- Home
- »
- Clinical Diagnostics
- »
-
Rapid Tests Market Size And Share, Industry Report, 2033GVR Report cover
![Rapid Tests Market Size, Share & Trends Report]()
Rapid Tests Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Instruments, Consumables), By Technology (Immunoassay, Molecular Diagnostics), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-028-8
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Rapid Tests Market Summary
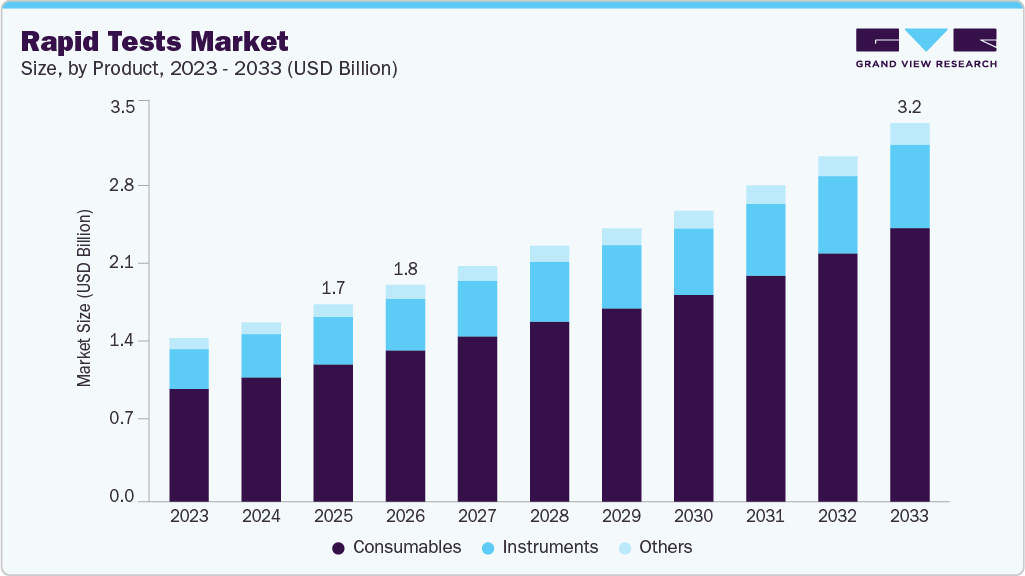
The global rapid tests market size was estimated at USD 1.69 billion in 2025 and is projected to reach USD 3.25 billion by 2033, growing at a CAGR of 8.30% from 2026 to 2033. The growth can be attributed to the growing geriatric population, rising target disease burden, and increasing adoption of self-testing & point-of-care products.
Key Market Trends & Insights
- By product, the consumable segment led the market with the largest revenue share of 69.51% in 2025.
- By technology, the immunoassay-based products segment led the market with the largest revenue share of 53.06% in 2025.
- Based on application, the upper respiratory tract infections segment led the market with the largest revenue share of 40.17% in 2025.
- Based on end use, the hospitals & clinics segment led the market with the largest revenue share of 39.37% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.69 Billion
- 2033 Projected Market Size: USD 3.25 Billion
- CAGR (2026-2033): 8.30%
- North America: Largest market in 2025
Upper respiratory tract infections (URIs) remain extremely common worldwide, with incidence rates exceeding 162,000 per 100,000 people in recent global estimates, underscoring the need for rapid differentiation of viral versus bacterial causes in clinical practice.Upper respiratory tract infections (URTIs) remain a major global health concern in 2025, with high incidence rates particularly in children under 5 and adults with underlying conditions. Children under 5 are the most affected group for URTIs, while 97% of pediatric RSV deaths occur in low- and middle-income countries.
