- Home
- »
- Clinical Diagnostics
- »
-
Self-testing Market Size And Share, Industry Report, 2033GVR Report cover
![Self-testing Market Size, Share & Trends Report]()
Self-testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Kits, Device, Strips), By Sample (Blood, Urine, Stool), By Application, By Distribution Channel, By Usage, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-113-2
- Number of Report Pages: 160
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Self-testing Market Summary
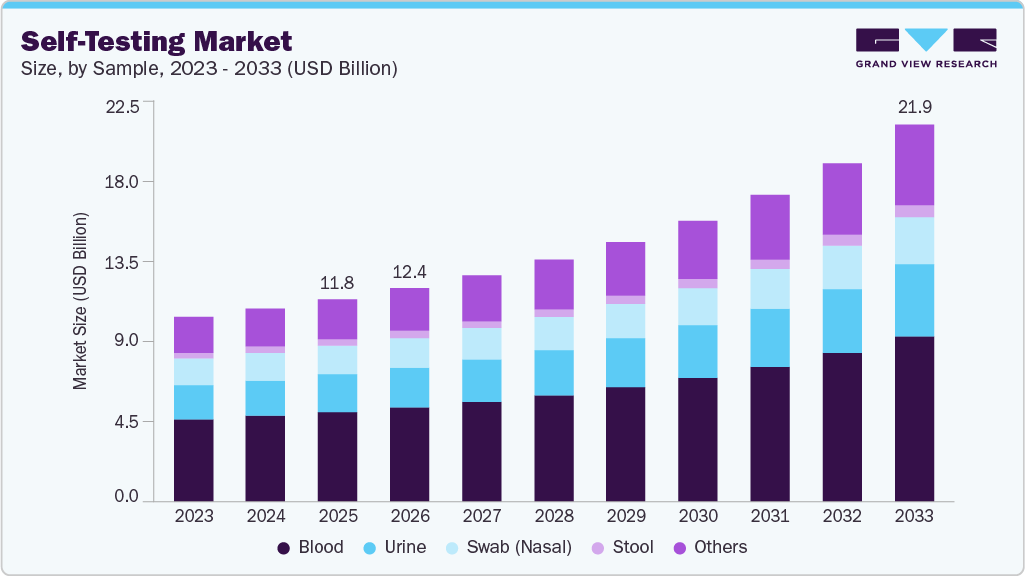
The global self-testing market size was estimated at USD 11.78 billion in 2025 and is expected to reach USD 21.92 billion by 2033, growing at a CAGR of 8.46% from 2026 to 2033. This growth is driven by increasing emphasis on personalized healthcare, technological advancements in rapid diagnostics, and rising demand for convenient and rapid diagnostics kits. Self-test/kits are used for rapid diagnosis of a wide variety of illnesses.
Key Market Trends & Insights
- By product, the kits segment held the highest market share of 40.52% in 2025.
- By sample type, the blood segment held the highest market share of 44.22% in 2025.
- Based on application, the diabetes and glucose tests segment held the highest market share of 28.29% in 2025.
- Based on distribution channel, the online segment held the highest market share of 55.85% in 2025.
