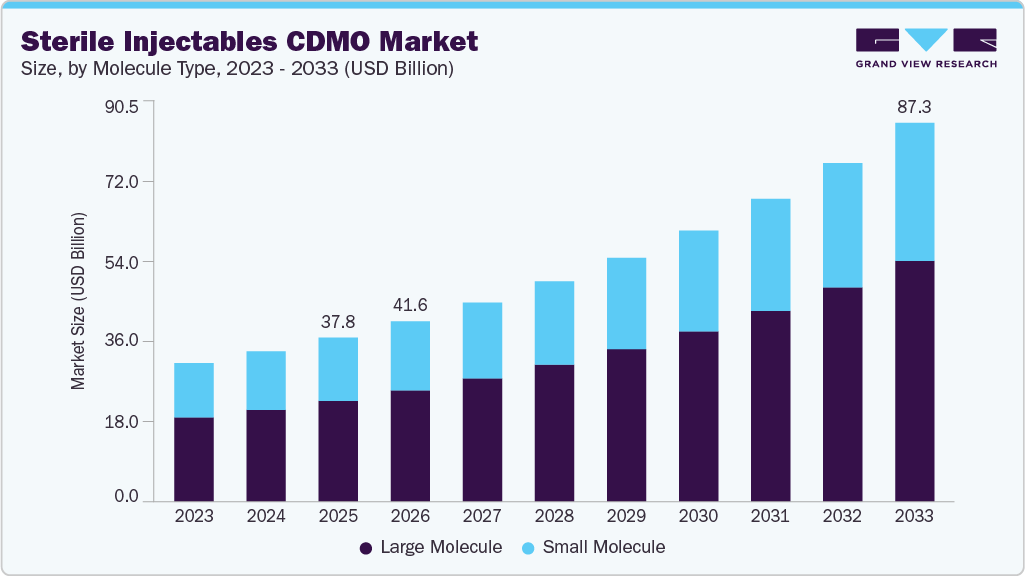
The global sterile injectables CDMO market size was estimated at USD 37.82 billion in 2025 and is projected to reach USD 87.34 billion by 2033, growing at a CAGR of 11.2% from 2026 to 2033. The market growth is driven by increasing drug development activities of biologics, biosimilars, and personalized medicines, drug approval processes, increasing complexity of therapeutics, and rising need for precise analytical validation and regulatory compliance.
Key Market Trends & Insights
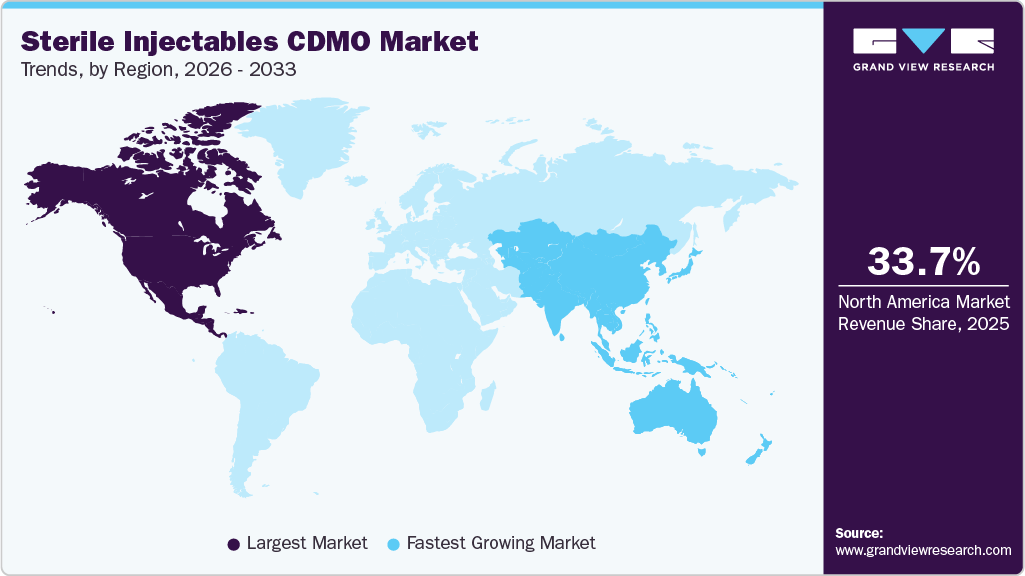
- North America held the largest market share in the sterile injectables CDMO market, accounting for 33.7% in 2025.