Furthermore, Respiratory syncytial virus (RSV) is a highly prevalent viral infection that particularly affects infants, young children, and older adults. Globally, RSV is estimated to cause approximately 33 million acute lower respiratory tract infections annually in children under five years, with over 3 million cases requiring hospitalization. In the U.S., RSV leads to 58,000-80,000 hospitalizations and 100-300 deaths among children under five each year, as well as 60,000-160,000 hospitalizations and 6,000-10,000 deaths among adults aged 65 and older. Seasonal outbreaks typically occur in the fall and winter, significantly impacting healthcare systems and underscoring the critical need for effective diagnostic and preventive measures.
Below is a comparison of Virus detections by subtype reported to FluNet, 01 October 2017 to 30 December 2024, from countries in the Northern hemisphere, as of 07 January 2025:
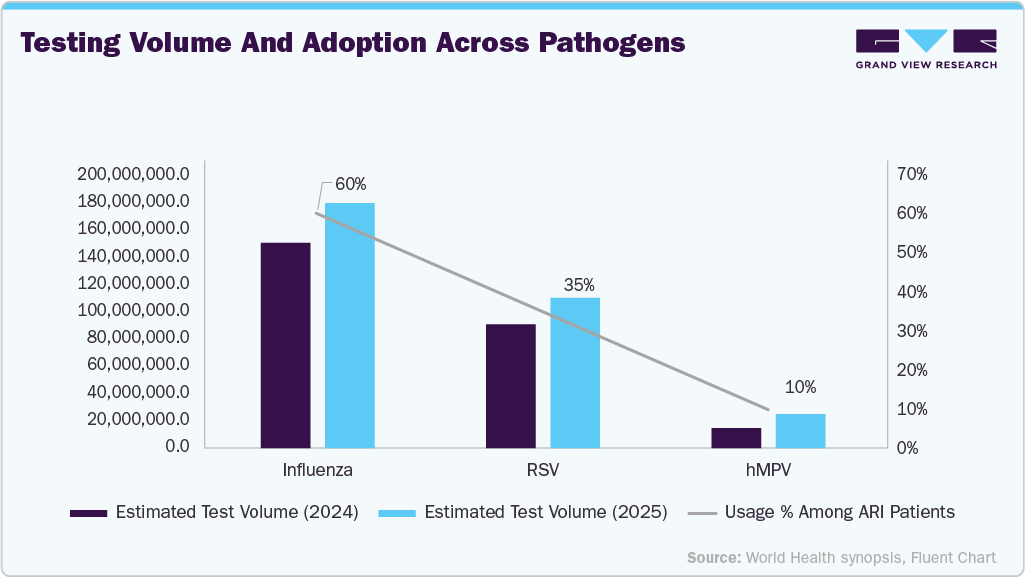
The dataset plots estimated global volumes of rapid diagnostics that correspond with observed respiratory pathogen activity:
-
Influenza rapid test demand accounts for the largest share of total respiratory testing due to its high seasonal prevalence and broad availability of RIDTs. This results in high growth of the rapid influenza diagnostic tests market over the forecast years.
-
RSV rapid test demand is significant and growing, driven by pediatric and high-risk population screening.
-
hMPV contributes a smaller but meaningful share, especially where multiplex testing platforms incorporate hMPV targets.
Collectively, these drivers reflect how seasonal respiratory trends translate into diagnostic demand, highlighting the seasonality and pathogen-specific test utilization that underpins market expansion.
Across infectious disease surveillance and public health programs, rapid tests are increasingly playing a strategic role. Governments and health authorities in 2025 are scaling up access to rapid diagnostic kits for endemic conditions beyond COVID-19; for example, the Indian Council of Medical Research (ICMR) has advocated rapid test availability for conditions such as Hepatitis B, sickle cell anemia, and syphilis at primary health centres, aiming to improve early diagnosis in underserved areas. Meanwhile, markets for specific rapid tests, such as rapid influenza diagnostic tests (RIDTs), are projected to grow steadily as seasonal flu and respiratory illnesses persist globally. This widening scope of disease targets is expanding the application base of rapid diagnostics beyond upper respiratory infections and sepsis screening into the arenas of infectious and non-communicable diseases.
