- Home
- »
- Pharmaceuticals
- »
-
Pharmaceutical Contract Manufacturing Market Report, 2033GVR Report cover
![Pharmaceutical Contract Manufacturing Market Size, Share & Trends Report]()
Pharmaceutical Contract Manufacturing Market (2026 - 2033) Size, Share & Trends Analysis Report By Service, By Product (Small Molecule, Biologics), By Drug, By Scale Of Operation, By Workflow, By Application, By End Use, By Company, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-520-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Pharmaceutical Contract Manufacturing Market Summary
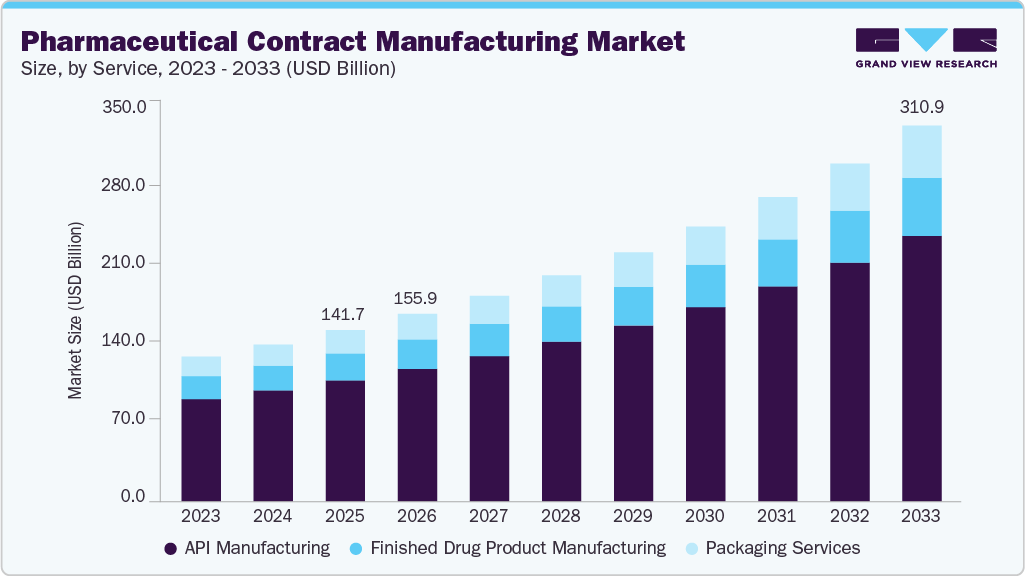
The global pharmaceutical contract manufacturing market size was estimated at USD 141.74 billion in 2025 and is projected to reach USD 310.97 billion by 2033, growing at a CAGR of 10.45% from 2026 to 2033. The demand for contract manufacturing is gaining momentum due to a surge in clinical as well as commercial workflow realignment among pharmaceutical manufacturers.
Key Market Trends & Insights
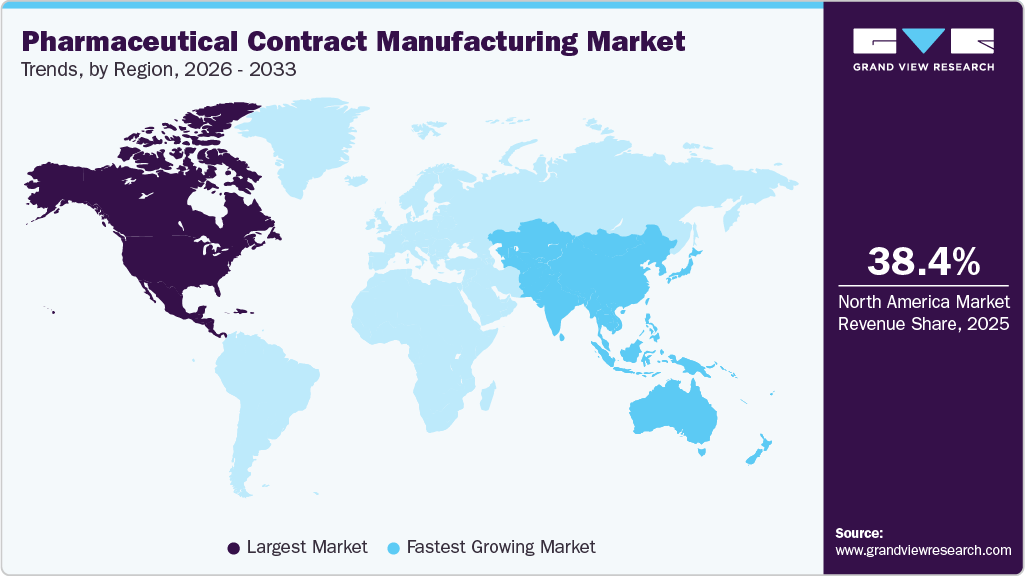
- North America pharmaceutical contract manufacturing industry held the largest share of 38.44% of the global market in 2025.