- U.S. is the largest market in North America and hold a substantial share by 2025.
- By molecule type, the large molecule segment dominated the sterile injectables CDMO market and accounted for 61.4% of global revenue in 2025.
- By product, the finished drug product segment accounted for the largest revenue share of 59.5% in 2025.
- By service, the manufacturing segment accounted for the largest revenue share of 57.1% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 37.82 Billion
- 2033 Projected Market Size: USD 87.34 Billion
- CAGR (2025-2030): 11.2%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, the increasing number of clinical trials, rising outsourcing of research activities by pharmaceutical and biotechnology companies to specialized contract research organizations (CROs), and ongoing technological advancements are further anticipated to fuel market growth. Furthermore, stringent regulatory requirements, expanding R&D investments, particularly in the emerging markets, and the growing emphasis on biomarker analysis and pharmacokinetic studies are expected to further accelerate market expansion.The increasing demand for cellular and genetic therapies is driving the market’s growth. With the expansion of therapy pipelines, there is a rising requirement for specialized manufacturing capabilities provided by contract manufacturing organizations (CMOs). For instance, according to ClinicalTrials.gov, as of March 2024, over 581 clinical trials were focused on cell therapies and related approaches for treating various diseases in North America is anticipated to drive an increase in contract manufacturing throughout the projected period.
Growth in markets of biosimilars, biologics, personalized medicine, orphan drugs, companion diagnostics, and adaptive trial designs, among others, is anticipated to boost the demand for sterile injectables CDMO. This surge is fueled by the high demand for biologics, complex manufacturing requirements, and regulatory support, which necessitate outsourcing to specialized CDMOs. These organizations provide the required expertise, infrastructure, and scalable manufacturing solutions that pharmaceutical companies need to reduce costs and accelerate time-to-market. Moreover, several companies are investigating new avenues, and the growing requirement to adhere to regulations is driving the demand for specialized service providers with proficiency in sterile injectables contract development and manufacturing affairs.
Furthermore, key initiatives undertaken by the companies in this sector are also expected to boost the market’s growth. For instance, in October 2025, Simtra BioPharma Solutions, a specialized CDMO focused on sterile injectable manufacturing, is advancing a two-continent expansion plan aimed at addressing rising global demand for complex, high-value therapies. Following its transition to an independent organization in 2023, the company has intensified investments across North America and Europe, strengthening its manufacturing footprint and expanding advanced capabilities. This strategic growth initiative underscores Simtra’s commitment to enhancing capacity, supporting customer pipelines, and delivering scalable, high-quality sterile solutions across key pharmaceutical markets.
The surge in global Alzheimer's disease cases, rising demand for cutting-edge gene therapies, a growing incidence of neurodegenerative diseases, and an increase in genetic disorders collectively propel the expansion of advanced therapies. A substantial number of major and small biotechnology companies are driving innovation in this field in terms of technology and manufacturing capabilities, thereby offering commercial opportunities for market expansion. Furthermore, the recent approval of new products in this market has further accelerated R&D activities in this marketplace.
Market Concentration & Characteristics
The sterile injectables CDMO market growth stage is medium, and the pace of market growth is accelerating. The sterile injectables CDMO market is characterized by a degree of innovation, Level of M&A activities, impact of regulations, service expansions, and regional expansions.
The sterile injectables CDMO market fortifies a high degree of innovation. Increasing focus on the adoption of advanced aseptic processing techniques that ensure the sterility of injectable products throughout the manufacturing process. This includes the use of isolators and restricted access barrier systems (RABS) to maintain aseptic conditions during filling and finishing operations. Additionally, advancements in lyophilization technology have improved the stability and shelf-life of injectable products, especially for biologics and complex molecules. High-containment manufacturing facilities have also emerged to address the growing demand for potent drug products, ensuring safe handling and production of hazardous substances.