Innovation and competitive dynamics are reshaping the competitive landscape and product development trajectory. Providers are increasingly integrating advanced technologies such as AI, machine learning, and microfluidics to enhance test sensitivity, multiplexing, and ease of use, which supports broader adoption in both clinical and at-home settings. Established diagnostic players and new entrants are also influencing market growth paths: for instance, Amazon’s entry into home diagnostics services across major Indian cities in mid-2025 triggered notable shifts in investor sentiment in the diagnostics sector. At the same time, large strategic moves like Abbott’s planned acquisition of Exact Sciences-makers of widely used cancer and genetic tests signal diagnostic companies’ push to diversify beyond traditional rapid infectious disease testing into broader screening portfolios, even as they balance quality assurance and regulatory challenges in an increasingly crowded market.
Furthermore, the introduction of mobile applications, such as cobas infinity point-of-care from Roche Diagnostics, to satisfy the requirements of the POC coordinator and aid in enhancing productivity by completing the task on the smartphone irrespective of the destination, can be expected to drive the adoption of POC molecular diagnostic products during the forecast period.
Key players have shifted their focus to commercializing home-based self-testing products in recent years, and the COVID-19 outbreak has propelled this trend. Amid government-imposed lockdowns, such products addressed clinical needs while promoting self-quarantine practices. Companies, such as Abbott, have products like BinaxNOW Card rapid test for influenza and COVID-19 that delivers results at home in 15 minutes. This product comes with a virtual guide and application to enable the testing process and display test results.
Market Concentration & Characteristics
The degree of innovation in the global rapid diagnostics tests market is high, as companies continuously develop and introduce more advanced, sensitive, and rapid diagnostic technologies. For example, the use of molecular diagnostics like RT-PCR has significantly improved detection accuracy, while rapid antigen tests and point-of-care (POC) devices enable faster results for timely patient management. Furthermore, the integration of AI and automation into diagnostic workflows has further enhanced the efficiency and accuracy of RSV testing, indicating a strong push for innovation in this space.
The level of mergers and acquisitions (M&A) activities in the rapid tests industry is medium. While major diagnostic companies have made strategic acquisitions to strengthen their product portfolios and expand market reach, such as Abbott's acquisition of Alere, the market is not seeing a high volume of M&A activity. Companies are primarily focusing on organic growth through research and development or partnerships to innovate and capture market share.
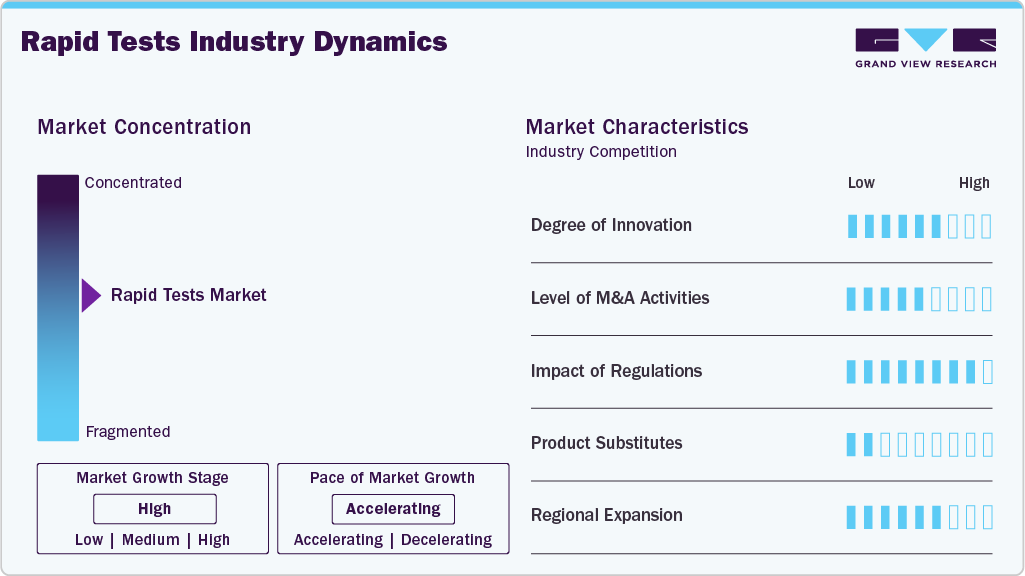
The impact of regulation is high, as regulatory bodies such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) play a critical role in ensuring the safety and effectiveness of diagnostic tests. Regulation plays a major role in shaping the rapid diagnostics market, as governments are strengthening validation and quality requirements to ensure test accuracy and reliability. Health authorities, such as the Indian Council of Medical Research (ICMR), continue to publish updated guidance on diagnostic evaluation and the appropriate use of rapid tests, especially for infectious diseases and antimicrobial resistance surveillance. These regulatory frameworks improve patient safety but also increase compliance and approval timelines for manufacturers.
