- Home
- »
- Clinical Diagnostics
- »
-
Pyrogen & Endotoxin Testing Market, Industry Report, 2033GVR Report cover
![Pyrogen And Endotoxin Testing Market Size, Share & Trends Report]()
Pyrogen And Endotoxin Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Instruments, Consumables, Services), By Technology (Bacterial Endotoxin Testing, Recombinant Endotoxin Detection), By Application, By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-873-6
- Number of Report Pages: 140
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Pyrogen & Endotoxin Testing Market Summary
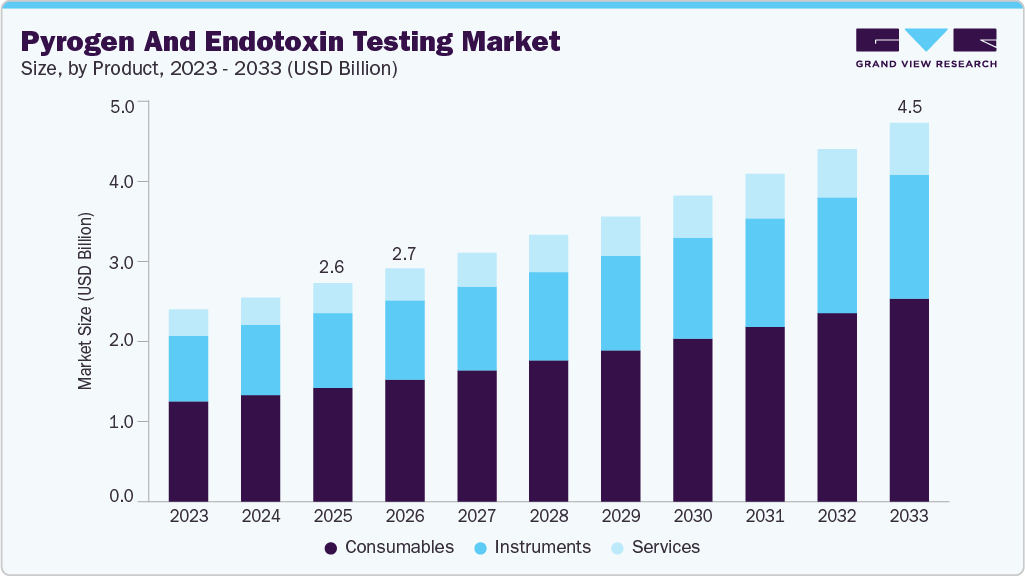
The global pyrogen and endotoxin testing market size was estimated at USD 2.56 billion in 2025 and is projected to reach USD 4.46 billion by 2033, growing at CAGR of 7.22% from 2026 to 2033. The increasing demand for pyrogen and endotoxin testing products in the pharmaceutical and biotechnology industries is a significant driver of market growth.
Key Market Trends & Insights
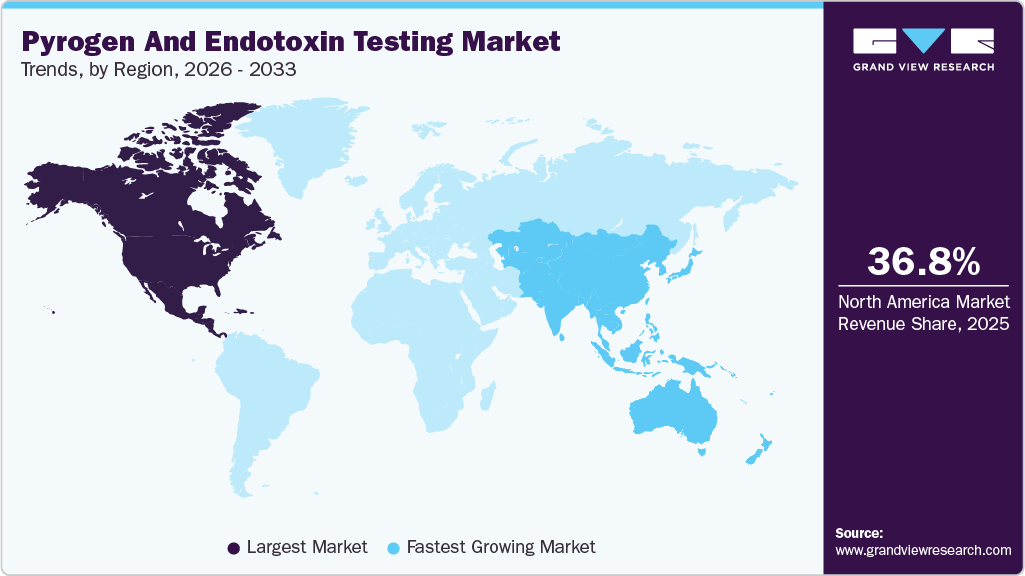
- North America dominated the global pyrogen and endotoxin testing market with the largest revenue share of 36.78% in 2025.