Regulations have a high impact on the market, influencing compliance requirements, quality standards, and market entry barriers. Regulatory agencies impose rigorous guidelines to enhance safety, efficacy, and manufacturing processes, impacting the time and cost associated with novel biologics development and commercialization. Moreover, Continuous updates and amendments in regulatory guidelines require companies to stay informed and adaptable. Biopharmaceutical companies find it critical to track updated regulatory guidelines, so they outsource regulatory affairs services to comply with updated regulatory standards.
The level of M&A (mergers and acquisitions) activities in the market is high. Several companies are undertaking mergers and acquisitions strategies to expand biologics regulatory affairs portfolios, gain access to advanced technologies, and enhance their market presence. For instance, in January 2026, Alcami Corporation expands RTP biologics and analytical capacity with a new facility and added sterile fill-finish lines, strengthening large-molecule development support and increasing competitiveness in the sterile injectables CDMO market.
Service expansion in the market is medium owing to increasing demand for specialized regulatory expertise and comprehensive support services in the biologics sector. Moreover, service providers are expanding their regulatory consulting, compliance services, and end-to-end support for biologics development to cater to customer demand and broaden revenue growth opportunities. For instance, in February 2024, Pharmascience announced a USD 120 million expansion of its sterile injectable manufacturing facility last fall at its Candiac site. This expansion marked a crucial milestone for the company, demonstrating its commitment to enhancing its manufacturing capabilities in the pharmaceutical sector.
The market is experiencing significant regional expansion, with service providers strategically expanding their presence to new geographic areas. Biologics companies are expanding into emerging markets in Asia-Pacific, Latin America, and MEA owing to growing healthcare needs and favorable regulatory environments. Thus, growing contract development and manufacturing organizations trend for regulatory affairs due to the rising need to navigate country-specific regulatory requirementsand skilled expertise.
Molecule Type Insights
The large molecule segment dominated the sterile injectables CDMO market and accounted for 61.4% of global revenue in 2025.The growth is driven by the rapid rise of biologics, including monoclonal antibodies, vaccines, and complex protein-based therapies that require aseptic fill-finish services for safe patient delivery. In addition, the growing demand for precision medicine has boosted the need for innovative treatments that target specific pathways or genetic markers associated with various conditions. Furthermore, constant advancements in biomanufacturing technologies are contributing to the segment’s growth.
In addition, the growing demand for precision medicine has boosted the need for innovative treatments that target specific pathways or genetic markers associated with various conditions. Furthermore, constant advancements in biomanufacturing technologies are contributing to the segment growth. Some of the innovations, such as single-use technologies, digital quality control, and continuous manufacturing processes, have made it more feasible for CDMOs to produce large molecules at scale, ensuring compliance with stringent regulatory requirements. These technological improvements reduce production costs and timeframes, making it more attractive for pharmaceutical companies to invest in the development of large molecules.
Small molecule is expected to grow at the second fastest CAGR over the forecast period, due to leading well-established infrastructure for their production, making them easier and less expensive to manufacture and have wider range of therapeutic applications as well as being less stringent in handling and storage compared to biologics, making them easier for CDMOs to manufacture.
Product Insights
The finished drug product segment accounted for the largest revenue share of 59.5% in 2025, driving the market growth due to increasing demand for ready-to-administer injectables, growth in biologics and high-potency therapies, and the need for compliant, scalable fill-finish capacity. Besides this, healthcare system focuses on supply-chain resilience and domestic manufacturing capacity also supports investment in sterile FDP infrastructure.
In addition, finished drug product segment is further sub-segmented to pre-filled syringes, vials and ampoules, specialty injectables, and others. Among these prefilled syringes segment is a rapidly expanding segment within the market, driven by increasing demand for ready-to-administer medicines, biologics, and emergency hospital therapies. Besides this, regulatory authorities such as the U.S. Food and Drug Administration emphasize aseptic processing, container-closure integrity, and human-factor safety in injectable delivery systems, supporting adoption of prefilled formats that reduce dosing errors and contamination risks.