- The pharmaceutical contract manufacturing in the U.S. is expected to grow significantly over the forecast period.
- By service, the API manufacturing segment held the largest market share of 70.68% in 2025.
- By product, the small molecule segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 141.74 Billion
- 2033 Projected Market Size: USD 310.97 Billion
- CAGR (2026-2033): 10.45%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The pharmaceutical industry is witnessing a shift toward assigning complex manufacturing processes to specialized partners like contract manufacturing organizations (CMOs), reinforcing the growth of the market.
Furthermore, most small-to-mid-sized pharmaceutical companies benefit from CMOs, which support them focus on their core competencies and tackle the complexities of production for a company contracting them. In addition, most contract manufacturing providers specialize in the availability of various sterile filling equipment that supports increased production, further enhancing the process of drug production without compromising quality. Besides, the surge in drug discovery techniques and growing focus on quality control systems to ensure that products meet industry standards drive the market growth.
Moreover, rapid technological advances are also reshaping contract manufacturing to improve efficiency while enabling effective drug customization. Technological advancements such as advanced manufacturing technologies, data analytics and artificial intelligence (AI) and quality by design (QbD) and process analytical technology (PAT) support continuous evolution and demand for new manufacturing capacities to accelerate drug development timelines. For instance, data analytics and AI are transforming various aspects of the pharmaceutical industry by harnessing major amounts of data generated throughout the manufacturing process, it can further support optimizing production, predict equipment failures, and ensure regulatory compliance. Moreover, CMOs with expertise in formulation development and clinical trial production are expected to boost the AI-driven discoveries into real-world treatments. Moreover, growing requirement for branded and generic drugs is one of the significant factors for the growth of the market.
Thus, the role of contract manufacturing (CMOs) in the pharmaceutical industry offers a comprehensive range of tailored solutions from pharmaceutical development to commercial services, further accelerating drug development, minimizing the cost, and improving efficiency.
Market Concentration & Characteristics
The pharmaceutical contract manufacturing industry growth stage is medium, and growth is accelerating. The market is characterized by the level of merger & acquisitions activities, degree of innovation, regulatory impact, product expansion, and regional expansions.
Pharmaceutical contract manufacturing is adopting innovation through continuous manufacturing, AI-driven process optimization, single-use technologies, and advanced biologics production. These advancements improve efficiency, enhance quality control, and allow for better scalability, positioning CMOs as essential partners in the evolving global pharmaceutical landscape.

Stringent global regulatory standards and the need to comply with agencies FDA, EMA, and ICH regulations drive quality, compliance, and transparency in pharmaceutical contract manufacturing. GMP requirements, data integrity standards, and evolving regulatory frameworks impact CMOs operations, ensuring safe, high-quality drug production while increasing costs, complexity, and supply chain scrutiny.
Mergers and acquisitions (M&A) in pharmaceutical contract manufacturing are surging, driven by technological advancements, and expanding service portfolios. In addition, the CMOs are acquiring specialized firms to enhance biologics, HPAPI, and sterile manufacturing capabilities, strengthening global supply chains and competitiveness.