Product expansion remains a key growth driver, with manufacturers increasingly launching rapid tests for broader applications, including influenza, RSV, streptococcus, hepatitis, and sepsis-related infections. Governments are also supporting wider access to rapid diagnostic kits at primary healthcare levels to improve early detection. This expansion of disease coverage is increasing the relevance of rapid testing across both acute and chronic care pathways.
Regional expansion is accelerating as emerging economies invest in decentralized healthcare access and infectious disease control. Countries in Asia and the Middle East are promoting rapid testing adoption through public health programs and improved primary care infrastructure. At the same time, North America and Europe continue to lead in advanced molecular rapid platforms, driven by strong hospital demand and regulatory oversight, supporting a globally expanding footprint for rapid diagnostics.
Product Insights
The consumables segment led the rapid diagnostic tests market with the largest revenue share of 69.51% in 2025. This can be attributed to high testing rates for the diagnosis of upper respiratory diseases and the commercialization of several PoC assays. Moreover, growing product launches and R&D initiatives pertaining to molecular diagnostic technologies, and rising demand for the Point-of-Care (POC) & self-test products, are expected to boost market growth in the forecast period. Whereas, Antibiotic-resistant infections are a major issue in the world, thereby boosting demand for consumables globally. Organizations are actively involved in addressing the issue. For instance, in April 2021, BD MAX’s Molecular Multi-Drug Resistant Tuberculosis (MDR-TB) Assay was included by WHO in its moderate-complexity automated NAAT MDx technologies, recognized for its high accuracy in TB testing, and was included under the updated guidelines on TB diagnostic tests.
Recently, in October 2025, Xpedite Diagnostics announced the launch of SwiftX Sepsis, an ultrasensitive pathogen cell-capture DNA extraction kit designed to isolate bacterial and fungal DNA directly from whole blood samples without the need for culture enrichment. The kit uses proprietary cell-capture chemistry optimized to selectively bind microbial cells even at very low concentrations (below 10 CFU/mL), overcoming a key challenge in sepsis research where human cell background often masks pathogen signal.
The instruments segment is also anticipated to grow at the fastest CAGR over the forecast period, driven by the increasing adoption of automated and multiplex molecular diagnostic platforms in hospitals and laboratories. Advanced systems that deliver faster turnaround times and higher sensitivity are gaining traction, especially for respiratory and syndromic testing.
A recent example of product advancement was seen in January 2024, when QIAGEN launched new QIAstat-Dx syndromic PCR panels in India, strengthening access to rapid molecular diagnostics for gastrointestinal and CNS infections. This highlights how innovation in testing instruments continues to expand the market beyond traditional rapid kits.
Technology Insights
The immunoassay segment led the market with the largest revenue share of 53.06% in 2025. Multiple rapid immunoassays with high efficacy are available on the market. Immunoassay-based rapid diagnostics for RSV include Directigen, QuickVue, Sofia, and BD Veritor, which are next-generation lateral flow Digital Immunoassay (DIA) techniques for the qualitative detection of RSV viral antigens. In addition, rapid RSV antigen is the most widely used test. Further, immunoassays such as chromatographic immunoassays offer high sensitivity and specificity for detecting RSV antigens, making them particularly valuable for the rapid diagnosis of acute respiratory infections, especially in pediatric and geriatric populations. These tests detect RSV antigens in patient samples, providing results in as little as 15-30 minutes, which is crucial for the timely management of patients.