The API segment is expected to grow significantly during the forecast period. The segment growth is driven by growing prevalence of chronic and infectious diseases requiring injectable biologics, vaccines, monoclonal antibodies, and high-potency drugs. In addition, this trend encourages pharmaceutical sponsors to outsource sterile API production to specialized CDMOs for scalable, compliant supply. Besides, technological advancements are improving API production quality and efficiency. Some of the innovations such as isolator technologies, closed-system transfers, single-use systems, continuous manufacturing, automation, and advanced aseptic filtration help reduce contamination risk, improve sterility assurance, and enhance overall manufacturing efficiency. These technologies also support the complex processing need of potent or sensitive API molecules, ensuring consistent, high-quality sterile injectable outputs.
Service Insights
The manufacturing segment accounted for the largest revenue share of 57.1% in 2025 and is expected to grow at the fastest CAGR over the forecast period. The segment’s dominance is primarily driven by the increasing outsourcing of large-scale commercial production of sterile injectable drugs by pharmaceutical and biotechnology companies. Sterile manufacturing requires highly specialized infrastructure, aseptic processing capabilities, and stringent regulatory compliance, which involve significant capital investments and operational complexity. In addition, growing geriatric population coupled with rising demand for biologics, high-potency therapies, and clinical-stage injectable products and increasing prevalence of chronic diseases are driving pharmaceutical companies to invest in R&D activities and launch new drugs in the market to meet the growing demand.
The robust demand for pharmaceutical products such as medications and small-molecule drugs can be attributed to flexible commercial manufacturing. Several pharmaceutical companies are turning to CDMOs to access advanced infrastructure and expertise due to the increasing influx of novel therapies and orphan drugs and higher returns on approved products. The increasing demand for new small molecule innovator APIs in the pharmaceutical industry is encouraging commercial CDMOs to expand their capabilities, which is expected to boost the market. For instance, in October 2025, Simtra BioPharma Solutions expanded its U.S. and European capacity with new isolator filling lines, ADC capabilities, and facilities, strengthening global sterile injectable manufacturing capacity and supporting the demand for complex biologic therapies.
The formulation development segment in the sterile injectables CDMO market is anticipated to witness the second fastest CAGR form 2026 to 2033, due to increasing demand for complex injectable therapeutics and specialized drug delivery systems. The rapid expansion of biologics, monoclonal antibodies, and advanced therapies such as gene and cell therapies has significantly increased the need for sophisticated formulation capabilities to ensure product stability, sterility, and bioavailability. In addition, pharmaceutical and biotechnology companies are increasingly outsourcing formulation development to CDMOs to reduce capital expenditure and accelerate time-to-market. Rising approvals of injectable drugs and the growing prevalence of chronic diseases requiring parenteral therapies are further supporting demand for advanced injectable formulations. These abovementioned factors are driving the formulation development services segment growth over the estimated timeframe.
Therapeutic Area Insights
The oncology segment accounted for the largest revenue share of 29.2% in 2025. The increasing number of cancer cases, rising R&D investments, and growing requirements for oncology drugs and biologics are some of the factors driving the segment growth. In addition, the robust pipeline of oncology drugs, such as monoclonal antibodies and immunotherapies, necessitates increased production capabilities. Thus, CDMOs play an important role in supporting pharmaceutical companies by providing the necessary infrastructure and expertise to manufacture these innovative treatments.
The central nervous system diseases segment is anticipated to grow at the fastest CAGR over the forecast period. This can be attributed to increasing prevalence of CNS disorders across the globe, attributed to factors such as aging populations, lifestyle changes, and environmental influences led to a growing need for effective treatments delivered via sterile injectables. Moreover, the rise in R&D activities focused on discovering novel therapies for CNS diseases spurred the demand for specialized manufacturing services provided by CDMOs.
Cardiovascular Diseases (CVDs) are among the major causes of death worldwide. According to the WHO report of July 2025, cardiovascular diseases caused about 19.8 million deaths worldwide, approximately 32.0% of all global fatalities with heart attacks and strokes accounting for 85.0% of these deaths, in 2022. Thus, advancements in small molecule innovator APIs are leading to the development of novel treatments for CVDs, which is expected to propel market growth.
Route of Administration Insights
The intravenous (IV) segment accounted for the largest revenue share of 32.4% in 2025, attributed to rapid onset and total bioavailability ensuring the drug dose reaches the bloodstream, bypassing first-pass metabolism. In addition, IV administration is also helpful when the drug dosages are low and highly viscous, which are difficult to absorb through tissues.
