- Home
- »
- Medical Devices
- »
-
Pharmaceutical CRO Market Size, Industry Report, 2033GVR Report cover
![Pharmaceutical CRO Market Size, Share & Trends Report]()
Pharmaceutical CRO Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Drug Discovery, Pre-Clinical, Clinical), By Molecule Type (Small Molecules, Large Molecules), By Services, By Therapeutic Area, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-470-0
- Number of Report Pages: 230
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Pharmaceutical CRO Market Summary
The global pharmaceutical CRO market size was estimated at USD 45.33 billion in 2025 and is projected to reach USD 83.31 billion by 2033, growing at a CAGR of 8.04% from 2026 to 2033. The industry is driven by the increasing rate of clinical research to boost demand for outsourcing services, the surge in the demand for personalized medicine and advanced therapeutics, rising mergers and collaboration activities in the contract research organization (CRO) market, growing demand for outsourcing services across developing economies and rising focus of life sciences companies on their core competencies.
Key Market Trends & Insights
- Asia Pacific pharmaceutical CRO market held the largest share of 40.45% of the global market in 2025.
- China pharmaceutical contract research organization market is witnessing new growth opportunities.
- Based on type, the clinical segment held the largest market share of 75.34% in 2025.
- Based on molecule type, the small molecules segment held the highest market share in 2025.
- Based on service, the clinical monitoring segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 45.33 Billion
- 2033 Projected Market Size: USD 83.31 Billion
- CAGR (2026-2033): 8.04%
- Asia Pacific: Largest market in 2025
Rising clinical research activity is driving global demand for pharmaceutical CRO outsourcing as drug development becomes more complex and costly. With over 569,000 registered clinical studies worldwide, pharmaceutical companies increasingly rely on CROs for trial management, regulatory compliance, and operational efficiency. Besides this, growing investment in biologics, personalized medicine, and multicenter global trials further expands outsourcing needs, particularly in cost-efficient regions such as Asia Pacific and Latin America. CROs support sponsors through specialized expertise, advanced technologies, and regulatory guidance. In addition, digital innovation, AI-driven analytics, and decentralized clinical trial models are transforming research execution, improving patient recruitment, accelerating timelines, and strengthening the strategic role of CROs globally.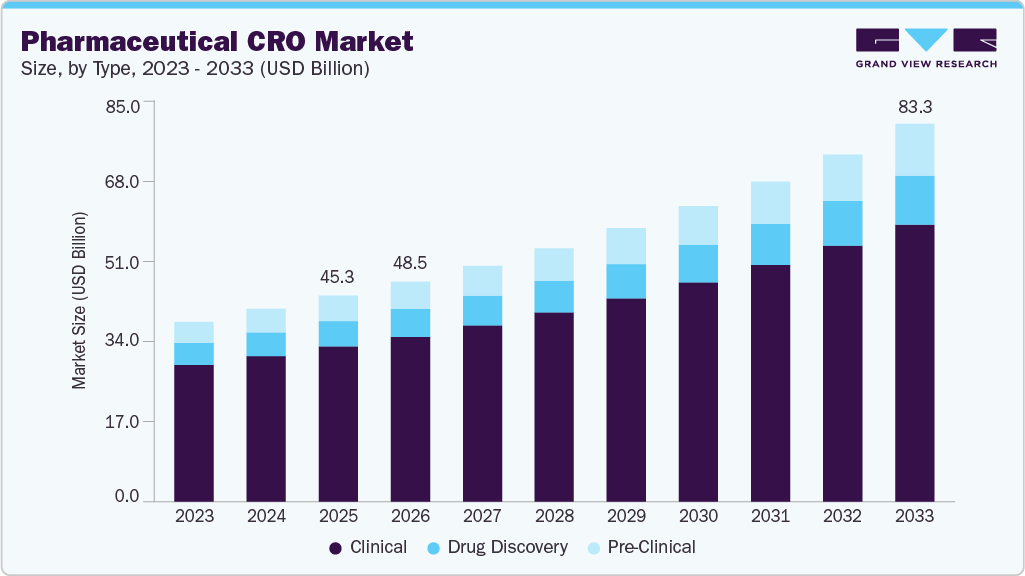
In addition, personalized medicine and advanced therapeutics that require complex research to evaluate safety, efficacy, and mechanisms of action are increasing reliance on pharmaceutical CROs for specialized preclinical and clinical studies. Besides this, the rising global incidence of cancer, neurodegenerative diseases, and genetic disorders, and demand for gene therapies are accelerating innovation and outsourcing needs. CROs are adopting advanced technologies, including AI-driven platforms, to improve trial design, patient recruitment, and operational efficiency. Similarly, growing approvals of novel therapies and increasing biotech innovation propel research activity, creating significant opportunities for CRO market expansion and technology-driven clinical development worldwide.
