- Home
- »
- Healthcare IT
- »
-
Regulatory Information Management System Market Report, 2033GVR Report cover
![Regulatory Information Management System Market Size, Share & Trends Report]()
Regulatory Information Management System Market (2026 - 2033) Size, Share & Trends Analysis Report By End Use (Pharmaceutical Sector, Medical Device Sector), By Region (North America, Europe, Asia Pacific, Middle East & Africa, Latin America), And Segment Forecasts
- Report ID: GVR-4-68039-971-6
- Number of Report Pages: 190
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
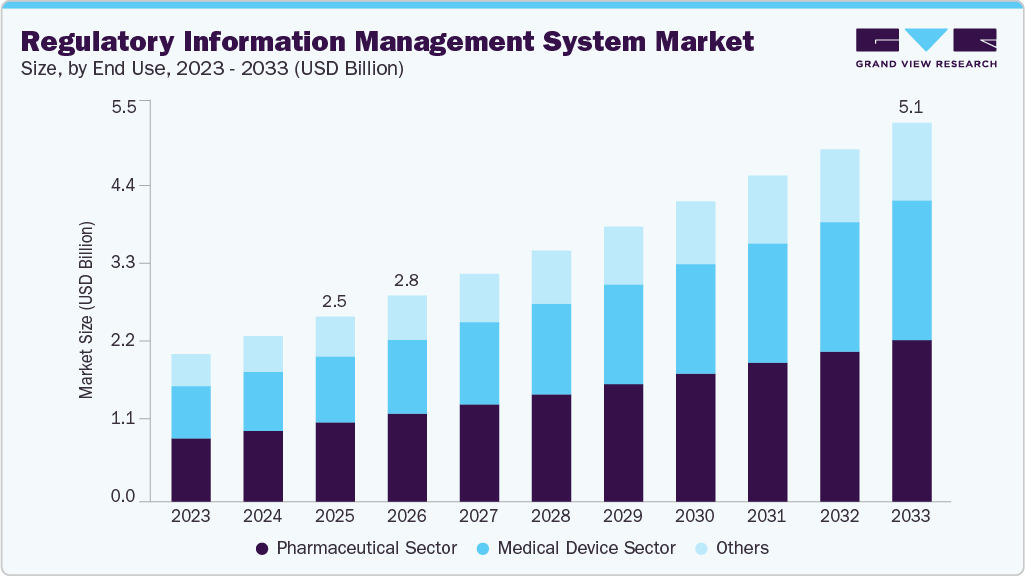
Regulatory Information Management System Market Summary
The global regulatory information management system market size was estimated at USD 2.50 billion in 2025 and is projected to reach USD 5.11 billion by 2033, growing at a CAGR of 9.10% from 2026 to 2033. The market's growth is driven by the growing volume of global regulatory submissions, the digital transformation of regulatory and compliance processes, rising adoption of cloud-based software solutions.
Key Market Trends & Insights
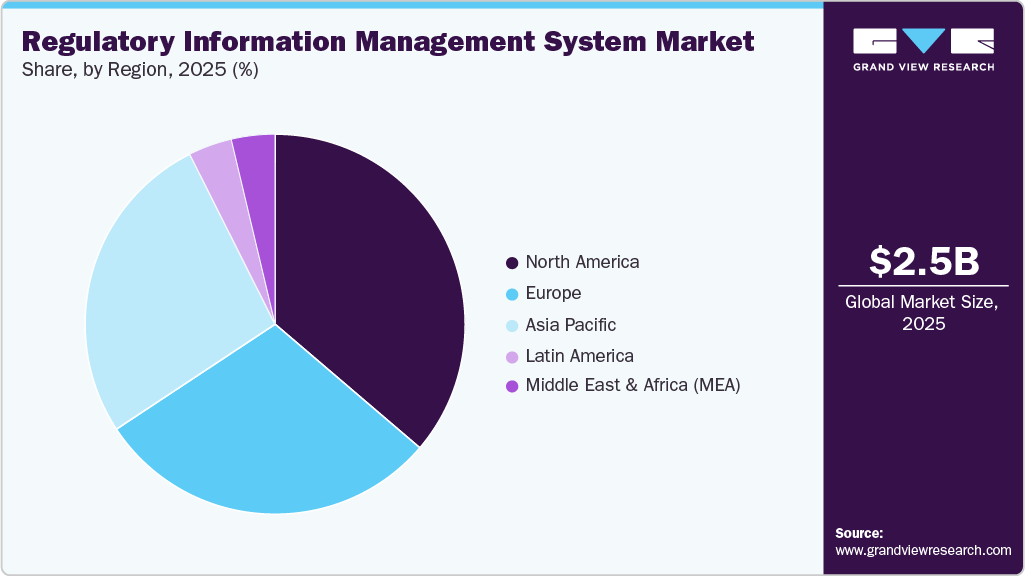
- North America dominated the market for regulatory information management market with a share of 36.83% in 2025.