The subcutaneous route of administration segment is anticipated to grow at the fastest CAGR over the forecast period. This can be attributed to transition toward patient-friendly subcutaneous biologics which is accelerating demand for specialized fill-finish, prefilled syringes, and wearable injectors. In addition, reduced clinic burden, improved adherence, and pipeline shifts to large-volume SC therapies are driving the market growth.
The subcutaneous (SC) route of administration eliminates the need for long-term IV catheters or infusion pumps, driving the development of complex biologics designed for SC delivery. In addition, SC infusions are generally associated with fewer side effects and shorter patient chair times in clinics and hospitals, supporting the growth of this administration segment. For instance, according to an article published by Elsevier B.V. in September 2025, a systematic review comparing IV and SC administration for oncology therapies found that SC delivery significantly reduced both patient chair time and healthcare provider time. Consequently, indirect cost savings such as reduced work loss for patients and caregivers were observed, contributing to improved treatment satisfaction.
End-use Insights
The pharmaceutical companies segment accounted for the largest revenue share of 51.6% in 2025, owing to the increasing trend of drug developers and large pharmaceutical firms outsourcing critical stages of sterile injectables development and production to specialized CDMOs. Pharmaceutical companies are focusing on core competencies such as discovery and clinical development, while relying on CDMOs for expertise in aseptic processing, regulatory compliance, and high-volume manufacturing.
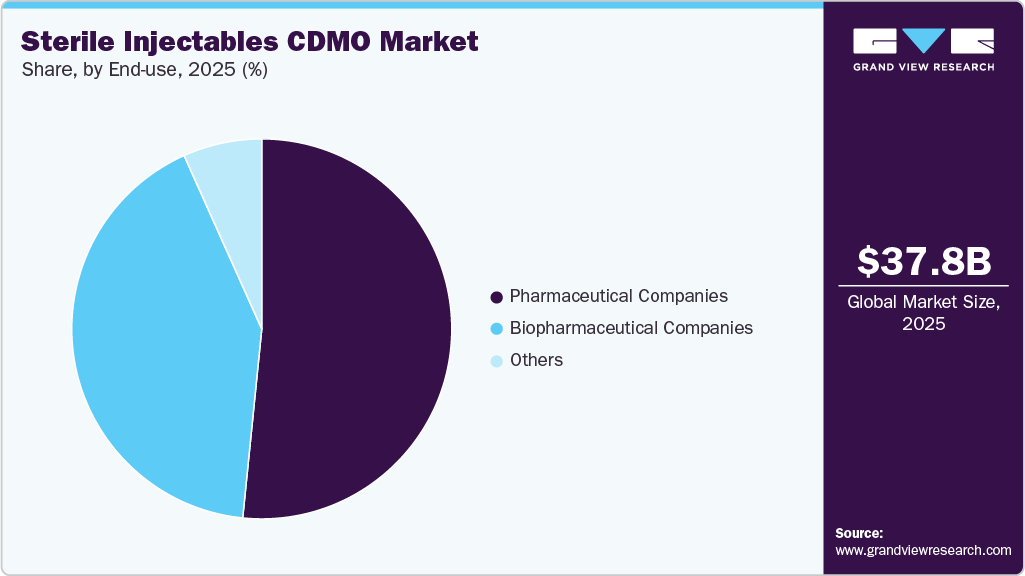
Biopharmaceutical companies are focusing on antibody drug conjugates (ADCs), mRNA therapeutics, and monoclonal antibodies (mAbs), along with orphan drugs. These formulations are sensitive and prone to contamination, highlighting the complexity of manufacturing these products, thus requiring specialized facilities and equipment for ensuring the integrity of the final drug. In addition, the target population for orphan drugs (rare diseases) is often small, needing smaller batches of drug volumes based on clinical trials conducted.
Regional Insights
North America held the largest market share in the sterile injectables CDMO market, accounting for 33.7% in 2025. The growth can be attributed to the establishment of contract manufacturing facilities, the increase in chronic diseases, and the advancement of biology. The expansion of the contract manufacturing market is significantly driven by the growing focus on biologics and biosimilars, which often necessitate sterile injectable formulations.