Furthermore, the pharmaceutical CRO industry is experiencing increased mergers, acquisitions, and collaborations to expand service capabilities, geographic reach, and technological expertise. Besides this, growing outsourcing of drug development has driven CRO consolidation, enabling integrated services across the clinical lifecycle. Some of the emerging economies such as India, China, Brazil, and South Africa attract outsourcing due to lower costs, diverse patient populations, modern infrastructure, and supportive regulatory reforms. In addition, faster patient recruitment and harmonized regulatory standards further accelerate clinical research and adoption of advanced technologies, including AI and data analytics, enhances trial efficiency and quality.
Moreover, outsourcing in the pharmaceutical and biotechnology sectors is primarily driven by the need to avoid large capital investments and improve operational efficiency. By delegating non-core functions to CROs, companies can focus on core activities such as drug discovery, development, and commercialization. In addition, rising R&D and preclinical testing activities are outsourced to emerging markets in Asia Pacific and the Middle East & Africa to reduce costs and access specialized capabilities. This trend is particularly prominent among small- and mid-sized life sciences companies lacking extensive infrastructure. Thus, outsourcing enables resource optimization, streamlined operations, and greater focus on advancing clinical development programs efficiently.
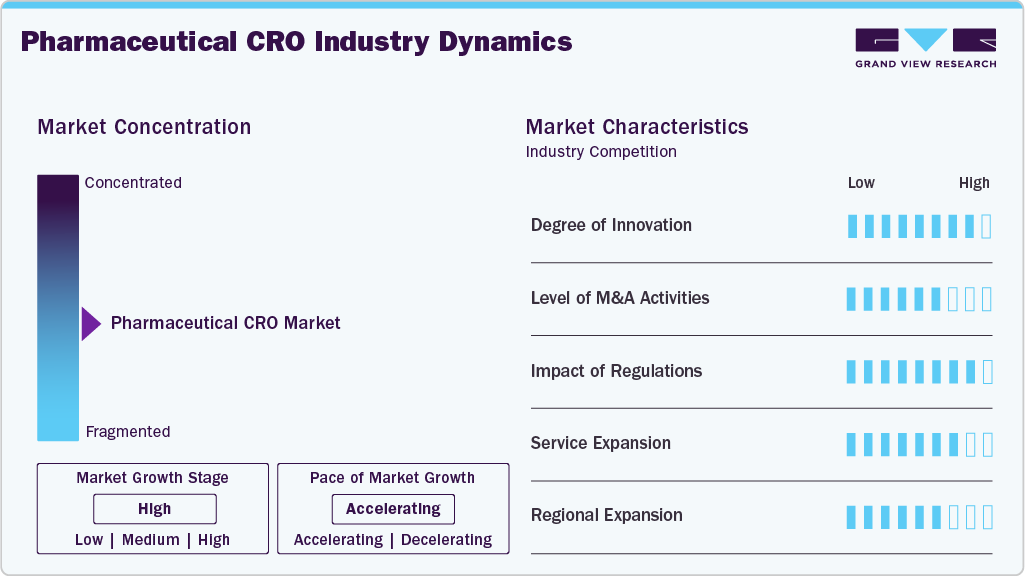
Market Concentration & Characteristics
The market growth stage is high, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion
CROs are rapidly integrating AI, predictive analytics, cloud-based data platforms, and real-world evidence capabilities to optimize protocol design, patient recruitment, and trial execution. In addition, technology-driven platforms such as the TriNetX-enable feasibility and recruitment capabilities adopted by ICON plc enhance enrollment efficiency, reduce timelines, and improve study outcomes.
Strict oversight from agencies such as the U.S. Food and Drug Administration and European Medicines Agency drives demand for CROs with strong compliance frameworks and inspection-ready operations. Besides this, sponsors retain regulatory accountability for outsourced work, increasing reliance on CROs with proven audit performance and global regulatory expertise.