The market for pharmaceutical contract manufacturing remains fragmented, with numerous CMOs catering to small molecules, biologics, and specialty drugs. Besides, cost advantages, and regulatory compliance have led the market competitive fragmentation.
Pharmaceutical contract manufacturing is witnessing rapid regional expansion, with CMOs investing in developed and developing economics to leverage cost efficiencies and favorable regulations, which further fuel market growth.
Service Insights
Based on service segment, the market is segmented into API Manufacturing, finished drug product manufacturing, and packaging services. Finished drug product manufacturing further includes oral solids, liquids, topical, and others. The API manufacturing segment captured the largest market share of 70.68% in 2025. This significant growth can be attributed to rising outsourcing trends, cost efficiency, and the need for regulatory compliance. Producing active pharmaceutical ingredients (APIs) is one of the most critical processes in pharmaceutical production, as it helps create lifesaving medications for patients.
The packaging services segment is expected to grow at the fastest CAGR over the forecast period. The growth is due to increasing demand for lifesaving pharmaceuticals and the rising partnerships among pharmaceutical companies and contract manufacturing organizations (CMOs) to provide a range of services, including manufacturing and packaging. In addition, the need for cost-effective packaging solutions and the requirement for packaging of lifesaving medications are crucial in ensuring that these products reach patients effectively.
Innovations in the primary packaging industry are also becoming more prevalent, particularly for small-batch production. Furthermore, pharmaceutical contract manufacturing has emerged significantly, leading to increased production capacity and reduced time-to-market for new product launches. This segment offers various packaging options, including blister packaging, bottle and vial packaging, ampoule and capsule packaging, tubes, sachets, pouches, and stick packs. These solutions help pharmaceutical companies enhance efficiency and maintain product integrity throughout the supply chain, from primary packaging and labeling to shipping, storage, and distribution to end users. This trend is expected to drive market growth over the estimated time.
Product Insights
Based on product segment, the market is segmented into small molecules and biologics. Biologics further includes monoclonal antibodies, vaccines, recombinant proteins, and others. The small molecules segment dominated the global market in 2025. Small-molecule drugs have been the backbone of the pharmaceutical industry for nearly a century. The segment growth is driven by the increasing volume of small molecule production; growing advantages CMOs offer will simplify manufacturing compared to large molecule biologics.
The biologics segment is expected to grow at significant CAGR over the forecast period. This growth is attributed to high investments and ongoing innovation in therapeutic & other medications. In addition, the increasing adoption of advanced technologies such as artificial intelligence, the rising outsourcing trends, and the growing research and development (R&D) activities by pharmaceutical companies are key factors propelling the segment's growth.
Drug Insights
On the basis of drug segment, the market is segmented into generics and branded. The generics segment accounted for the largest market share in 2025. The segment growth is driven by rising patent expirations, increased outsourcing by pharmaceutical companies, and rising requirements for cost-effective medicines. Besides, continuous manufacturing trends and technological advancement have led the segment to grow. Furthermore, the low manufacturing costs have encouraged various pharmaceutical companies in developed economies to outsource their manufacturing activities to CMOs.
The branded segment is expected to grow at a considerable CAGR during the forecast period. The branded drug contract manufacturing market is experiencing steady growth as pharmaceutical companies increasingly outsource production to Contract Manufacturing Organizations (CMOs). Moreover, due to the rising complexity of drug formulations, most pharmaceutical manufacturers producing branded drugs are turning to contract manufacturing organizations for their expertise in advanced formulations, biologics, sterile injectables, and novel drug delivery systems.
Scale of Operation Insights
On the basis of the scale of the operation segment, the market is segmented into batch and continuous segments. The batch segment dominated the market in 2025. A key trend in the market is batch outsourcing, as it offers smaller companies the opportunity to save operational costs and significantly reduces the preparation and adjustment of equipment before commencing the production of each batch. Such factors are anticipated to drive the segment growth.