Furthermore, countries such as the U.S. and Canada benefit from the growing requirement for domestic production of complex biologics, biosimilars, and specialty generics, which strengthens supply security and reduces dependency on imports. In addition, the growing availability of advanced technology in the region is expected to drive market growth. For instance, in October 2025, Jubilant Pharmova Limited strengthened its contract manufacturing in North America by commissioning a third sterile injectable production line at its Washington facility. The site is managed by its wholly owned subsidiary, Jubilant HollisterStier (JHS). The USD 132 million investment is designed to support complex injectable projects, improve manufacturing efficiency and precision, and expand output, to double the plant’s overall sterile production capacity in Spokane.
U.S. Sterile Injectables CDMO Market Trends
U.S. is the largest market in North America and hold a substantial share by 2025. This growth is driven by a strong pipeline of new drug approvals and a commitment to innovative treatments, particularly in oncology and the management of chronic diseases. Further, the number of companies providing Contract Development and Manufacturing Organization (CDMO) services for sterile injectables is increasing. The rise in approvals for innovative therapies, especially in oncology and chronic conditions, is expected to fuel market growth during the forecast period.
Europe Sterile Injectables CDMO Market Trends
Europe’s sterile injectable CDMO market is experiencing steady expansion, driven by increasing demand for biologics, complex molecules, and personalized therapies. The UK benefits from a strong pharmaceutical base, supportive regulatory environment, and advanced manufacturing infrastructure, enabling efficient production and compliance with MHRA standards. Automated filling lines and advanced quality systems enhance throughput while ensuring consistent product quality. Germany’s market growth is supported by a mature pharmaceutical ecosystem, strategic European positioning, and a rising preference for outsourcing complex biologics and biosimilars. Investment in modular sterile filling and advanced aseptic capabilities strengthens production capacity, while BfArM and regional authorities enforce EU GMP and Annex 1 compliance with Qualified Person oversight.
The sterile injectables CDMO market in Germany is expected to witness significant growth, driven by the increasing demand for biopharmaceuticals and the rising prevalence of chronic diseases. The shift toward more complex drug formulations, particularly biologics and biosimilars, necessitated advanced manufacturing capabilities that several pharmaceutical companies prefer to outsource. This trend is further fueled by the need for cost-effective solutions and the ability to scale production quickly in response to market demands.
The UK sterile injectables CDMO market is anticipated to grow significantly over the coming decade. Growth can be attributed to the increasing focus on biologics and complex molecules, which often require sterile injectable delivery, which is expected to drive the market. In addition, the UK's supportive regulatory environment and the presence of FDA- and EMA-approved R&D and manufacturing facilities attract foreign investment, boosting the market.
Asia Pacific Sterile Injectables CDMO Market Trends
The Asia Pacific market is anticipated to grow at the fastest CAGR of 12.3% from 2026 to 2033. The growth is driven by rising demand for biologics, specialty injectables, and complex formulations. Japan has become a strategic hub due to its advanced technological infrastructure, robust R&D ecosystem, and regulatory efficiency, which supports faster clinical trials and product approvals. Strong government incentives streamlined PMDA review processes, and a focus on supply chain reliability position the country as a high-capability center for both domestic and global production. China’s market growth is fueled by increasing outsourcing of manufacturing by domestic and international pharmaceutical companies. Investments in advanced fill-finish systems, lyophilization, and prefilled syringes, coupled with strong regulatory oversight under the NMPA, support high-quality sterile manufacturing. Chinese CDMOs are leveraging technical expertise, regulatory compliance, and innovative platforms to serve both clinical and commercial-stage biologics, enhancing the region’s competitiveness.
China Sterile Injectables CDMO Marketis witnessing growth due to itsexpanding R&D activities, evolving regulatory frameworks, a diverse patient population, and rising demand for high-quality injectable therapies. Domestic and international pharmaceutical companies increasingly outsource manufacturing to Chinese CDMOs, leveraging local expertise, advanced infrastructure, and cost efficiency to accelerate development and commercial timelines. Government initiatives, foreign investments, and a strong focus on process innovation and quality assurance further support sector expansion.