- Based on usage, the reusable segment held the highest market share of 81.90% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 11.78 Billion
- 2033 Projected Market Size: USD 21.92 Billion
- CAGR (2026-2033): 8.46%
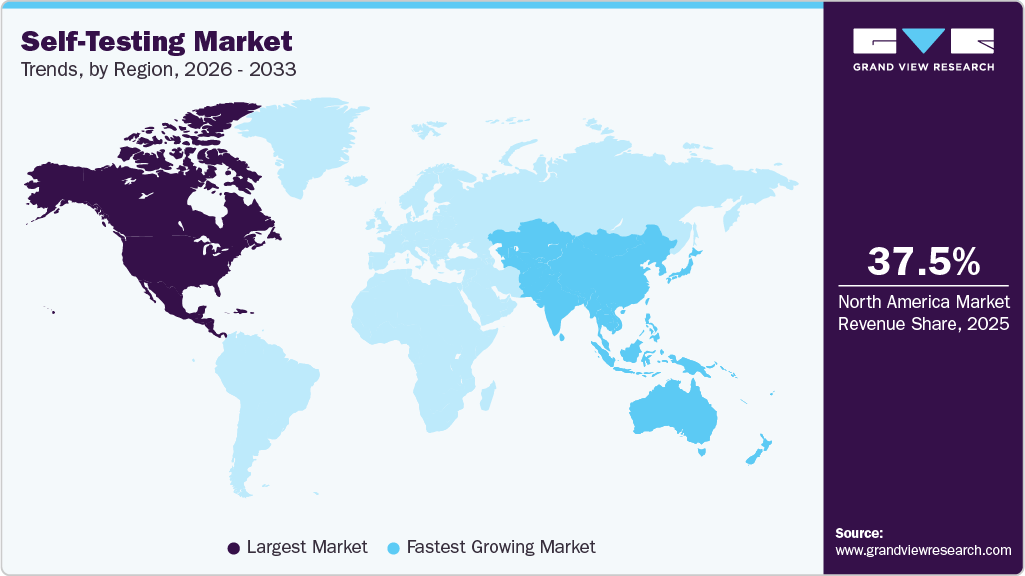
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Furthermore, rising awareness about the benefits of early diagnosis and regular monitoring has boosted the uptake of self-testing devices, and an increasing number of product launches are also expected to have a positive impact on the market. The COVID-19 pandemic has accelerated the need and emphasis on self-testing kits with an aim to increase the rate of diagnosis. To reduce the risk of serious disease, hospitalizations, or death, many organizations have implemented various efforts. For instance, the Administration for Strategic Preparedness and Response (ASPR) and NIH collaborated to launch the Home Test to Treat programme in January 2024. This programme is a virtual community health intervention that will provide free COVID health services, such as telehealth consultations, home treatments, and at-home rapid tests, in a few selected communities. Such factors further boost the market.Growing regulatory approvals are driving market expansion by instilling confidence in self-testing products. As more diagnostic tools receive approval from reputable regulatory bodies, including the WHO and FDA, they gain broader acceptance and credibility. This encourages adoption by both consumers and healthcare providers, facilitating access to accurate, reliable testing options. Regulatory approvals not only ensure product safety but also create pathways for market growth, promoting the development and availability of new self-testing solutions across various healthcare sectors. For instance, in July 2022, Innova secured a CE mark for its rapid antigen self-test, allowing the company to distribute the product across European markets. This approval provides a significant boost to the availability of at-home COVID-19 testing solutions, enabling easier access for consumers and enhancing the capacity to manage the pandemic. The CE mark indicates that the test meets the European Union's safety, health, and environmental protection standards, reinforcing confidence in its reliability and effectiveness.
In addition, increasing funding and investments by various organizations, as well as market players, for the development of robust and novel diagnostics has created beneficial opportunities for the market. For instance, in January 2022, the Florida Atlantic University (FAU) approved funding of USD 1.3 million to NIH for the development of quick and automated HIV self-kits. Furthermore, in April 2022, India-based start-up Cervicheck received approval from the Central Drugs Standard Control Organization (CDSCO) for its commercialization in India. The kit is used for the diagnosis of Human Papillomavirus (HPV).
Integration with telehealth services represents another major advancement driving the adoption of self-testing solutions. Remote consultation capabilities allow individuals to perform tests at home and immediately connect with healthcare professionals for interpretation, diagnosis, or treatment recommendations. This seamless connection between testing and clinical guidance improves healthcare accessibility, particularly for individuals in remote or underserved areas. Telehealth integration also supports chronic disease management by enabling continuous monitoring and timely medical interventions without requiring in-person visits. Healthcare systems are increasingly adopting digital ecosystems that combine diagnostics, data analytics, and virtual care, creating new opportunities for self-testing technologies. As telemedicine becomes more widely accepted, the demand for connected diagnostic tools that integrate with virtual healthcare platforms is expected to grow substantially. For instance, in April 2025, the Intro-Act Healthtech Newsletter, citing BioSpace news, reported that an AI-enabled home sleep apnea test wearable had received FDA 510(k) clearance, providing multi-night testing and severity identification validated against polysomnography, demonstrating the growing convergence of digital health, home diagnostics, and remote care technologies.
Market Concentration & Characteristics
The self-testing industry is marked by a high degree of innovation, propelled by continuous advancements in diagnostic technologies. In April 2025, Researchers in the United States developed an integrated nucleic acid testing device designed for HIV self-testing that interfaces with a USB-connected analyzer. The prototype allows users to collect a small finger-prick blood sample, process it through a microfluidic reagent cartridge, and obtain nucleic acid test results (via RT-LAMP amplification) in approximately 60 minutes. This innovation demonstrates proof of concept for highly sensitive, portable molecular testing outside traditional laboratory environments, potentially expanding the capabilities of HIV self-tests beyond rapid antibody detection to include direct viral nucleic acid analysis.