- The pyrogen and endotoxin testing industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product, the consumables segment led the market with the largest revenue share of 52.25% in 2025.
- By technology, the bacterial endotoxin testing (BET) segment led the market with the largest revenue share of 65.08% in 2025.
- By application, the product safety & batch release testing segment led the market with the largest revenue share of 42.89% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.56 Billion
- 2033 Projected Market Size: USD 4.46 Billion
- CAGR (2026-2033): 7.22%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The rapid expansion of these industries, coupled with the rising number of new therapeutic launches, is expected to propel the demand for pyrogen and endotoxin testing products further. This growth highlights the critical role of pyrogen and endotoxin testing in ensuring the safety and efficacy of pharmaceutical and biotechnological products.Several regulatory bodies and governments are taking various initiatives to train and create awareness about pyrogen and endotoxin testing. Government initiatives worldwide are pivotal in accelerating this transition. In June 2024, the 179th session of the European Pharmacopoeia (Ph. Eur.) Commission marked a significant advancement for animal welfare and contemporary pyrogen and endotoxin testing practices. During this session, the Commission approved the removal of the Rabbit Pyrogen Test (RPT) from 57 revised texts and introduced a comprehensive new general chapter on Pyrogenicity. This decision effectively abolished the RPT requirement across all Ph. Eur. texts, solidifying the transition toward in vitro testing methodologies, such as the Monocyte Activation Test (MAT).
The scope of the pyrogen and endotoxin testing industry is expanding significantly as technological advancements improve testing sensitivity, accuracy, and compliance with evolving regulatory standards. Increasing emphasis on ensuring the safety of pharmaceuticals, biologics, vaccines, and medical devices has driven the demand for advanced testing platforms capable of detecting both endotoxin and non-endotoxin pyrogens. The industry is gradually transitioning from traditional animal-based methods toward modern in vitro technologies that offer faster turnaround times and improved reproducibility. For instance, in May 2025, Minerva Biolabs GmbH introduced the NAT-MAT system, a next-generation pyrogen-detection platform that integrates the Monocyte Activation Test (MAT) with digital PCR. The system measures cytokine gene expression, including IL-1β and TNF-α, enabling more sensitive and rapid detection compared to conventional ELISA-based MAT assays. Such innovations highlight the expanding scope of advanced molecular and cell-based diagnostic technologies within pharmaceutical quality control processes.
In January 2026, BETMAT Biotechnology launched high-performance sustainable endotoxin testing services designed to meet evolving quality control requirements in pharmaceutical and medical device manufacturing. The new service focuses on providing reliable bacterial endotoxin testing (BET) solutions that comply with international pharmacopeial standards while supporting sustainability initiatives in laboratory testing. The service launch reflects the growing demand for efficient, standardized, and environmentally responsible endotoxin-detection technologies in pharmaceutical manufacturing. BETMAT’s testing infrastructure incorporates advanced automated instrumentation and strict quality management systems aligned with global pharmacopeial requirements, including USP, EP, BP, and ChP, ensuring traceable and reliable results.
Furthermore, the development of integrated testing platforms is strengthening the role of automated, standardized pyrogen-detection solutions across pharmaceutical manufacturing workflows. The NAT-MAT platform includes ready-to-use macrophage cells, nucleic acid extraction kits, and a triplex digital PCR assay, enabling reliable and automated testing for both quality control and in-process monitoring. These developments reflect the growing shift toward comprehensive testing systems that can improve efficiency and reduce manual laboratory procedures.
In addition, the ability of such technologies to support complete pyrogen and endotoxin testing creates opportunities to gradually replace traditional animal-based methods, such as the Rabbit Pyrogen Test. As regulatory agencies and industry stakeholders increasingly promote animal-free testing approaches, the adoption of advanced in vitro pyrogen detection technologies is expected to expand, thereby broadening the market scope and encouraging continued innovation in endotoxin and pyrogen and endotoxin testing solutions.