Strategic alliances, platform integrations, and acquisitions remain central to capability expansion, geographic reach, and service differentiation. Partnerships with technology providers, healthcare networks, and biotech sponsors enable CROs to deliver end-to-end solutions across discovery, development, and commercialization. For instance, in January 2026, Worldwide Clinical Trials agreed to acquire Catalyst Clinical Research, strengthening oncology expertise, expanding global site reach, and intensifying consolidation trends across the pharmaceutical CRO market.
Some of the leading CROs provide integrated services spanning feasibility assessment, protocol optimization, site management, data analytics, pharmacovigilance, and multi-country trial coordination. The expansion into real-world data, decentralized trials, and advanced analytics supports complex therapeutic research, including oncology and gene therapy.
CROs are expanding global delivery networks across Asia-Pacific, Latin America, and emerging regions to access diverse patient populations and cost efficiencies. The hybrid and decentralized trial models, supported by digital infrastructure and regional operational hubs, enable faster recruitment and broader geographic coverage.
Opportunity Analysis
The global expansion of clinical trial activity is significantly increasing demand for CRO services. ClinicalTrials.gov reports over 569,000 registered studies across more than 225 countries, reflecting the growing globalization of drug development. Besides this, regulatory data indicate that clinical and manufacturing oversight is conducted outside the U.S. in a large number, with a majority of FDA inspections occurring at foreign sites and a large proportion of regulated facilities located internationally. These trends highlight the shift toward globally distributed development models and growing reliance on CROs for regional execution, regulatory coordination, and compliance support.
In addition, the FDA’s Bioresearch Monitoring program reinforces that sponsors retain responsibility for outsourced activities, increasing demand for CROs with strong compliance frameworks and inspection-ready quality systems. As clinical trials become more complex and geographically dispersed, pharmaceutical and biotechnology companies particularly small and mid-sized firms are increasingly outsourcing monitoring, data management, and regulatory operations, supporting sustained CRO market growth.
Impact of U.S. Tariffs on the Global Pharmaceutical Contract Research Organization (CRO) Market
U.S. tariffs on imported pharmaceuticals, laboratory equipment, and research inputs can indirectly influence the global CRO market by increasing operational and supply chain costs. Higher import duties on active pharmaceutical ingredients, biologics materials, and specialized instruments may raise clinical development expenses for sponsors, encouraging cost optimization through outsourcing to CROs in lower-cost regions. Similarly, tariffs can disrupt cross-border supply chains, prompting companies to diversify research locations and expand partnerships with international CROs. In addition, regulatory and trade uncertainties also encourage sponsors to rely on CROs with strong regional infrastructure and local compliance expertise. However, tariffs may increase costs for CROs dependent on imported equipment or reagents, potentially affecting pricing structures. Thus, while tariffs introduce short-term cost pressures and operational adjustments, they can strengthen outsourcing demand by driving geographic diversification, strategic collaboration, and greater reliance on globally distributed CRO service networks.
Technological Advancements
Technological advancements are transforming the pharmaceutical CRO market by enhancing efficiency, data quality, and patient-centric research. Some of the advancements include AI-driven trial design and patient recruitment, decentralized and hybrid clinical trial technologies, advanced data management and real-time analytics, automation and robotic process integration and advanced biomarker and precision medicine capabilities. Artificial intelligence and machine learning improve trial design, site selection, and patient recruitment through predictive analytics.

Besides this, decentralized and hybrid clinical trial technologies enable remote monitoring, telemedicine integration, and digital patient engagement, expanding participation and improving retention. In addition, advanced data management platforms and real-time analytics provide centralized oversight, support risk-based monitoring, and enhance regulatory compliance across global studies. Furthermore, automation technologies, including robotic process automation, streamline documentation, data processing, and regulatory workflows, reducing manual errors and accelerating timelines. In addition, advances in genomics, biomarker analysis, and precision medicine capabilities allow CROs to support targeted therapies and complex therapeutic areas such as oncology and rare diseases.
Pricing Model Analysis
In the pharmaceutical CRO market, pricing models are designed to align project risk, value delivery, and partnership duration. The milestone-based pricing links payments to predefined achievements such as study initiation, enrollment targets, or regulatory submissions, improving accountability and distributing financial risk. Whereas, value-based pricing reflects the strategic impact of CRO services, including faster timelines, improved recruitment outcomes, and regulatory success.