The self-testing industry has experienced a moderate level of merger and acquisitions (M&A) activity, driven by the need for companies to diversify their product portfolios and enhance their technological capabilities. These acquisitions often target smaller, innovative firms specializing in niche diagnostic solutions, enabling larger players to stay competitive.
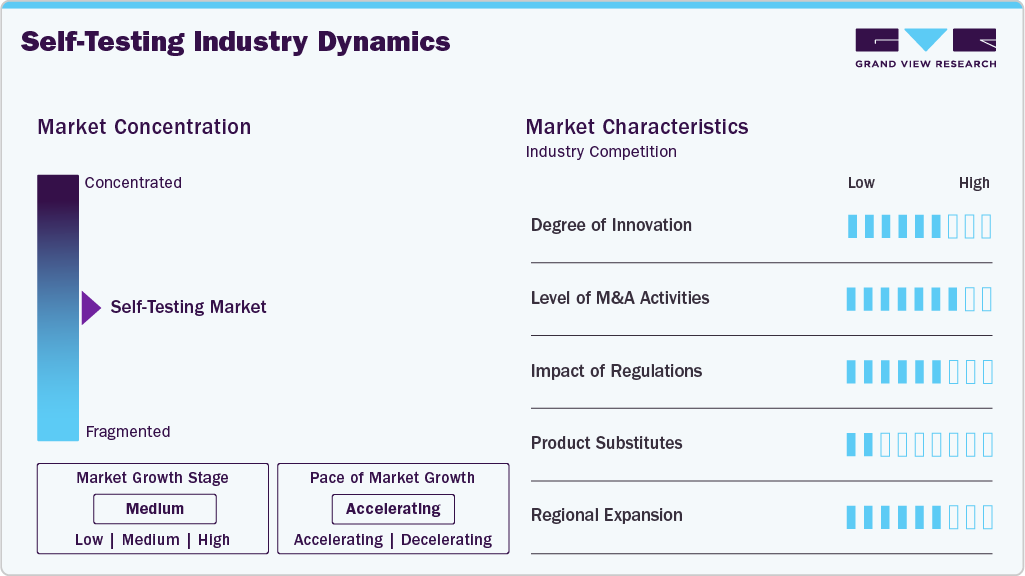
Regulations play a critical role in shaping the self-testing industry by establishing mandatory guidelines for product safety, accuracy, and efficacy. Regulatory frameworks like those from the U.S. FDA, European CE marking, and other regional authorities ensure that self-testing devices meet stringent quality standards. While these regulations build consumer trust, they also pose challenges for manufacturers, such as increased development costs and extended time-to-market. The swift approval of emergency-use diagnostic kits during the COVID-19 pandemic, however, showcased the industry's ability to adapt and innovate under regulatory guidelines.
Product substitution in the self-testing industry is robust, driven by the need to address emerging diagnostic trends and unmet needs. As new health concerns arise, manufacturers are expanding their offerings to include a diverse range of self-testing kits for conditions like infectious diseases, chronic illnesses, and genetic predispositions. This product expansion is further driven by advancements in materials science, enabling the development of more reliable, cost-effective, and user-friendly alternatives to traditional laboratory testing.
Regional expansion in the self-testing industry is characterized by targeted efforts to penetrate emerging markets while strengthening footholds in developed regions. Companies are leveraging strategic partnerships, collaborations, and localized manufacturing to address region-specific healthcare challenges and regulatory requirements. For instance, the growing demand for self-testing kits in Asia-Pacific, Latin America, and Africa highlights untapped opportunities, particularly in rural and underserved areas.
Sample Insights
Blood sample dominated the overall market with the largest revenue share of 44.22% in 2025. The dominance of the segment can be attributed to the increasing use of blood samples for diagnostics due to their non-invasiveness, ability to provide comprehensive insights into various health conditions, and advancements in diagnostic technologies. Digital connectivity with mobile applications also supports automated data tracking and clinical decision support. For instance, in January 2024, the U.S. Food and Drug Administration (FDA) reported that the Q-Pad A1c Test System was cleared based on a clinical study involving 198 participants, where 97.5% of users successfully collected valid blood samples, and results showed equivalence to laboratory venous blood testing.