Increasing investment in R&D by the government and manufacturing companies for product development is one of the major factors contributing to the market's growth. New product development and launch is a key strategy adopted by manufacturers to maintain their dominance and to get maximum revenue share in the sector. As stated by the European Federation of Pharmaceutical Industries and Associations (EFPIA), in 2023, the research-based pharmaceutical industry invested around USD 48.65 billion in R&D in Europe in 2022. Hence, the need for pyrogen and endotoxin testing products is anticipated to upsurge during the forecast period.
Furthermore, the pyrogen and endotoxin testing industry is driven by the increasing demand for therapeutic drugs. Several companies are actively involved in developing new drug therapies. For instance, in 2023, the FDA approved 55 new drugs. Pyrogen testing in drug development assesses the presence or absence of pyrogens in parenteral pharmaceutical products. Moreover, the sterility of any drug does not indicate that it is pyrogen-free. Therefore, drugs that are expected to be sterile must be tested for the presence of pyrogens to control febrile reactions in patients.
Market Characteristics & Concentration
The market growth stage is medium, and the pace is accelerating. The innovation in the pyrogen and endotoxin testing industry is driven by technological advancements, rising demand for pyrogen and endotoxin testing, and the need for animal-free, reliable testing methods. The degree of innovation in the pyrogen and endotoxin testing industry is considered medium to high, with the pace of technological advancement steadily accelerating as the pharmaceutical and biotechnology industries expand globally. The need for reliable detection of bacterial endotoxins and other pyrogens in parenteral drugs, biologics, vaccines, and medical devices has encouraged the development of more advanced testing technologies. Traditional methods such as the Limulus Amebocyte Lysate (LAL) assay and Rabbit Pyrogen Test have long served as industry standards; however, the market is witnessing a gradual shift toward innovative in vitro testing approaches that offer greater sensitivity, reproducibility, and faster turnaround times.
The pyrogen and endotoxin testing industry sees high levels of mergers and acquisitions (M&A) activity as leading life sciences and diagnostic companies aim to strengthen their technological capabilities and expand their testing portfolios. Large pharmaceutical testing service providers and laboratory technology companies frequently pursue strategic acquisitions to broaden their assay menus, improve testing accuracy, and enhance laboratory automation capabilities. These transactions enable companies to integrate advanced endotoxin detection technologies, strengthen regulatory compliance expertise, and expand service offerings across the pharmaceutical, biotechnology, and medical device industries.

The pyrogen and endotoxin testing industry is subject to stringent regulatory oversight due to the critical role these tests play in ensuring the safety of pharmaceutical products, biologics, vaccines, and medical devices. Regulatory agencies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and other international health authorities require strict compliance with quality standards before pharmaceutical products can enter the market. These agencies mandate comprehensive testing protocols to detect endotoxins and pyrogens during drug manufacturing, batch release, and quality control processes.
Product expansion in the pyrogen and endotoxin testing industry is accelerating as companies continue to broaden their portfolios of testing kits, reagents, instruments, and automated detection systems. Increasing pharmaceutical production and the rapid growth of biologics and injectable drugs are driving the demand for diverse testing solutions capable of ensuring sterility and product safety. Manufacturers are introducing new reagents and assay kits designed to improve sensitivity, reduce testing time, and enhance laboratory efficiency. The expansion of recombinant endotoxin detection technologies, particularly recombinant Factor C assays, represents a key product innovation aimed at reducing dependence on traditional animal-derived reagents.
Regional expansion plays a crucial role in the growth of the pyrogen and endotoxin testing industry as companies seek to capitalize on emerging opportunities in rapidly developing pharmaceutical manufacturing hubs. While North America and Europe remain mature markets with well-established regulatory frameworks and advanced laboratory infrastructure, significant growth opportunities are emerging in Asia-Pacific, Latin America, and the Middle East. Countries such as China, India, and South Korea are experiencing rapid expansion of pharmaceutical and biotechnology industries, supported by increasing government investment in healthcare infrastructure and drug manufacturing capabilities.