The continuous segment is expected to experience significant growth at a specific (CAGR) during the forecast period. Pharmaceutical continuous manufacturing (CM) is considered a crucial technology in the pharmaceutical contract manufacturing process, with a growing emphasis on producing innovative drugs. The segment growth can be attributed to greater efficiency, cost reduction, and improved product quality.
Workflow Insights
On the basis of the workflow segment, the market is segmented into clinical and commercial. The commercial segment dominated the global market in 2025 owing to a strong pipeline for pharmaceutical drug development, and large-scale commercial manufacturing, among others. In addition, several pharmaceutical companies are turning to commercial contract manufacturing to access advanced infrastructure and expertise due to the increasing influx of novel therapies & orphan drugs and higher returns on approved products.
The clinical segment is anticipated registering a significant CAGR during the forecast period. The segment is driven by growing number of clinical trials and increasing demand for specialized CMO services. Such factors are anticipated to drive the market.
Application Insights
On the basis of the application segment, the market is segmented into oncology, infectious diseases, neurological disorders, cardiovascular diseases, metabolic disorders, autoimmune diseases, respiratory diseases, ophthalmology, gastrointestinal disorders, hormonal disorders, hematological disorders, and others.
The oncology segment dominated the global market and accounted for a largest revenue share in 2025. The segment is expected to grow due to the increasing number of cancer cases worldwide. For instance, the Cancer Atlas mentioned that there will be 29 million cancer cases across the globe by 2040. Recent trends in pharmaceutical products have shifted with growing R&D for cancer treatments.
The cardiovascular diseases segment is expected to experience significant growth at a specific (CAGR) during the forecast period. The segment is driven by the increasing prevalence of cardiovascular diseases, which has fueled the segment's growth. For instance, according to World Heart Report 2023, it revealed that over half a billion people globally are affected with cardiovascular diseases, resulting in nearly a million deaths. Hence, the growing innovation of pharmaceutical products is expected to propel market growth.
End Use Insights
On the basis of the end use segment, the market is segmented into pharmaceutical and biotechnology companies. The pharmaceutical companies segment dominated the market in 2025 owing to the increasing trend of outsourcing end-to-end services, especially among small and mid-sized pharmaceutical companies that lack expertise in drug development.
The biotechnology companies’ segment is expected to experience significant growth at a specific (CAGR) rate during the forecast period. Increasing preference for outsourcing among biotechnology, midsized biotechnology, and emerging companies to gain better knowledge of outsourcing pharmaceutical drugs has led the growth of market. Likewise, an increasing number of new pharmaceutical drugs contribute to market growth.
Company Size Insights
On the basis of the company size segment, the market is segmented into small, medium, and large. The small company size segment dominated the market with the largest revenue share in 2025. The high growth of the segment is mainly due to the presence of growing employee strengths, facilities and increasing venture funding.
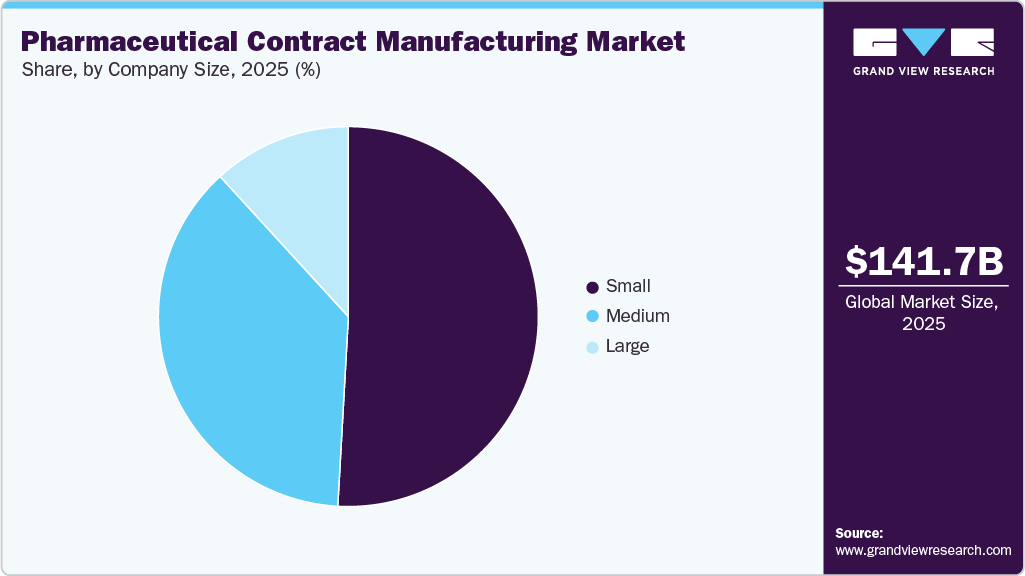
The medium segment is expected to grow at a significant CAGR during the forecast period. Most medium or “midsized” companies have 100 to 500 employees participating in the end-to-end development of innovative drugs; however, their concentration is limited to specific therapy areas.