Urine sample is expected to grow at the fastest growth rate over the forecast period, owing to the increasing use of urinalysis for various disease diagnostics. These tests are commonly used for detecting a range of conditions, including urinary tract infections (UTIs), kidney function, and pregnancy. The segment is particularly appealing for at-home diagnostics, driven by rising health awareness and the growing preference for convenient, accessible healthcare solutions. The availability of rapid reagent strip technologies also allows results to be generated within minutes, increasing consumer adoption. For instance, in October 2024, clinical data discussed at the American College of Physicians meeting and reported by MedCentral indicated that women experienced urinary tract infections up to 30 times more frequently than men, and as many as 40% of women who developed a UTI experienced another infection within 6 months, demonstrating sustained demand for accessible urine-based testing solutions.
Product Insights
The kits segment dominated the market with a revenue share of 40.52% in 2025 and is anticipated to grow at the fastest CAGR over the forecast period. The kits offer convenience, ease of use, and the ability to monitor various health conditions, such as pregnancy, diabetes, and infections, from the comfort of home. Innovations in test accuracy, user-friendly designs, and the expansion of distribution channels are fueling the segment's popularity. The segment growth is attributed to the growing focus of key players on investing in R&D and the development of new products. Increasing consumer preference for convenience and privacy, particularly for sensitive health conditions, is also driving demand for kit-based diagnostics. For instance, in May 2025, Reuters reported that the U.S. Food and Drug Administration had approved the first at-home cervical cancer screening kit following evaluation in a clinical study involving more than 600 women aged 25 to 65 years, demonstrating increasing regulatory acceptance of consumer-operated diagnostic kits.
The strips segment held considerable market share in 2025, owing to the high usage of strips and affordability. Testing strips are commonly used for monitoring blood glucose levels, urinalysis, ketone detection, and other diagnostic purposes, making them indispensable in chronic disease management. Their affordability, portability, and ease of use make them a preferred choice among consumers, particularly for daily health monitoring. Increasing geriatric population, growing cases of chronic diseases, and technological advancement in the medical sector are projected to boost the segment's revenue share.
Application Insights
The diabetes and glucose tests application segment held the largest revenue share of 28.29% in 2025, due to the rapidly increasing global prevalence of diabetes and prediabetes conditions. Diabetes requires continuous monitoring of blood glucose levels to prevent complications such as cardiovascular disease, kidney failure, and nerve damage, which directly drives demand for home-based testing solutions. For instance, in November 2024, Reuters reported that approximately 828 million adults globally were living with diabetes in 2022, nearly double the number recorded in 1990, while about 445 million adults aged 30 years and older were not receiving treatment, highlighting the urgent need for accessible self-testing solutions.
STD/STI tests are projected to showcase the fastest CAGR over the forecast period. Self-testing offers a discreet and convenient alternative to clinic-based screening, especially for individuals facing stigma, privacy concerns, or limited healthcare access. Home-based tests support timely detection and help reduce delays associated with traditional diagnostic pathways, improving disease management and treatment outcomes. For instance, in September 2025, the World Health Organization reported that more than 1 million sexually transmitted infections were acquired worldwide among individuals aged 15 to 49 years, emphasizing the urgent demand for accessible and scalable testing solutions such as self-testing kits.
Distribution Channel Insights
The online segment dominated the market with a market share of 55.85% in 2025, as online retailers offer greater accessibility of Self-testing products, allowing easier access to consumers regardless of their location. Online channels offer wider options of self-testing products than offline channels, which also provide access to niche products. Furthermore, the convenience of e-commerce platforms and direct-to-consumer (DTC) websites enables individuals to purchase self-test kits directly and easily from the comfort of their homes.
Offline channels are projected to hold the fastest CAGR over the forecast period. Significant investments by companies in building their own distribution networks are projected to accelerate segment growth. In addition, an increasing number of distribution agreements within companies is also estimated to have a positive impact on the segment growth by 2030. This channel continues to play a vital role in regions where access to online platforms is limited or where consumers prefer in-person consultations. The offline segment is particularly important for markets with regulatory requirements that necessitate direct interactions with healthcare professionals.
Usage Insights
The reusable segment held the largest revenue share of 81.90% in 2025. The segment is further projected to grow with the fastest CAGR value over the forecast period. They are commonly used for health monitoring purposes, such as blood glucose testing or ovulation prediction. Reusable kits are appealing to consumers seeking long-term value and a more sustainable approach to healthcare, while also providing the convenience of home testing. The segment's growth is supported by innovations in durability and ease of cleaning.