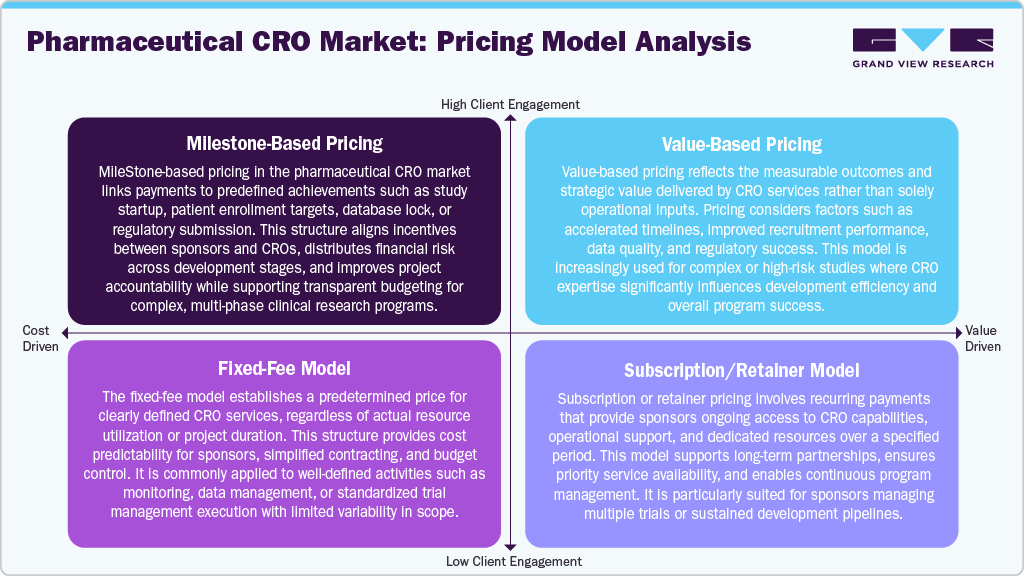
The fixed-fee model offers a predetermined cost for clearly defined services, providing budget predictability and simplified contracting for standardized activities. In addition, subscription or retainer models involve recurring payments that ensure ongoing access to CRO expertise, dedicated resources, and operational support. These models support long-term collaboration and continuous program management.
Type Insights
On the basis of type, the clinical segment held the largest market share of 75.34% in 2025. This segment includes phase I, II, III, and IV trial services. The segment is driven by the increasing volume of drug development programs. Pharmaceutical companies are outsourcing clinical trials to CROs to gain access to specialized expertise, global trial networks, and advanced technologies, while also managing costs and operational efficiency. Other factors driving this demand include the rapid expansion of drug pipelines, the globalization of clinical trials, and the need to navigate complex regulatory requirements across multiple markets. In addition, the increasing number of small and mid-sized biotech companies, the adoption of decentralized and digital trial solutions, and the trend toward long-term strategic partnerships with CROs have further boosted demand for the pharmaceutical CRO market.
The preclinical segment is driven by scientific, regulatory, and economic factors, as these studies generate critical safety, efficacy, and pharmacokinetic data before human trials. Some of the pharmaceutical companies outsource in vitro, in vivo, and toxicology studies to CROs due to specialized expertise and infrastructure requirements. In addition, technological advancements, including high-throughput screening, organ-on-chip models, genetically modified animals, and AI-based predictive tools, enhance study accuracy and efficiency.
Molecule Type Insights
On the basis of the molecule type, the small molecule segment held the largest share in 2025. Small-molecule therapies continue to drive drug development and outsourcing across discovery and clinical stages. For instance, in 2025, the U.S. Food and Drug Administration approved 46 novel drugs, including 34 small-molecule treatments across oncology, metabolic, and infectious diseases. In addition, early-stage outsourcing is strengthening through external discovery platforms such as AION Labs, supported by AstraZeneca, Merck, Pfizer, and Teva, reflecting rising sponsor reliance on external ecosystems for accelerated lead identification and optimization.