Products such as the BinaxNOW RSV test from Abbott and the Sofia RSV FIA from Quidel are examples of successful chromatographic immunoassay-based diagnostics. These tests have contributed significantly to the market due to their cost-effectiveness compared to molecular assays, making them accessible to a broader range of healthcare providers.
The molecular diagnostics segment is anticipated to grow at the fastest CAGR over the forecast period. In the rapid tests industry, molecular diagnostics products have a substantial presence, which is expected to drive the market in the coming years. The FDA has approved RT-PCR-based POC assays, such as Focus Diagnostics' Simplexa Flu A/B & RSV, BioFire’s Film Array RVP, and Cepheid's GeneXpert Flu A/B, for the detection of pneumonia or streptococcus infections. In January 2024, QIAGEN introduced two new multiplex real-time PCR panels for its QIAstat-Dx platform in India: A Gastrointestinal Panel 2 and a Meningitis/Encephalitis Panel, joining its existing respiratory SARS-CoV-2 test. These syndromic panels detect multiple pathogens simultaneously (e.g., 15 CNS pathogens, 22 GI agents) in about 60-80 minutes, enabling faster, more accurate clinical decisions than traditional culture methods.
Application Insights
The upper respiratory tract infections segment led the rapid diagnostic tests market with the largest revenue share of 40.17% in 2025. URTIs remain one of the most common reasons for outpatient visits and hospital triage, driving strong demand for rapid diagnostic solutions that can quickly differentiate between viral and bacterial infections. Rapid diagnostic tests (RDTs) for upper respiratory tract infections (URTIs) are evolving toward multiplex molecular assays, offering fast, accurate, and near-patient detection of viruses such as RSV, influenza, and COVID-19 to improve antibiotic stewardship. Recent news highlights that rapid molecular testing, such as Xpert Xpress Flu/RSV, can significantly reduce unnecessary antibiotic prescriptions in pediatric outpatients. This is expected to drive the rapid influenza diagnostic tests market
The antibiotic-resistant infections segment is anticipated to grow at the fastest CAGR over the forecast period. Rising antimicrobial resistance has increased the urgency for rapid diagnostics that can guide appropriate antibiotic use and reduce unnecessary prescriptions. Public health agencies globally continue to emphasize diagnostic-driven stewardship strategies. In line with this preparedness focus, in April 2024, HealthTrackRx partnered with the U.S. CDC to launch a rapid PCR test for H5N1, demonstrating how molecular rapid testing is expanding to address emerging infectious threats and support surveillance efforts.
End Use Insights
The hospitals & clinics segment led the market with the largest revenue share of 39.37% in 2025, driven by the critical need for rapid diagnostic results in emergency care, inpatient management, and infection control. Hospitals rely heavily on rapid tests for respiratory infections, sepsis screening, and antimicrobial decision-making, where immediate results can directly impact treatment pathways and patient outcomes. The availability of rapid molecular platforms within clinical environments has further strengthened hospital demand, supporting the segment’s dominant market position.