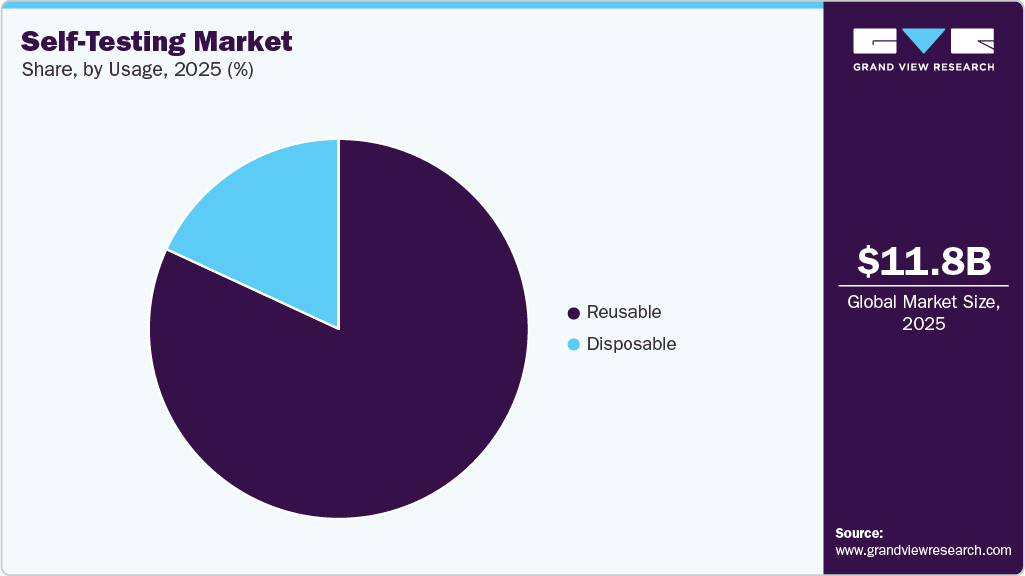
Reusable kits are expected to hold considerable market share during the forecast period. Self-testing provides new opportunities to detect, diagnose, and address patients and their treatment process. Disposable kits offer remarkable usability due to their convenient and user-friendly nature. They eliminate the need for complex preparation or cleaning, ensuring a hygienic and hassle-free experience. Such advantages of disposable kits and improved safety are likely to accelerate segment growth during the forecast period.
Regional Insights
The self-testing market in North America dominated the overall global market and accounted for a 37.53% share in 2025, attributed to growing cases of chronic conditions that require quick diagnosis, technological advancement in the healthcare sector, and availability of key players, which are projected to boost the revenue growth in the region. An increasing number of FDA approvals for new products also boosts market growth. For instance, in May 2022, empowerDX announced the launch of an at-home celiac disease genetic risk test. The kits perform molecular testing and provide accurate results.
U.S. Self-Testing Market Trends
The self-testing market in the U.S. held a significant share of North America in 2024, driven by the combination of factors, including high health awareness, advanced healthcare infrastructure, and the increasing prevalence of chronic conditions such as diabetes and hypertension. The market has also benefited from the widespread adoption of telemedicine and digital health solutions, which have driven the demand for at-home diagnostic kits. In addition, in August 2025, this momentum continued with FDA approval of bioLytical’s INSTI HIV Self-Test, offering results in approximately one minute and setting new expectations for rapid consumer diagnostics. In July 2025, it also highlighted the strategic response of mature players, as BD announced its FDA submission for an at-home HPV self-collection screening solution aimed at expanding cervical cancer prevention.
Europe Self-Testing Market Trends
The self-testing market in Europe is experiencing significant growth. The growth is supported by rising consumer demand for accessible healthcare solutions and government initiatives promoting preventive care. The increasing burden of chronic diseases, such as cardiovascular disorders and cancer, has fueled the adoption of self-testing kits for early detection and monitoring. Technological advancements and the growing acceptance of at-home diagnostics across European countries have also played a pivotal role in driving market expansion.
The UK self-testing market is experiencing significant growth primarily due to heightened health awareness and the National Health Service (NHS) initiatives encouraging early diagnosis and self-monitoring. In June 2023, UK-based Linear Diagnostics secured funding from the National Institute for Health and Care Research (NIHR) to develop a 20-minute rapid test for gonorrhea and chlamydia, addressing the need for faster, more accessible STI diagnostics. Such advancements highlight Europe’s focus on enhancing self-testing accuracy and convenience, reducing reliance on clinical visits, and expanding access to early detection solutions.