Product Insights
The consumables segment led the market with the largest revenue share of 52.25% in 2025 and is projected to grow at the fastest CAGR during the forecast period. The growing adoption of in vitro and non-animal testing methods has intensified the demand for advanced consumables. In June 2024, FUJIFILM Wako Chemicals launched the PYROSTAR Neo+ and LumiMAT, advancing the market with innovative, sustainable consumables. These products exemplify the shift toward recombinant and animal-free testing methods, reflecting the industry's focus on innovation, regulatory compliance, and sustainability.
The instruments segment is expected to grow at a significant CAGR during the forecast period, owing to the technological advancements in products. For instance, the PyroDetect System is a verified non-animal alternative to the rabbit test, offering high-quality in vitro detection of both non-endotoxin and endotoxin contamination.
Technology Insights
The bacterial endotoxin testing (BET) segment led the market with the largest revenue share of 65.08% in 2025. The segment’s dominance is primarily attributed to its long-standing regulatory acceptance and widespread use in pharmaceutical and medical device quality control processes. BET methods such as gel clot, chromogenic, and turbidimetric assays remain the standard approaches recommended in pharmacopeial guidelines, including the United States Pharmacopeia and the European Pharmacopoeia, making them essential for batch release testing of injectable drugs, vaccines, and biologics. Pharmaceutical manufacturers rely heavily on BET technologies to detect endotoxin contamination in sterile drug formulations, ensuring compliance with global regulatory standards. For instance, in 2025, BETMAT Biotechnology launched high-performance endotoxin testing services using validated BET methods, including kinetic turbidimetric, kinetic chromogenic, and gel clot assays, to support pharmaceutical and medical device manufacturers.
The recombinant endotoxin detection segment is anticipated to grow at the fastest CAGR over the forecast period. This growth is driven by the increasing demand for sustainable and animal-free testing alternatives. Recombinant Factor C (rFC) assays eliminate the need for horseshoe crab blood used in traditional LAL tests, addressing both environmental concerns and supply chain limitations. Pharmaceutical companies and regulatory agencies are gradually recognizing recombinant detection technologies as reliable alternatives for endotoxin detection. Continuous innovation in recombinant testing platforms and improvements in the sensitivity of these assays are expected to significantly accelerate their adoption in pharmaceutical quality control workflows.
Application Insights
The product safety & batch release testing segment led the market with the largest revenue share of 65.08% in 2025, driven by stringent regulatory requirements that mandate endotoxin and pyrogen testing before pharmaceutical products are released into the market. Injectable drugs, vaccines, biologics, and implantable medical devices require rigorous quality control procedures to ensure patient safety and prevent adverse immune responses caused by endotoxin contamination. Pharmaceutical manufacturers perform endotoxin testing at multiple stages of drug production, particularly during final product batch release. Regulatory authorities worldwide enforce strict guidelines requiring endotoxin testing to comply with pharmacopeial standards.
The manufacturing process & raw material control segment is anticipated to grow at the fastest CAGR over the forecast period. The need to detect endotoxin contamination early in the drug manufacturing process is becoming increasingly important to avoid costly batch failures and ensure consistent product quality. Pharmaceutical manufacturers are implementing endotoxin testing during raw material screening, water system monitoring, and in-process production stages. This proactive approach helps companies maintain regulatory compliance and reduce the risk of contamination throughout the production lifecycle.