Sterile injectables CDMO market in Japan is becoming a preferred destination for biopharmaceuticals and global contract research organizations (CROs). The growth is supported by advanced technological infrastructure, policy reforms, and a developed R&D ecosystem that enables faster patient recruitment and clinical trial execution. The market has benefited from expansions such as Fujifilm’s December 2025 completion of CDMO facility at Toyama Prefecture and AGC Biologics’ May 2025 upgradation of its Yokohama site, which enhance capacity for high-value biologics and specialty injectables.
Latin America Sterile Injectables CDMO Market Trends
The expansion of innovative biologics manufacturing capabilities across key Latin American countries is collectively strengthening the region’s sterile injectables CDMO market. Investments in GMP-compliant facilities, technology-enabled aseptic production, and integrated fill-finish operations are reducing import dependence while improving local access to high-complexity biologics and specialty injectable therapies.
Brazil sterile injectables CDMO market is growing due to the development of advanced domestic biologics manufacturing capabilities. As biologics and high-complexity APIs increasingly require aseptic processing, sterile fill-finish, and GMP-compliant clinical manufacturing, the presence of advanced local capabilities reduces import dependence and strengthens supply chain resilience.
MEA Sterile Injectables CDMO Market Trends
Middle Eastern countries such as South Africa, Saudi Arabia, the United Arab Emirates (UAE), and Kuwait are prospering economies. The MEA sterile injectables CDMO market is expected to witness significant growth, driven by evolving regulatory environments and increasing demand for high-quality pharmaceutical products. In the Middle East, countries such as Saudi Arabia and the UAE are investing heavily in healthcare infrastructure, creating a conducive environment for CDMOs to expand their operations.
South Africa sterile injectables CDMO market is strengthening due tothe increasing approval and adoption of long-acting injectable therapies which reflects growing confidence in advanced sterile biologic manufacturing and regulatory capability. As new complex injectables gain regulatory approval for public health programs, the demand for reliable manufacturing, fill-finish capacity, and supply chain coordination expands accordingly.
Key Sterile Injectables CDMO Company Insights
Some of the key players operating in this market are Boehringer Ingelheim GmbH, Baxter BioPharma Solutions (Simtra BioPharma Solutions), Vetter Pharma, Recipharm AB, Aenova Group, Fresenius Kabi, Unither Pharmaceuticals, FAMAR Health Care Services, Cipla, NextPharma Technologies, Ajinomoto Bio-Pharma Services, PCI Pharma Services, IDT Biologika, Alcami Corporation, Fareva Group, Eurofins CDMO, Siegfried Holding AG among others.
The market is evolving due to key initiatives for growing requirements for biologics and biosimilar, facility expansions, and adoption of advanced technology. Moreover, companies are pursuing mergers and acquisitions to enhance market presence while diversifying services for niche therapeutics and compliance needs. For instance, in January 2026,Vetter Pharma announced its expansion to build a new manufacturing facility in the Saarland region (Southwest Germany) to strengthen its European footprint, supported by regional advantages. The company has invested ~567.99 million USD (480 EUR) in its first phase of construction.
Key Sterile Injectables CDMO Companies:
The following key companies have been profiled for this study on the sterile injectables CDMO market.
- Boehringer Ingelheim GmbH
- Baxter BioPharma Solutions
- Vetter Pharma
- Recipharm AB
- Aenova Group
- Fresenius Kabi
- FAMAR Health Care Services
- NextPharma Technologies
- Ajinomoto Bio-Pharma Services
- PCI Pharma Services
- IDT Biologika
- Alcami Corporation
- Fareva Group
- Eurofins CDMO
- Siegfried Holding AG

Recent Developments:
-
In January 2026, Vetter Pharma announced its expansion to build a new manufacturing facility in the Saarland region (Southwest Germany) to strengthen its European footprint, supported by regional advantages. The company has invested ~567.99 million USD (480 EUR) in its first phase of construction.
-
In May 2025, FAMAR Health Care Services Group acquired a sterile production site from MiP Pharma in Homburg (Germany). This strategic milestone will result in strengthening the geographical footprint of the company in high-value dosage forms, thereby magnifying its capabilities in lyophilized and aseptic fill and finish.