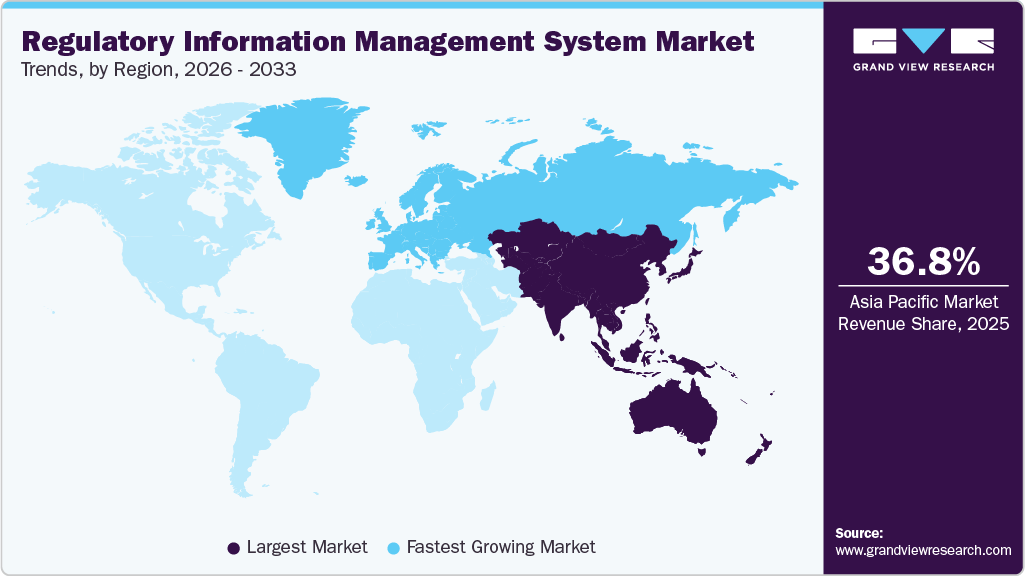
- Europe is estimated to be the fastest-growing region over the forecast period.
- Based on end use, the Pharmaceutical Sector segment held the dominant market share of 42.61% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.50 Billion
- 2033 Projected Market Size: USD 5.11 Billion
- CAGR (2026-2033): 9.10%
- North America: Largest market in 2025
- Europe: Fastest growing market
The growing number of regulatory submissions worldwide has significantly accelerated the adoption of regulatory information management system software, as pharmaceutical, biotechnology, and medical device companies increasingly pursue approvals across multiple geographies simultaneously. Rising R&D activity, faster development timelines, post-pandemic pipeline recovery, and expansion into emerging markets led to a sharp increase in regulatory filings, variations, renewals, labeling updates, and post-approval change submissions. At the same time, regulators such as the FDA, EMA, and national authorities require more frequent lifecycle updates, electronic submissions, and structured data formats. Managing this expanding submission volume through manual or fragmented processes is inefficient and error-prone, driving the need for centralized RIMS platforms to ensure consistency, visibility, and compliant global regulatory operations.
Implementing regulatory compliance software within an organization ensures data security and privacy, maintaining the reliability & credibility of information used in the regulatory process. Companies such as Freyr, with their advanced Integrated Regulatory Management System (IRMS), emphasize their commitment to upholding the highest data security and regulatory compliance standards. This dedication increases trust and reliability in client relationships. This demonstrates the growing appeal of RIM software, which is in accordance with the primary input, which specifically states that conventional RIM is no longer in use:
“In order to comply with the requirements for drug approval, traditional methods of RIM are not possible to follow because they take a lot of time, and time is crucial in the pharmaceutical industry. If we compromise on timing, we risk losing the drug's royalties or experiencing other similar consequences, so adopting such technological advancements as Veeva Vaults RIM software gives us the leverage of working effectively.”
- Pharmaceutical companies market expert, India.
For instance, a well-known pharmaceutical business noted in its customers' feedback for Veeva's Vault RIM Suite that RIM is one of the change and digitalization strategies that they are continually forcing themselves to think about when exploring the data-driven regulatory information solution.

Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, the impact of regulations, the level of partnership & collaboration activities, the degree of innovation, and regional expansion. For instance, the regulatory information management systems market is slightly fragmented, with many providers entering the market. The degree of innovation, the level of partnerships & collaboration, and the impact of regulations on the industry are high. However, the regional expansion observes moderate growth.
The degree of innovation in the market seems moderate in 2025 Innovation is driven by the need for centralized, efficient, and compliant regulatory operations. Advances in digital integration, AI-enabled analytics, and automated workflow management are enabling healthcare and life sciences companies to streamline global submissions, maintain accurate and up-to-date regulatory data, support faster approvals, and ensure consistent compliance across multiple markets.

Partnerships and collaborations in the RIMS market are increasing to strengthen digital infrastructure, streamline compliance, and centralize regulatory data. Companies are collaborating with technology vendors and consulting firms to enhance submission workflows and ensure global compliance. For instance, in March 2025, Rimsys partnered with KPMG to advance digital transformation in the MedTech industry by combining Rimsys’ regulatory software expertise with KPMG’s advisory and business transformation capabilities. The partnership aims to help MedTech companies modernize their regulatory management processes, scale RIMS readiness, strengthen compliance, and accelerate innovation and product market access.
Regulations play a crucial role in the regulatory information management system market by ensuring accurate, compliant, and auditable management of regulatory data. Regional differences in rules, such as those set by the FDA in the U.S., EMA in Europe, and CDSCO in India, create operational complexity that requires tailored compliance strategies. At the same time, these regulations drive investments in centralized data management, digital workflows, staff training, and quality assurance, ensuring RIMS platforms support safe, reliable, and fully compliant regulatory operations across global markets.
RIMS market players are expanding globally to meet diverse regulatory needs, offering localized compliance support and tailored solutions across North America, Europe, Asia-Pacific, and the Middle East. For instance, in October 2023, ArisGlobal opened a new office in Mysore, India, strengthening regional capabilities and enhancing product development and customer support. Such expansions help improve regional compliance, engagement with local regulators, and efficiency in managing global regulatory operation
End Use Insights
Based on end-use, the pharmaceutical segment led the market with the largest revenue share of 42.61%in 2025. The pharmaceutical industry faces numerous regulations and compliance requirements, necessitating robust Regulatory Information Management (RIM) systems. In addition, increasing number of clinical trials and regulatory approval submissions in the pharmaceutical sector is a potential growth driver. For instance, in August 2023, Calyx's RIM system played a crucial role in a global pharmaceutical company's successful submission to the FDA during an eCTD 4.0 implementation pilot program.
The medical device segment is expected to grow at significant CAGR over the forecast period. RIM systems offer a centralized platform for efficient data management and regulatory documentation, leading many medical device companies to shift to these applications. For instance, in October 2022, Veeva Systems announced that over 350 companies have adopted Veeva Vault RIM Suite apps, showcasing its presence in the market.
Furthermore, growing emphasis on product safety and quality control is propelling the adoption of RIM systems. In sectors such as biologics, stringent regulatory oversight is crucial for patient safety while manufacturing complex biological products. Similarly, in the cosmetics and food sectors, adherence to regulatory guidelines is vital for consumer safety and satisfaction. RIM systems offer an effective solution for managing regulatory submissions, product registrations, and compliance documentation, enhancing quality control and risk management. For instance, in October 2022, China reinforced safety and technical standards for cosmetics, significantly impacting the demand for RIM systems in the cosmetics industry.
Regional Insights
North America dominated the market for regulatory information management market with a share of 36.83% in 2025. In 2025, North America dominates the global regulatory information management system (RIMS) market, driven by the presence of multiple RIMS providers offering comprehensive regulatory solutions. In addition, government initiatives in North America aim to implement RIMS by fostering collaboration with various stakeholders. According to MTaPS's December 2022 update, the Promoting Quality of Medicines Plus (PQM+) and USAID MTaPS planned to collaborate with stakeholders to establish interoperable and integrated RIMS 2 years ago.
U.S. Regulatory Information Management System Market Trends
The regulatory information management system market in the U.S. driven by the increasing complexity of adhering to U.S. FDA regulations and managing product information across multiple states and global submissions. Challenges such as dynamic regulatory updates, manual processes, complex enterprise systems, and resource constraints make compliance management difficult for U.S. life sciences companies. Leading firms, are adopting RIMS to centralize regulatory data, track submission status, manage FDA correspondence, and monitor compliance obligations.