End-use Insights
The pharmaceutical & biopharmaceutical companies segment led the market with the largest revenue share of 46.89% in 2024. The growth is attributed to the rising production of pharmaceuticals, biopharmaceuticals, and other biologic products. Key players offering pyrogen and endotoxin testing instruments include Lonza Group, Thermo Fisher Scientific, and FUJIFILM Wako Chemicals, which are known for their broad portfolio of endotoxin detection products, including LAL-based assays and in vitro pyrogen detection
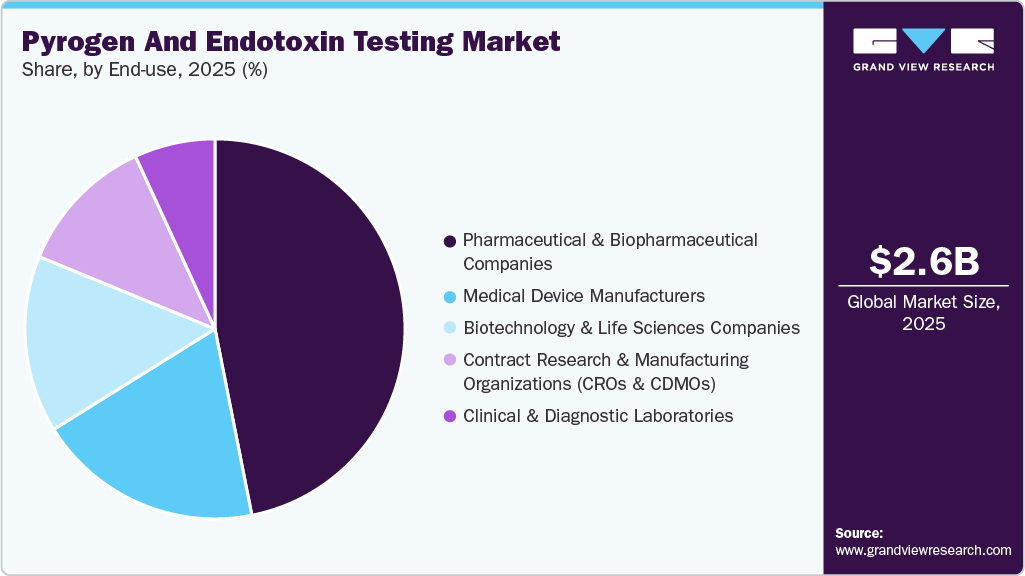
The contract research & manufacturing organizations (CROs & CDMOs) segment is expected to grow at the fastest CAGR over the forecast period. CROs and CDMOs provide specialized testing services, including endotoxin and pyrogen testing, to help pharmaceutical and biotechnology companies meet regulatory requirements. As outsourcing becomes a common strategy for accelerating drug development and reducing operational costs, these organizations are expanding their laboratory testing capabilities and adopting advanced endotoxin detection technologies to meet growing client demand.
Regional Insights
North America pyrogen and endotoxin testing market dominated the global industry with the largest revenue share of 36.78% in 2025. The growth is attributed to the rising number of chronic diseases needing developments in drug therapy. The growing adoption of ethical and efficient in vitro pyrogen tests has become a cornerstone of this dominance. Traditional animal-based tests, such as the Rabbit Pyrogen Test, are being steadily replaced by advanced alternatives, including the Monocyte Activation Test and LAL-based assays, which use chromogenic, turbidimetric, and gel clot methods.
U.S. Pyrogen And Endotoxin Testing Market Trends
The pyrogen and endotoxin testing market in the U.S. accounted for the largest market revenue share in North America in 2025. A recent development in January 2024 exemplifies this evolution: Charles River Laboratories International, Inc. introduced the Endosafe Trillium rCR cartridge, a groundbreaking advancement in BET technology. This innovation integrates their flagship Endosafe cartridge with recombinant cascade reagent (rCR) technology, providing an animal-free alternative that enhances testing efficiency, accelerates manufacturing processes, and supports sustainability initiatives.
Europe Pyrogen And Endotoxin Testing Market Trends
The pyrogen and endotoxin testing market in Europe is characterized by a robust yet intricate landscape, driven by stringent regulatory frameworks, the growth of the pharmaceutical and biotechnology industries, and the increasing adoption of innovative testing methods. Valued significantly in recent years, the market shows strong growth potential fueled by rising investments in healthcare research, expansion of drug development activities, and heightened focus on patient safety
The UK pyrogen and endotoxin testing market is expected to grow at a significant CAGR over the forecast period, driven by the growing number of drug approvals. Demand is primarily driven by the pharmaceutical, biotechnology, and medical device sectors, where ensuring product sterility and safety is paramount.
The pyrogen and endotoxin testing market in France is expanding rapidly. The growth is attributed to the growing initiatives in the therapeutic drugs field. The country is undertaking several initiatives to increase the access to drugs for the targeted patients in France.
The Germany pyrogen and endotoxin testing market is expected to grow at a significant CAGR over the forecast period. The pharmaceutical and biotechnology sectors, which heavily rely on pyrogen and endotoxin testing to ensure the safety of their products, continue to expand in the country. For instance, the 12th PharmaLab Congress will be held in Düsseldorf/Neuss, Germany, from November 25-27, 2024, focusing on advancements in analytical and microbiological quality control for the pharmaceutical industry.