The large molecules are the fastest-growing segment in the market. Large-molecule drug development, including monoclonal antibodies, recombinant proteins, vaccines, and cell and gene therapies, continues to drive demand for specialized outsourced research and clinical services. These modalities involve complex bioanalytical workflows, immunogenicity assessment, and customized clinical trial designs, expanding the scope of CRO engagement across preclinical evaluation and late-stage development. The technical complexity of biologic programs increases the need for comparative studies, bioequivalence assessment, and immunogenicity testing, supporting sustained demand for CRO services across complex biologic development pathways.
Service Insights
On the basis of service, the clinical monitoring segment held the largest revenue share in 2025. Clinical monitoring is crucial for ensuring the integrity and compliance of clinical trials. It oversees the clinical trial conduct, verifies data accuracy, and ensures adherence to regulatory standards, making it an important component of the drug development process. For instance, in January 2024, ICON plc announced offering clinical trial services to BioNTech and Pfizer investigational COVID-19 vaccine programme. The company has implemented extensive remote clinical monitoring and source data verification which has highlighted the growing reliance on technology in clinical trials.
On the other hand, the regulatory and medical affairs services segment is the fastest-growing segment over the forecast period, as global trials face increasingly complex requirements. CROs help sponsors navigate guidance from the U.S. Food and Drug Administration and European Medicines Agency, manage post-marketing safety, and align scientific communication with compliance expectations. In addition, growing regulatory complexity is driving outsourcing demand. CROs are investing in advanced regulatory technologies and strategic partnerships to enhance submission efficiency and regulatory readiness. For instance, in October 2025, Parexel International Corporation partnered with Weave Bio to integrate AI-powered automation into regulatory authoring and submission preparation, accelerating approvals and improving regulatory readiness.
Therapeutic Area Insights
On the basis of therapeutic area, the oncology segment held the largest revenue share in 2025, owing to the rising prevalence of cancer, including lung cancer, breast cancer, colorectal cancer, and prostate cancer. For instance, according to The Cancer Atlas, the number of cancer cases is expected to reach 29 million globally by 2040. Moreover, the growing patient population is expected to encourage pharmaceutical and biotechnology companies to expand their oncology drug pipelines, thereby increasing the outsourcing of clinical research activities to CROs. In addition, oncology drug development involves complex clinical trial designs, biomarker-based studies, and targeted therapies that require specialized expertise and advanced research infrastructure. CROs help companies identify suitable patients, manage regulatory requirements, and handle clinical trial data, making oncology trials faster and more efficient.
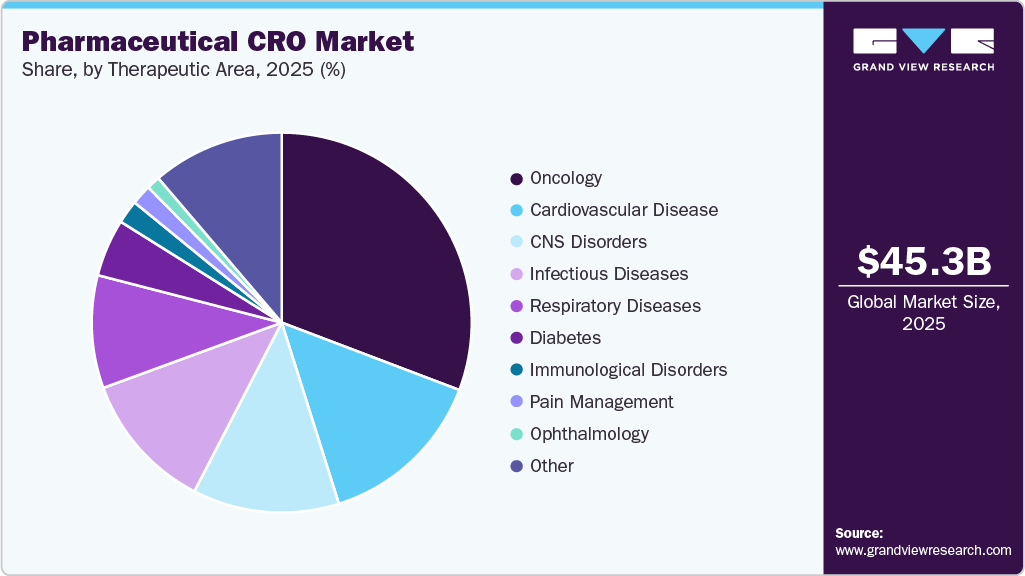
The CNS disorders segment is amongst the fastest growing segments, due to the rising prevalence of neurological disorders such as Alzheimer’s, Parkinson’s, epilepsy, and multiple sclerosis, which is accelerating CNS drug development. According to the NCD Alliance, about 3.4 billion people live with neurological conditions worldwide, expanding the patient pool and investment in research. Besides this, due to complex trial designs, long durations, and specialized assessments, sponsors rely on CROs for execution and compliance. In addition, CNS drug development has shown high failure rates, which has created an increase in the demand for experienced research partners and advanced clinical trial design strategies. For instance, according to Cytel, nearly 85% of Phase II-III studies fail due to disease complexity, subjective assessments, and high placebo response rates. These challenges make CNS clinical trials more complicated and time-consuming to conduct.