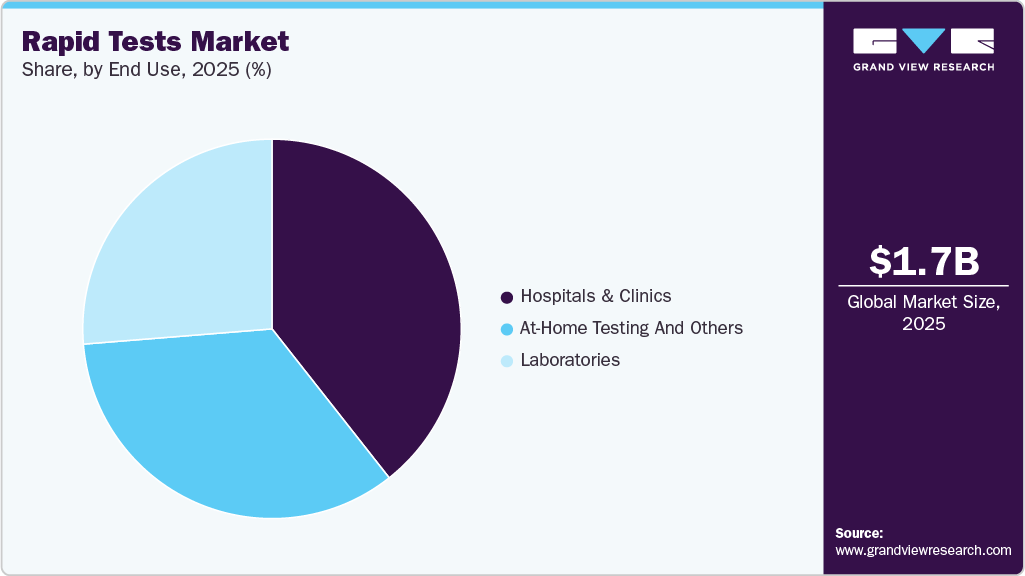
The at-home testing and others segment is anticipated to grow at the fastest CAGR over the forecast period, owing to increasing consumer awareness, convenience-driven healthcare behavior, and the expanding availability of self-testing kits, which are accelerating this trend. At-home rapid diagnostics are becoming more relevant not only for infectious diseases but also for routine monitoring and early detection outside traditional healthcare facilities. The broader shift toward decentralized care, supported by digital health integration and retail distribution, is expected to drive the continued growth of home-based rapid testing solutions globally.
Regional Insights
North America dominated the global rapid tests market with the largest revenue share of 48.48% in 2025. The growth can be attributed to the high incidences of upper respiratory diseases & antibiotic-resistant infections, high healthcare expenditure, and the launch of technologically advanced products. Furthermore, supportive government initiatives and a rise in awareness about the use of PoC & rapid diagnostics for the diagnosis of various diseases in the region are fueling the market growth.
U.S. Rapid Tests Market Trends
The rapid tests market in the U.S. accounted for the largest market revenue share in North America in 2025. The growth can be attributed to the high incidences of upper respiratory diseases, sepsis & antibiotic-resistant infections, high healthcare expenditure, and the launch of technologically advanced products.
In June 2025, a real-world clinical study at a major U.S. academic medical center reported that implementing a rapid sepsis diagnostic test was associated with a 42% reduction in mortality and a mean 2-day decrease in hospital length of stay among patients with suspected sepsis. The test enables clinicians to identify sepsis earlier than traditional culture-based methods, prompting faster initiation of appropriate antimicrobial therapy and supportive care.
Further, in August 2025, the U.S. Food and Drug Administration (FDA) granted clearance to Triverity’s rapid sepsis diagnostic test (the Exact Dx sepsis assay) for use in acute care settings. This clearance allows the test to be used at or near the point of care to rapidly detect biomarkers associated with sepsis, supporting clinicians in early diagnosis and risk stratification. These developments underscore a broader shift in the United States toward rapid, actionable diagnostics in critical care. Reduced mortality and shorter hospital stays directly enhance clinical and operational value, incentivizing uptake of advanced sepsis tests across hospital systems.
Europe Rapid Tests Market Trends
The rapid tests market in Europe is anticipated to grow at a considerable CAGR during the forecast period, driven by the presence of developed economies such as Germany, Spain, the UK, France, and Italy. The increasing prevalence of chronic diseases and rising technological advancements are among the key factors driving the market in the region. The main cause of death in women in Europe is breast cancer, which is the underlying reason for the high demand for these tests leading to a positive impact on rapid cancer tests market.