Asia Pacific Pyrogen And Endotoxin Testing Market Trends
The pyrogen and endotoxin testing market in the Asia Pacific is estimated to grow at the fastest CAGR of 8.01% over the forecast period. Many manufacturers target Asian countries, such as China & India, for drug discovery, development, and production. In addition, clinical research organizations are focusing on Asian countries for clinical trials. The availability of less stringent government regulations for drug development, the vast genome pool, and the rapidly developing healthcare infrastructure in this region are some of the major factors responsible for the growth of the sector in this region.
The China pyrogen and endotoxin testing market held a significant share in the Asia Pacific in 2025. The growth is attributed to the increasing presence of key players in the country, owing to the reasonable manufacturing cost. Government policies promoting quality and safety in medical devices and drugs have led to the widespread use of LAL and turbidimetric tests for batch release testing, while the country’s focus on biotechnology innovation encourages the adoption of rabbit tests for specialized products.
The pyrogen and endotoxin testing market in Japan is dominated by global leaders such as Charles River Laboratories and bioMérieux SA, as well as regional players like FUJIFILM Wako Pure Chemical Corporation, which has a strong presence due to its expertise in pyrogen detection kits. In June 2024, FUJIFILM Wako Pure Chemicals Corporation introduced the LumiMAT Pyrogen Detection Kit (LumiMAT) and PYROSTAR Neo+ as innovative alternatives to traditional methods for detecting pyrogens and endotoxins.
Middle East & Africa Pyrogen And Endotoxin Testing Market Trends
The pyrogen and endotoxin testing market in the Middle East and Africa (MEA) includes South Africa, Saudi Arabia, and several emerging pharmaceutical markets across the Gulf Cooperation Council and African countries. Countries such as South Africa and Saudi Arabia are increasingly investing in healthcare infrastructure, pharmaceutical manufacturing, and regulatory modernization, which is driving the demand for endotoxin and pyrogen testing solutions. The region is witnessing growing pharmaceutical imports and domestic production of sterile drugs, vaccines, and medical devices, which require strict quality control procedures, including endotoxin testing. Governments across the region are implementing initiatives to strengthen pharmaceutical production capabilities and reduce reliance on imports, thereby encouraging the establishment of local manufacturing facilities and laboratory testing centers. In addition, international pharmaceutical companies are expanding partnerships with regional manufacturers and contract research organizations to support drug development and manufacturing activities.
The Saudi Arabia pyrogen and endotoxin testing market is expanding due to the country’s growing pharmaceutical manufacturing sector and government initiatives to strengthen domestic drug production. Under the Vision 2030 healthcare transformation strategy, Saudi Arabia is investing heavily in pharmaceutical research, biotechnology development, and advanced healthcare infrastructure. These initiatives are encouraging multinational pharmaceutical companies to establish local manufacturing facilities and collaborate with domestic producers. As the production of injectable drugs, vaccines, and biologics increases, the need for reliable endotoxin and pyrogen testing solutions has become more critical to ensure product safety and regulatory compliance.
Key Pyrogen And Endotoxin Testing Company Insights
Some of the leading players in the pyrogen and endotoxin testing industry are Lonza and Charles River Laboratories. These companies are actively engaged in business initiatives, such as product launches and the acquisition of companies. Moreover, these companies have a global presence, which helps them access affordable manufacturing facilities and a broad customer base. These factors cumulatively strengthen their presence in the pyrogen and endotoxin testing industry.
The emerging players in the pyrogen and endotoxin testing industry mostly focus on partnerships and collaborations to increase their presence in the pyrogen and endotoxin testing industry. Companies generally offer a compact product portfolio and focus on capturing local markets.
Key Pyrogen And Endotoxin Testing Companies:
The following key companies have been profiled for this study on the pyrogen and endotoxin testing market.
- Charles River Laboratories
- bioMérieux
- Associates of Cape Cod, Inc.
- FUJIFILM Wako Pure Chemical Corporation
- Bioendo Technology
- GenScript
- ACROBiosystems
- Thermo Fisher Scientific, Inc.
- Merck KGaA
- Sartorius AG
- Eurofins Scientific
- WuXi AppTec
Recent Developments
-
In May 2025, Minerva Biolabs GmbH announced the launch of the NAT-MAT system, an advanced pyrogen detection platform that combines the Monocyte Activation Test (MAT) with digital PCR technology. The system measures gene expression of cytokines IL-1β and TNF-α, enabling faster and more sensitive detection of both endotoxin and non-endotoxin pyrogens compared to conventional ELISA-based MAT assays.