The self-testing market in Germany is experiencing significant growth, driven by high adoption rates of innovative self-diagnostic tools, supported by robust healthcare policies, which have further strengthened the market. Germany's aging population and increasing prevalence of chronic illnesses have also amplified the demand for reliable and user-friendly self-testing solutions, contributing to the market's upward momentum.
Asia Pacific Self-Testing Market Trends
The self-testing market in the Asia Pacific is experiencing the fastest growth. A rising number of activities related to research & development of novel therapeutics for infections, improving the healthcare sector, and government initiatives to reduce the disease burden are key factors driving regional market growth. Other factors, such as increasing healthcare reforms, expanding healthcare infrastructure, growing population, rising incidence of chronic diseases, and increasing number of local companies entering the market, are also estimated to drive the market during the forecast period. The critical need for self-testing kits in the Asia-Pacific region, particularly in countries like Thailand, where high disease prevalence demands accessible and efficient diagnostic tools. Self-test kits can play a pivotal role in early detection, empowering individuals to seek timely treatment and support public health goals. In Thailand, recent data from the Ministry of Health indicates that over 2 million people are currently living with chronic hepatitis B, underscoring its status as a significant public health challenge. The World Health Organization (WHO) has launched a global campaign aiming to eradicate hepatitis B by 2030, reflecting the urgency to address this pressing issue.
China self-testing market is growing, driven by a large population with increasing health awareness and a growing burden of chronic diseases such as diabetes and cardiovascular disorders. Government initiatives to expand healthcare access and promote early diagnosis have further driven market demand. Furthermore, the availability of affordable self-testing kits and the rapid expansion of e-commerce platforms have also played a critical role in market development.
The self-testing market in Japan is experiencing growth driven by the country’s focus on preventive healthcare, and high adoption rates of advanced technologies have contributed to the market's expansion. Furthermore, Japan's strong healthcare infrastructure and consumer preference for user-friendly, accurate diagnostic tools are driving innovation in the market.
India self-testing market is experiencing growth, driven by India faces critical health challenges, including the rising prevalence of diseases like dengue fever, with nearly 300,000 cases reported in 2023. Early diagnosis is essential to avoid severe complications, but many cases remain undetected until symptoms worsen. The growing need for self-testing solutions in India is evident, as they provide a timely and accessible way to detect health issues, helping individuals manage their conditions before they escalate into more serious concerns. Furthermore, UTIs affect around 30% of Indian women, and if untreated, they can lead to serious health problems, including kidney damage. In addition, the rise in early menopause, occurring as early as age 40, can lead to anxiety and depression if undiagnosed. To address such issues, manufacturers such as Mankind Pharmaoffer RAPID NEWS self-test kits, which are accessible, easy-to-use solutions for detecting dengue, UTIs, and early menopause, empowering individuals to test privately and reliably from home.
Latin America Self-Testing Market Trends
The self-testing market in Latin America is experiencing significant growth, due to the region's improving healthcare infrastructure and growing middle-class population with access to disposable income have also spurred demand for self-testing solutions. In addition, collaborations between local manufacturers and global players are enhancing market penetration.
Brazil self-testing market is shaped by an evolving regulatory landscape that emphasizes product safety and accuracy. The growing prevalence of conditions like diabetes and hypertension has increased demand for self-monitoring solutions. Moreover, government-led health campaigns and partnerships between public and private sectors are driving market growth, particularly in urban areas where access to healthcare facilities is limited.
Middle East And Africa Self-Testing Market Trends
The self-testing market in the MEA is expanding as awareness of chronic and infectious diseases is rising across the region. Initiatives to improve healthcare access, especially in underserved areas, have enhanced the adoption of self-testing kits. The growing affordability of these solutions and the region's increasing reliance on digital health technologies are additional contributors to market growth.
Saudi Arabia self-testing market is characterized by rapid innovation, growing competition, and a dynamic regulatory environment. The government’s Vision 2030 initiative, which aims to modernize the healthcare sector and promote preventive care, has accelerated the adoption of self-diagnostic tools. The market is also benefiting from a tech-savvy population, increasing investments in healthcare, and a strong focus on localized manufacturing to meet growing demand.