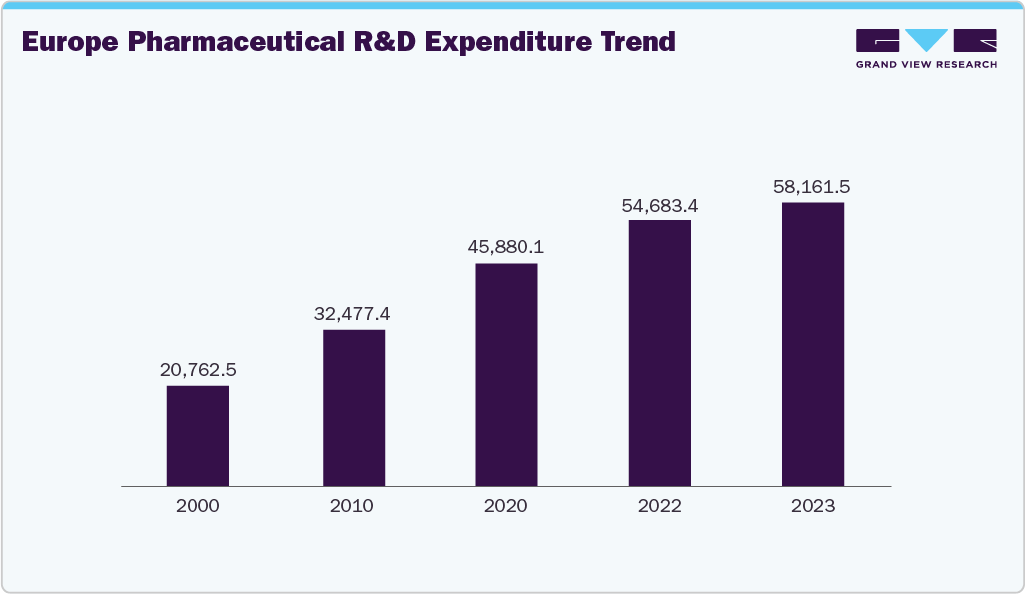
Europe Regulatory Information Management System Market Trends
The regulatory information management system market in Europe is expected to grow at the fastest CAGR over the forecast period. This can be attributed to the changing regulatory scenarios in the life science industry and increasing adoption of advanced technologies and automation in regulatory frameworks is expected to drive demand for RIMS in the region. Companies such as Rimsys, Calyx, and Kalypso offer solutions to streamline regulatory processes. For instance, Rimsys, a global provider of RIM systems, implemented its regulatory management software to automate the European Union in Vitro Diagnostics Regulation (EU IVDR) General Safety and Performance Requirements (GSPRs) for a global player in the IVD market.
The UK regulatory information management system market is being driven by the rising regulatory complexity and increasing digitalization of regulatory operations in the UK are driving adoption of regulatory information management systems. Leading pharmaceutical and medical device companies are implementing RIM solutions to improve efficiency and decision-making. For example, in April 2022, Mundipharma International deployed Veeva Systems’ RIM platform across 120 local affiliates and two major product portfolios. Additionally, emerging UK regulatory initiatives, such as the Innovative Devices Access Pathway (IDAP) Pilot, are expected to further support demand for RIM systems during the forecast period.
Growth of the Germany regulatory information management systems market is driven by factors such as growing emphasis on regulatory compliance, increasing cyber threats, and the need for robust data security. Pharmaceutical & medical device companies recognize the importance of adopting regulatory solutions to manage compliance complexities. For instance, in January 2023, CHEPLAPHARM Arzneimittel GmbH, a German pharma company, selected Generis Enterprise Technology Limited to offer its CARA Life Science Platform for merging & standardizing regulatory, RIM, labeling, and quality processes across domains. These factors are expected to drive demand for RIM systems in the country over the forecast period.
Asia Pacific Regulatory Information Management System Market Trends
Asia Pacific region is anticipated to experience the rapid growth during the forecast period. Factors contributing to this growth include the increasing number of pharmaceutical-based products & medical devices and a growing concern for regulatory compliance. There is a notable increase in the demand for RIMS to handle regulatory concerns and data maintenance requirements
The Japan regulatory information management system market is witnessing growth due to the increasing complexity of Japan’s regulatory clearance processes. This in turn is driving adoption of regulatory information management software in the country to manage rising compliance costs and requirements efficiently. While pharmaceuticals remain a key segment, other regulated industries, including chemical compliance solutions offered by UL LLC, are also contributing to the growth of Japan’s regulatory information management market.
The growth of regulatory information management system market in India is contributed to increasing regulatory scrutiny and frequent policy updates across the pharmaceutical and medical device sectors. Expanding oversight by CDSCO, wider coverage of medical device regulations, and rising electronic submission and lifecycle reporting requirements are driving companies to adopt RIM systems to centralize regulatory data and streamline compliance management.