Regional Insights
North America pharmaceutical contract research organization market held a significant share in 2025, supported by a strong pharmaceutical and biotechnology sector, advanced research infrastructure, and high healthcare spending. U.S. leads globally in healthcare expenditure, driving demand for outsourced research services. CROs are evolving into strategic partners that manage drug development across preclinical, clinical, and post-approval stages, reflecting growing complexity in clinical trials. The rise of biologics, cell and gene therapies, and precision medicine has increased development costs and timelines, encouraging companies to outsource research activities. In addition, strategic collaborations, service expansion, and technological adoption by CROs continue to support market growth across the region.
U.S. Pharmaceutical CRO Market Trends
The pharmaceutical contract research organization market in the U.S. accounted for the highest market share in the North America market, owing to increasing R&D costs and the growing trend of outsourcing clinical trials. In addition, large pharmaceutical companies and smaller biotech firms are relying on CROs to conduct preclinical studies, clinical trials, and regulatory support, enabling faster and more cost-effective drug development. The market is dominated by a few major players, including IQVIA, Labcorp, ICON plc, Parexel International Corporation, and Medpace, which compete on the basis of service range, therapeutic expertise, technological capabilities, and global reach. Besides full-service CROs, mid-sized and specialized companies offer niche services in specific therapeutic areas or advanced technologies such as AI-driven trial design and decentralized clinical trials.
Canada pharmaceutical contract research organization market is expected to grow at a significant CAGR during the forecast period. The pharmaceutical industry has outsourced drug R&D programs and manufacturing through collaborations with academic institutions, small and medium-sized enterprises (SMEs), government research centers, and specialized research and manufacturing organizations. In Canada, CROs have become critical in this ecosystem, providing comprehensive services that accelerate product development and enhance manufacturing efficiency.
Europe Pharmaceutical CRO Market Trends
The pharmaceutical contract research organization market in Europe is evolving due to the presence of reputed pharmaceutical companies, numerous drug discoveries, and increased demand for CROs. In addition, many CROs specializing in drug discovery have their centers in European countries like the UK, Germany, and France, which is expected to contribute to market growth in this region. Moreover, the growing trend of outsourcing services to European countries is attributed to the increasing demand for pharmaceutical CRO across the region. The regional market is expected to be driven by a large patient population across Eastern & Western Europe and a centralized submission process for clinical trials. In addition, rising demand for new drugs, the expansion of new facilities, and emerging R&D activities are changing market scenarios rapidly.
Germanypharmaceutical contract research organization market held the highest share in 2025. The country has a growing number of CRO service providers that support leading biopharmaceutical companies. Outsourcing research and development services helps pharmaceutical companies in reducing infrastructure and manufacturing costs and improving operational efficiency, increasing the adoption of CRO services in the country. Moreover, the pharmaceutical CRO market in Germany is witnessing consolidation, as larger organizations acquire or merge with smaller service providers to expand capabilities and strengthen market presence.
The pharmaceutical contract research organization market in the UK is fueled by the presence of several academic and research institutes, which are constantly working towards research and development activities by entering into the partnerships with pharmaceutical companies. Moreover, the government’s support for biotechnology and life sciences through funding initiatives and favorable regulatory frameworks is attracting both domestic and international CROs.