Key Self-testing Company Insights
The market is projected to show lucrative growth as major market players are focusing on various strategic initiatives such as the launch of new products, mergers & acquisitions, and expansion in other regions. For instance, in March 2025, the U.S. Food and Drug Administration (FDA) granted marketing authorization to Visby Medical for the Visby Medical Women’s Sexual Health Test, marking the first diagnostic test for chlamydia, gonorrhea, and trichomoniasis that can be purchased and used entirely at home without a prescription. The single-use kit includes a self-collected vaginal swab and a powered testing device that transmits results to the Visby Medical app, with results available in approximately 30 minutes.
Key Self-testing Companies:
The following key companies have been profiled for this study on the self-testing market.
- Geratherm Medical AG
- Cardinal Health
- OraSure Technologies, Inc.
- bioLytical Laboratories Inc.
- PRIMA Lab SA.
- BD
- F. Hoffmann-La Roche Ltd.
- Bionime Corporation
- Abbott
- Everlywell, Inc.
Recent Developments
-
In February 2026, UK-based NHS-licensed digital healthcare platform Evaro entered into a strategic partnership with U-Test Diagnostics, a provider of rapid home self-testing kits. The partnership aims to combine self-testing capabilities with Evaro’s clinician-review and prescription fulfillment services, improving the overall patient experience by enabling users to seamlessly move from self-test delivery through to medical guidance and treatment.
-
In July 2025, BD (Becton, Dickinson and Company) announced it had submitted a new at-home HPV test application to the U.S. Food and Drug Administration. The test enables people to self-collect a vaginal sample at home using a simple swab about the size of a Q-tip, improving comfort and privacy. Once mailed to the lab, the sample enters the BD COR automated system, which prepares, analyzes, and reports results with minimal manual handling, improving efficiency and quality.
-
In September 2024, Mankind Pharma announced the launch of rapid self-test kits aimed at addressing critical health challenges in India. These kits are designed to provide quick and accurate results for various health conditions, thereby enhancing accessibility to healthcare services. The initiative is part of Mankind Pharma's commitment to improving public health and empowering individuals to take charge of their health management.
-
In July 2024, the World Health Organization (WHO) prequalified the OraQuick HCV self-test manufactured by OraSure Technologies, which is used to enhance access to hepatitis C testing and diagnosis. This self-test builds on the OraQuick HCV Rapid Antibody Test.
Self-testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 12.41 billion
Revenue forecast in 2033
USD 21.92 billion
Growth rate
CAGR of 8.46% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 - 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, sample, application, distribution channel, usage
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Geratherm Medical AG; Cardinal Health; OraSure Technologies, Inc.; bioLytical Laboratories Inc.; PRIMA Lab SA; BD; F. Hoffmann-La Roche Ltd.; Bionime Corporation; Abbott; Everlywell, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Self-testing Market Report Segmentation
This report forecasts revenue growth at global, regional, & country levels and provides an analysis of industry trends in each of the subsegments from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the global self-testing market report based on product, sample, application, distribution channel, usage, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Kits
-
Devices
-
Strips
-
-
Sample Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood
-
Urine
-
Stool
-
Swab
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood Pressure Test
-
Diabetes and Glucose Tests
-
Cholesterol and Triglycerides Tests
-
Pregnancy Test
-
STD /STI Test
-
Urinary Tract Infection Test
-
Anemia Test
-
Allergy Test
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Online
-
Offline
-
-
Usage Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Disposable
-
Reusable
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global self-testing market size was estimated at USD 11.78 billion in 2025 and is expected to reach USD 12.42 billion in 2026.
b. Some key players operating in the self-testing market include Geratherm Medical AG, Cardinal Health, OraSure Technologies, Inc., bioLytical Laboratories Inc., PRIMA Lab SA, BD, F. Hoffmann-La Roche Ltd., Bionime Corporation, Abbott, Everlywell, Inc.
b. Key factors that are driving the market growth include increasing emphasis on personalized healthcare, technological advancements in rapid diagnostics, and rising demand for convenient and rapid diagnostics kits.
b. The global self-testing market is expected to grow at a compound annual growth rate of 8.46% from 2026 to 2033 to reach USD 21.92 billion by 2033.
b. North America dominated the self-testing market with a share of 37.89% in 2025. This is attributable to growing cases of chronic conditions that requires quick diagnosis, technological advancement in the healthcare sector and availability of key players in the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