The UK rapid tests market is driven by the increasing prevalence of respiratory infectious diseases, technological advancements, and the development of healthcare infrastructure. Seasonal surges in influenza, RSV, and other upper respiratory infections have increased demand for rapid antigen and molecular point-of-care testing, particularly in hospitals and community care settings. The UK is also witnessing the steady adoption of multiplex respiratory panels that enable clinicians to differentiate multiple pathogens in a single test run, improving treatment decisions and reducing unnecessary antibiotic use. Technological advancements, including faster PCR-based platforms and improved home-testing options, are expanding rapid testing beyond traditional laboratories.
The rapid tests market in Germany is driven by advancements in technology and investments made by various organizations toward RSV and URT screening initiatives. Germany also benefits from high healthcare spending and a well-established reimbursement environment, enabling faster integration of advanced point-of-care diagnostics. Recent innovation trends include syndromic testing panels that can detect multiple respiratory pathogens simultaneously, improving clinical workflow efficiency. With ongoing investments in infectious disease surveillance and healthcare modernization, Germany is expected to remain a key European market for rapid diagnostics, particularly in hospital-based and emergency care applications.
Asia Pacific Rapid Tests Market Trends
The rapid tests market in the Asia Pacific is estimated to grow at the fastest CAGR during the forecast period. The region is witnessing strong uptake of molecular rapid testing platforms as governments and private providers invest in advanced laboratory capabilities. The expansion of home-testing models and pharmacy-based diagnostics is also accelerating market penetration, particularly in urban centers. Overall, Asia Pacific’s combination of large unmet diagnostic needs, improving healthcare infrastructure, and rapid technology adoption positions it as a high-growth region in the global rapid tests landscape.
The China rapid tests market is being driven by various factors. The country has expanded its focus on infectious disease preparedness, encouraging wider adoption of rapid antigen and molecular diagnostic tools in hospitals and public health settings. Rapid testing plays a key role in managing seasonal influenza outbreaks, respiratory syndromes, and broader infection control efforts. China also has a strong domestic diagnostics manufacturing base, supporting the availability of cost-effective rapid test kits and consumables.
The rapid tests market in Japan is one of the most advanced countries in this region. Japan’s market is expected to grow rapidly. Hospitals and laboratories in Japan are increasingly adopting high-performance molecular rapid testing platforms that deliver quick, reliable results, enabling faster clinical decision-making. Japan also benefits from strong regulatory oversight and high-quality healthcare delivery systems, which encourage the adoption of validated rapid diagnostic technologies.
Latin America Rapid Tests Market Trends
The rapid tests market in Latin America has exhibited significant growth in the past few years. Rapid testing is increasingly being adopted for respiratory infections, sexually transmitted diseases, and hospital-acquired infections, particularly in urban healthcare centers. Many countries in the region are working to strengthen diagnostic capabilities through public health initiatives and the expansion of private laboratories.
The Brazil rapid tests market is anticipated to grow at a significant CAGR during the forecast period. The Brazil has the largest population, and respiratory infections remain a major driver of demand for rapid antigen and molecular testing, particularly in hospital and emergency care environments. In addition, respiratory infections remain a major driver of demand for rapid antigen and molecular testing, particularly in hospital and emergency care environments.
Middle East and Africa Rapid Tests Market Trends
The rapid tests market in Middle East & Africa are strengthening diagnostic preparedness for respiratory infections, antimicrobial resistance, and hospital-acquired infections. Rapid testing is particularly important in regions where access to centralized laboratories may be limited, making decentralized and portable diagnostic solutions essential.
The Saudi Arabia rapid testing market is expanding due to increased healthcare modernization, rising awareness of infectious diseases, and government-led investments under Vision 2030. Rapid diagnostics are gaining adoption across hospitals and clinical laboratories, particularly for respiratory infections, sepsis screening, and antimicrobial stewardship efforts. The country is also strengthening its public health preparedness, supporting demand for advanced molecular rapid testing platforms that deliver faster, more accurate results. Key players in the Saudi rapid diagnostics space include global companies such as Abbott, Roche, BD, Siemens Healthineers, and bioMérieux, as well as regional distributors and healthcare providers. Growth is further supported by the expansion of private healthcare facilities and diagnostic chains.