Asia Pacific Pharmaceutical CRO Market Trends
The pharmaceutical contract research organization market in Asia Pacific held the largest share of 40.45% in 2025 and is amongst the fastest growing region over the forecast period, driven by cost advantages, a large patient pool, and growing adoption of global R&D and regulatory standards. The region benefits from expanding outsourcing hubs, skilled workforce availability, and increasing competition among service providers. Besides this, rising collaborations between global and local organizations support broader therapeutic coverage and faster development timelines. Some of the countries such as China, Japan, and India are emerging as key destinations due to regulatory reforms, advanced healthcare infrastructure, and strong clinical capabilities.
China pharmaceutical contract research organization market is witnessing new growth opportunities due to growing outsourcing among pharmaceutical companies, stringent standards around patient safety in clinical trials, easy access to a large pool of patients, and low operating costs. In addition, rising investments in the healthcare sector to upgrade infrastructure and boost advancements in technologies, along with the rising adoption of global R&D standards to meet international requirements, are key factors responsible for the growing demand for outsourcing services in China.
The pharmaceutical contract research organization market in Japan is characterized by increasing pipeline drug candidates, cost-effectiveness, and a proactive approach to competitive manufacturing techniques. In addition, the country is considered a significant location for clinical trials due to its well-established clinical development knowledge, clinical investigational sites with well-trained site staff & regulatory processes. In addition, increased competition in the marketplace is compelling pharmaceutical companies to outsource drug production to CROs, resulting in enhanced process oversight, accelerated manufacturing timelines, and superior quality assurance in contrast to conventional batch manufacturing techniques, all of which support new product development.
India pharmaceutical contract research organization market is experiencing rapid expansion, attributed to the rising cost of drug discovery that has led to pressure on market players and innovators to recover revenue lost to generics, increasing patent expiration, rising collaborations to find biologics, and surging R&D costs. Besides, the country has become one of the preferred sites for clinical trials due to its developing and cost-effective healthcare sector. Growing investment in healthcare, an increasing number of established CROs, skilled professionals, and logistical advantages are contributing to the growth of the Indian pharmaceutical CRO market.
Latin America Pharmaceutical CRO Market Trends
The pharmaceutical contract research organization market in the Latin America region is driven by the rising number of clinical trials conducted across the region, growing healthcare needs, and expanding pharmaceutical research programs, which are encouraging sponsors to include Latin American countries in global clinical development plans. The region offers lower trial execution costs and access to large patient populations, which support faster patient enrollment and improve study timelines. These factors are increasing the use of CROs for trial setup, site management, patient recruitment, and data handling across multiple countries in the region.
Brazil pharmaceutical contract research organization market is expanding as pharmaceutical companies running clinical trials rely on CROs to manage local study execution. These organizations support key functions such as site coordination, patient recruitment, monitoring, and data management across multi-center trials conducted at leading hospitals and research institutions. CRO effectiveness in Brazil largely depends on strong local operational teams and the capability to oversee studies across geographically dispersed sites. In addition, providers with established in-country infrastructure and long-term collaborations with hospitals and investigators are better positioned to secure complex and large-scale clinical research programs.
Middle East and AfricaPharmaceutical CRO Market Trends
The pharmaceutical contract research organization market in the Middle East & Africa (MEA) is witnessing significant growth, driven by higher healthcare spending and the development of hospital research units, trial sites, and laboratory capacity across key markets. Public and private investment is improving site readiness, investigator availability, and operational support functions, enabling sponsors to place a higher volume of industry-sponsored studies within the region. This expansion is improving on-ground execution across patient recruitment, site coordination, and clinical data collection for pharmaceutical development programs.
South Africa pharmaceutical contract research organization market is expanding due to established providers supporting industry-sponsored clinical trials across high-burden therapeutic areas. Local firms deliver services including feasibility assessment, monitoring, recruitment coordination, and project management for Phase I-IV studies through hospital and investigator networks. In addition, competitive advantage depends on strong investigator relationships, access to trial-ready sites, proven execution capability, regulatory familiarity, and reliable operational risk management across diverse clinical settings.
The pharmaceutical contract research organization market in the UAE is experiencing growth, fueled by access to clinical research infrastructure in Dubai and Abu Dhabi and the ability to execute sponsor-led pharmaceutical trials within national approval timelines. CRO selection by pharmaceutical sponsors is driven by investigator coverage, speed of site activation, and delivery performance in multi-center studies. For instance, in 2025, IROS, an Abu Dhabi-based contract research organization under the M42 group, partnered with BioSapien to conduct an oncology clinical trial in Abu Dhabi, illustrating active CRO participation in sponsor-led pharmaceutical development programs in the country.