Latin America Regulatory Information Management System Market Trends
The growth of regulatory information management system market in Latin America is contributed to increasing regulatory complexity and evolving country-specific requirements across the pharmaceutical and medical device sectors. Regulatory bodies such as ANVISA ((Agência Nacional de Vigilância Sanitária) are strengthening oversight, expanding documentation requirements, and mandating electronic submissions, driving companies to adopt RIM systems to centralize regulatory data, manage multi-country filings, and ensure consistent compliance across the region.
Middle East & Africa Regulatory Information Management System Market Trends
The growth of the regulatory information management system market in the Middle East & Africa is supported by regulatory modernization, expanding healthcare activity, and increasing adoption of digital compliance tools. Strengthening regulatory frameworks and electronic submission requirements across key markets are driving companies to adopt RIMS solutions to manage regulatory data and ensure consistent compliance across the region.
Key Regulatory Information Management System Company Insights
Key players in the regulatory information management system market are adopting innovative strategies, offering personalized infusion formulations, mobile and in-clinic services, and digital booking platforms to enhance accessibility and patient experience. Integration of smart infusion technologies and partnerships with wellness centers, fitness facilities, and luxury providers are further driving adoption and expanding market reach.
Key Regulatory Information Management System Companies:
The following key companies have been profiled for this study on the regulatory information management system market.
- Veeva Systems
- Kalypso (Rockwell Automation)
- DDi
- Körber AG
- ArisGlobal
- PhlexGlobal
- AmpleLogic
- Ennov
- MasterControl Solutions, Inc.
- Rimsys
- Ithos Global Inc. (Cordance Group)
- LORENZ Life Sciences Group
- IQVIA
- EXTEDO
Recent Developments
-
In April 2023, Ennov acquired Samarind, a provider of RIM solutions. The aim behind the acquisition was to strengthen its global position in the RIM software market.
-
In February 2023, ArisGlobal acquired Amplexor Life Sciences, combining Amplexor’s regulatory, quality, and safety software with ArisGlobal’s LifeSphere platform to create a more comprehensive life sciences regulatory SaaS offering.
-
In February 2023, ArisGlobal launched a new RIM solution named Investigational Product RIMS. The launch aims to cater to the increasing demands of the life sciences and medical device companies.
-
In January 2022, Cordance Group completed the acquisition of Ithos Global, a provider of regulatory compliance and formulation management software for the cosmetics industry. The acquisition allows Ithos Global to leverage Cordance’s operational expertise and financial resources to scale its business, expand technology offerings, and enhance support for customers
Regulatory Information Management System Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.78 billion
Revenue forecast in 2033
USD 5.11 billion
Growth rate
CAGR of 9.10% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market Value in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
End use and region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden; Switzerland; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Veeva Systems; Kalypso (Rockwell Automation); DDi; Körber AG; ArisGlobal; PhlexGlobal; AmpleLogic; Ennov; MasterControl Solutions, Inc.; Rimsys; Ithos Global Inc. (Cordance Group); LORENZ Life Sciences Group; IQVIA; EXTEDO
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Regulatory Information Management System Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global regulatory information management system market report based on solution type, deployment mode, end use and regions.
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Sector
-
Medical Device Sector
-
Others (Biologics, Cosmetics, Foods, etc.)
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
Switzerland
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global regulatory information management system market size was estimated at USD 2.50 billion in 2025 and is expected to reach USD 2.78 billion in 2026.
b. The global regulatory information management system market is expected to grow at a compound annual growth rate of 9.10% from 2026 to 2033 to reach USD 5.11 billion by 2033.
b. Some of the prominent players in the regulatory information management system market include: VVeeva Systems, Kalypso (Rockwell Automation), DDi, Körber AG, ArisGlobal, PhlexGlobal, AmpleLogic, Ennov, MasterControl Solutions, Inc., Rimsys, Ithos Global Inc. (Cordance Group), LORENZ Life Sciences Group, IQVIA, EXTEDO
b. North America dominated the regulatory information management system market with a share of 36.83% in 2025. Given the well-established and technologically advanced pharmaceutical enterprises in the U.S. and Canada, alongside the expanding medical device sector, North America is expected to have a significant presence in the global regulatory information management system market in 2023. Moreover, the region benefits from numerous suppliers offering integrated regulatory solutions such as RIM and regulatory submissions.
b. Key factors driving the market growth include the growing volume of global regulatory submissions, digital transformation of regulatory and compliance processes, rising adoption of cloud-based software solutions.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