-
In December 2024, Ellab acquired PharmaProcess in Italy and Switzerland, enhancing its life science services. The partnership integrates PharmaProcess's regulatory expertise with Ellab’s compliance solutions, providing end-to-end support for pharmaceutical and biotech clients in these regions.
-
In September 2024, Lonza Walkersville began the expansion of its endotoxin assay production facility in Walkersville, MD. The 18,000-square-foot upgrade will enhance manufacturing capacity to meet the rising demand for endotoxin assays used in ensuring the safety and compliance of injectable drugs and medical devices.
-
In June 2024, FUJIFILM Wako Pure Chemicals launched two new pyrogen and endotoxin tests: LumiMAT Pyrogen Detection Kit, a next-generation monocyte activation test, and PYROSTAR Neo+, a recombinant protein reagent for bacterial endotoxin detection, available globally in July 2024.
Pyrogen And Endotoxin Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.74 billion
Revenue forecast in 2033
USD 4.46 billion
Growth rate
CAGR of 7.22% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, technology, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Mexico; Argentina; Saudi Arabia; Kuwait; UAE; South Africa
Key companies profiled
Charles River Laboratories; bioMérieux; Associates of Cape Cod, Inc.; FUJIFILM Wako Pure Chemical Corporation; Bioendo Technology; GenScript; ACROBiosystems; Thermo Fisher Scientific, Inc.; Merck KGaA; Sartorius AG; Eurofins Scientific; WuXi AppTec.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Pyrogen And Endotoxin Testing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global pyrogen and endotoxin testing market report based on product, technology, application, end-use, and region.
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Consumables
-
Limulus Amebocyte Lysate (LAL) Endotoxin Test Kits
-
Chromogenic & Turbidimetric Reagents
-
Recombinant Factor C (rFC) Reagents
-
Monocyte Activation Test (MAT) Kits and Reagents
-
Control standard endotoxins and buffers
-
Endotoxin-free Lab Consumables
-
-
Instruments
-
Endotoxin Testing Systems
-
Microplate and Kinetic Readers
-
Automated Endotoxin Detection Systems
-
Incubators and Sample Preparation Units
-
-
Services
-
Contract Endotoxin and Pyrogen and Endotoxin Testing for IVD Manufacturers
-
Method Validation and Compliance Testing
-
Laboratory Contamination and Quality Assurance testing
-
-
-
Technology Outlook (Revenue, USD Billion, 2021 - 2033)
-
Bacterial Endotoxin Testing (BET)
-
Gel Clot Method
-
Chromogenic Method
-
Turbidimetric Method
-
-
Recombinant Endotoxin Detection
-
Pyrogen Detection
-
Monocyte Activation Test (MAT)
-
Rabbit Pyrogen Test
-
-
-
Application Outlook (Revenue, USD Billion, 2021 - 2033)
-
Product Safety & Batch Release Testing
-
Manufacturing Process & Raw Material Control
-
Validation & Regulatory Compliance Testing
-
Laboratory & Clinical Environment Contamination Control
-
-
End-use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Manufacturers
-
Biotechnology & Life Sciences Companies
-
Contract Research & Manufacturing Organizations (CROs & CDMOs)
-
Clinical & Diagnostic Laboratories
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global pyrogen and endotoxin testing market size was estimated at USD 2.56 billion in 2025 and is expected to reach USD 2.74 billion in 2026.
b. The global pyrogen and endotoxin testing market is expected to grow at a compound annual growth rate of 7.22% from 2026 to 2033 to reach USD 4.46 billion by 2033.
b. North America dominated the pyrogen and endotoxin testing market with a share of 36.78% in 2025. This is attributable to rising number of chronic diseases needing developments in drug therapy. The growing adoption of ethical and efficient in vitro pyrogen tests has become a cornerstone of this dominance.
b. Some key players operating in the pyrogen and endotoxin testing market include Charles River Laboratories, bioMérieux, Associates of Cape Cod, Inc., FUJIFILM Wako Pure Chemical Corporation, Bioendo Technology, GenScript, ACROBiosystems, Thermo Fisher Scientific, Inc., Merck KGaA, Sartorius AG, Eurofins Scientific, and WuXi AppTec.
b. Key factors that are driving the market growth include increasing demand for pyrogen and endotoxin testing products in the pharmaceutical and biotechnology industries, rapid expansion of these industries, coupled with the rising number of new therapeutic launches.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