Key Pharmaceutical CRO Companies Insights
The mature CROs compete through scale, global networks, and integrated service platforms, while emerging players differentiate via specialization, agility, and customized solutions. Ongoing investments in analytics, automation, and global site networks are strengthening competitive positioning and supporting sustained market growth. For instance, in January 2026, IQVIA partnered with Boehringer Ingelheim to deploy its DaaS+ platform, accelerating commercial data harmonization and reinforcing CRO roles in post-trial analytics and lifecycle commercialization support.
Key Pharmaceutical CRO Companies:
The following key companies have been profiled for this study on the pharmaceutical contract research organization market.
- Parexel International (MA) Corporation.
- ICON plc
- Laboratory Corporation of America Holdings
- Thermo Fisher Scientific Inc.
- Medpace
- IQVIA
- CTI Clinical Trial & Consulting
- WuXi AppTec
- Veeda Clinical Research
Recent Developments
-
In January 2026, PSI launched SYNETIC, an AI-powered platform improving site identification, startup speed, and enrollment predictability, reinforcing technology-driven efficiency and competitiveness across the pharmaceutical CRO market.
-
In November 2025, Adicon agreed to acquire Crown Bioscience, creating a global end-to-end laboratory services platform and intensifying cross-border consolidation, expanding integrated discovery-to-diagnostics capabilities within the pharmaceutical CRO market.
-
In November 2025, Ergomed opened offices in Buenos Aires and expanded into Brazil and Colombia, improving regional access, regulatory agility, and patient diversity for global CRO studies.
Pharmaceutical CRO Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 48.47 billion
Revenue forecast in 2033
USD 83.31 billion
Growth rate
CAGR of 8.04% from 2026 to 2033
Historical year
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, molecule type, service, therapeutic area and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Parexel International (MA) Corporation.; ICON plc; Laboratory Corporation of America Holdings; Thermo Fisher Scientific Inc.; Medpace; IQVIA; CTI Clinical Trial & Consulting; WuXi AppTec; Veeda Clinical Research
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Pharmaceutical CRO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global pharmaceutical contract research organization market report based on type, molecule type, service, therapeutic area, and region.
-
Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Drug Discovery
-
Target Validation
-
Lead Identification
-
Lead Optimization
-
-
Pre-Clinical
-
Clinical
-
Phase I Trial Services
-
Phase II Trial Services
-
Phase III Trial Services
-
Phase IV Trial Services
-
-
-
Molecule Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Small Molecules
-
Large Molecules
-
-
Service Outlook (Revenue, USD Billion, 2021 - 2033)
-
Project Management/Clinical Supply Management
-
Data Management
-
Regulatory/Medical Affairs
-
Medical Writing
-
Clinical Monitoring
-
Quality Management/ Assurance
-
Biostatistics
-
Investigator Payments
-
Laboratory
-
Patient And Site Recruitment
-
Technology
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Billion, 2021 - 2033)
-
Oncology
-
CNS Disorders
-
Infectious Diseases
-
Immunological Disorders
-
Cardiovascular Disease
-
Respiratory Diseases
-
Diabetes
-
Ophthalmology
-
Pain Management
-
Other
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global pharmaceutical contract research organization market size was valued at USD 45.33 billion in 2025 and is projected to reach USD 48.47 billion by 2026.
b. The global pharmaceutical contract research organization is projected to reach USD 83.31 billion by 2033, growing at a CAGR of 8.04% from 2026 to 2033.
b. Based on type, the clinical segment dominated the pharmaceutical CRO market accounted for the largest revenue share of 75.34% in 2025.
b. Some key players operating in the pharmaceutical CRO market include Parexel International (MA) Corporation.; ICON plc; Laboratory Corporation of America Holdings; Thermo Fisher Scientific Inc.; Medpace; IQVIA; CTI Clinical Trial & Consulting; WuXi AppTec; Veeda Clinical Research.
b. Key factors that are driving the pharmaceutical contract research organization market growth include increasing rate of clinical research to boost demand for outsourcing services, surge in the demand for personalized medicine and advanced therapeutics, rising mergers and collaboration activities in the CRO market, growing demand for outsourcing services across developing economies and rising focus of life sciences companies on their core competencies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