Key Rapid Tests Company Insights
The major market players are focused on adopting strategic initiatives such as launches, mergers & acquisitions, and partnerships. Furthermore, several players are focusing on the development of testing services, thereby boosting the demand.
For instance, in June 2023, Thermo Fisher Scientific, Inc. collaborated with Pfizer, Inc. to enhance the NGS testing for patients suffering from breast and lung cancer.
Key Rapid Tests Companies:
The following key companies have been profiled for this study on the rapid tests market.
- BD (Becton, Dickinson and Company)
- bioMérieux SA
- Thermo Fisher Scientific, Inc.
- F. Hoffmann-La Roche Ltd.
- Danaher Corporation
- DiaSorin S.P.A
- Abbott
Recent Developments
-
In February 2026, MedGenome unveiled MetaSeq, a metagenomic next-generation sequencing (mNGS) test that detects over 1,400 clinically relevant pathogens directly from blood by analysing microbial cell-free DNA. This culture-free assay, which delivers results in about 5 days, is positioned to overcome the limitations of conventional blood culture methods, particularly in the diagnosis of bloodstream infections (BSIs).
-
In November 2025, the U.S. Food and Drug Administration awarded Breakthrough Device designation to a new molecular diagnostic platform developed by Nanopath for detecting complicated urinary tract infections (cUTIs) directly from urine samples. Designation recognizes tests that could significantly improve the diagnosis or treatment of serious conditions and provides Nanopath with access to accelerated regulatory feedback and a prioritized review pathway.
-
In August 2025, U.S. Food and Drug Administration (FDA) granted clearance to Triverity’s rapid sepsis diagnostic test (the Exact Dx sepsis assay) for use in acute care settings. This clearance allows the test to be used at or near the point of care to rapidly detect biomarkers associated with sepsis, supporting clinicians in early diagnosis and risk stratification
-
In January 2025, FDA cleared the Inflammatix TriVerity first-in-class molecular test received 510(k) clearance, analyzing 29 immune-related genes in blood to differentiate bacterial/viral infections and provide a rapid risk score for sepsis.
Rapid Tests Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.86 billion
Revenue forecast in 2033
USD 3.25 billion
Growth rate
CAGR of 8.30% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, technology, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Spain; Italy; Denmark; Norway; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
BD (Becton, Dickinson and Company); bioMérieux SA; Thermo Fisher Scientific, Inc.; F. Hoffmann-La Roche Ltd.; Danaher Corporation; Diasorin S.P.A; Abbott.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Rapid Tests Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 - 2033. For this report, Grand View Research has segmented the global rapid tests market report based on the product, technology, application, end use, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Consumables
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Immunoassay
-
Molecular Diagnostics
-
Other technologies
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Upper respiratory tract infections
-
Influenza and Parainfluenza virus
-
Streptococcus
-
Respiratory syncytial virus
-
-
Antibiotic resistant infections
-
Sepsis
-
Bacterial Sepsis
-
Fungal Sepsis
-
Others
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinics
-
Laboratories
-
At-home testing and Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players in the rapid tests market include bioMérieux SA; Thermo Fisher Scientific, Inc.; F. Hoffmann-La Roche Ltd.; Danaher Corporation; DiaSorin S.P.A; and Abbott amongst others.
b. Key factors driving the rapid tests market growth include the rising incidence of target diseases and increasing demand for rapid diagnosis.
b. The global rapid tests market size was estimated at USD 1.69 billion in 2025 and is expected to reach USD 1.86 billion in 2026.
b. The global rapid tests market is expected to grow at a compound annual growth rate of 8.3% from 2026 to 2033 to reach USD 3.25 billion by 2033.
b. Based on technology, the immunoassay segment dominated the market with a share of 53.06% in 2025, owing to the availability of multiplex tests for the simultaneous diagnosis of flu, RSV, and COVID-19.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