-
In April 2024, Delpharm acquired the Meppel pharmaceutical manufacturing facility in the Netherlands from Astellas. This acquisition marks a significant strategic move for Delpharm as it expands its manufacturing capabilities and strengthens its presence in the pharmaceutical industry.
In February 2024, Novo Holdings, under the Novo Nordisk Foundation, acquired Catalent for USD 16.5 billion. This acquisition is part of Novo Holdings’ strategy to utilize its Wegovy cash and expand its manufacturing capabilities. The deal includes Novo Nordisk acquiring three of Catalent’s fill-finish sites from Novo Holdings for USD 11 billion upfront. These sites specialize in sterile drug filling and are expected to enhance Novo Nordisk’s manufacturing network for current and future treatments for diabetes and obesity. The acquisition is set to increase Novo Nordisk’s filling capacity starting in 2026 gradually
Sterile Injectables CDMO Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 41.56 billion
Revenue forecast in 2033
USD 87.34 billion
Growth rate
CAGR of 11.2% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Molecule type, product, service, therapeutic area, route of administration, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Switzerland; Ireland; Poland; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; Colombia; Chile; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar; Turkey
Key companies profiled
Boehringer Ingelheim GmbH; Baxter BioPharma Solutions; Vetter Pharma; Recipharm AB; Aenova Group; Fresenius Kabi; Famar; NextPharma Technologies; Ajinomoto Bio-Pharma Services, PCI Pharma Services; IDT Biologika; Alcami Corporation; Fareva Group; Eurofins CDMO; Siegfried Holding AG.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Sterile Injectables CDMO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global sterile injectables CDMO market report based on molecule type, product, service, therapeutic area, route of administration, end-use, and region.
-
Molecule Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecule
-
Large Molecule
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
API
-
Finished Drug Product
-
Pre-filled Syringes
-
Vials and Ampoules
-
Specialty Injectables
-
Others
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Formulation Development
-
Analytical & Testing Services
-
Method Development & Validation
-
Stability Testing
-
Drug Substance
-
Stability Indicating Method Validation
-
Accelerated Stability Testing
-
Photostability Testing
-
Other Stability Testing Methods
-
Extractable & Leachable Testing
-
Others
-
-
-
Manufacturing
-
Clinical Trial Manufacturing
-
Commercial Manufacturing
-
Aseptic Fill-Finish Services
-
-
Packaging
-
Storage
-
Cold
-
Non-cold
-
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Cardiovascular Diseases
-
Central Nervous System Diseases
-
Infectious Disorders
-
Musculoskeletal Diseases
-
Hormonal Diseases
-
Others
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Subcutaneous (SC)
-
Intravenous (IV)
-
Intramuscular (IM)
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biopharmaceutical Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
Switzerland
-
Ireland
-
Poland
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
Colombia
-
Chile
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
Turkey
-
-
Frequently Asked Questions About This Report
b. The global sterile injectables CDMO market size was estimated at USD 37.82 billion in 2025 and is expected to reach USD 41.56 billion in 2026.
b. The global sterile injectables CDMO outsourcing market is expected to grow at a compound annual growth rate of 11.19% from 2026 to 2033 to reach USD 87.34 billion by 2033.
b. North America dominated the sterile injectables CDMO market with a share of 33.75% in 2025. This is attributable to the establishment of contract manufacturing facilities, the rise in chronic diseases, and advances in biology. The expansion of the contract manufacturing market is driven primarily by the growing focus on biologics and biosimilars, which often require sterile injectable formulations.
b. Some key players operating in the sterile injectables CDMO market include Boehringer Ingelheim GmbH, Baxter BioPharma Solutions, Vetter Pharma, Recipharm AB, Aenova Group, Fresenius Kabi, FAMAR Health Care Services, NextPharma Technologies, Ajinomoto Bio-Pharma Services, PCI Pharma Services, IDT Biologika, Alcami Corporation, Fareva Group, Eurofins CDMO, Siegfried Holding AG
b. Key factors driving market growth include a growing focus on developing innovative therapeutics, a preference for personalized medicine, a changing regulatory landscape, increasing outsourcing trends, the adoption of one-stop-shop CDMOs, and growing R&D spending, among others.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