Regional Insights
North America Pharmaceutical Contract Manufacturing Market Trends
The North American pharmaceutical contract manufacturing industry accounted for the largest share of 38.44% in 2025. This can be attributed to several factors, including technological advancements, growing demand for pharmaceutical products, and the increasing outsourcing of pharmaceutical production to third-party manufacturers.
U.S. Pharmaceutical Contract Manufacturing Market Trends
The pharmaceutical contract manufacturing industry in the U.S. held the largest share in 2025. This growth is driven by the expansion of the overall drug market and increasing trends in outsourcing. In addition, the development of generics has significantly benefited the market, as pharmaceutical manufacturers outsource as much as 80% of their production to contract manufacturing organizations (CMOs).
Furthermore, CMOs significantly enhance the existing pharmaceutical product portfolio of pharmaceutical companies. The market provides state-of-the-art facilities, quality control measures, and resilient supply chains for production. Hence growing need for innovative drugs, the rising prevalence of diseases, and the increasing presence of pharmaceutical companies have all led to a increased demand for CMOs services.
Europe Pharmaceutical Contract Manufacturing Market Trends
The pharmaceutical contract manufacturing industry in Europe is anticipated to experience significant growth due to stringent regulatory requirements and an increasing demand for biologics & specialty drugs. This trend has emphasized the need for innovation capabilities, which fuels the demand for pharmaceutical contract manufacturing services in the region.
Germany's pharmaceutical contract manufacturing industry held the largest share in 2025. This market is primarily driven by the expanding pharmaceutical industry and the increasing demand for contract manufacturing organization (CMO) services that support the production of life-saving medications for regional healthcare markets.
The UK's pharmaceutical contract manufacturing industry is anticipated to grow over the forecast period. This growth is primarily due to the growing need for contract manufacturing for efficient and increased pharmaceutical production to reduce the time to market, further fueling the market growth.
Asia Pacific Pharmaceutical Contract Manufacturing Market Trends
Asia Pacific pharmaceutical contract manufacturing industry is projected to register a significant CAGR during the forecast period. This growth can be attributed to various factors, such as growing pharmaceutical product requirements, the presence of well-established pharmaceutical companies, technology adoption, and several opportunities in this region that drive market growth.
China's pharmaceutical contract manufacturing industry held the largest share in 2025. The Chinese CMO market is driven by the rapidly expanding pharmaceutical drug industry and the advantages offered by contract manufacturing organizations (CMOs). The advantages include cost-effectiveness and advanced manufacturing capabilities to innovate new drugs.
Japan pharmaceutical contract manufacturing industry is the second-largest in Asia Pacific region, and it is expected to provide a robust opportunity for pharmaceutical contract manufacturing services thereby driving market growth. The country is experiencing an increasing aging population and a rising incidence of diseases, leading to a rising requirement for various pharmaceutical drugs and contract manufacturing services.
The pharmaceutical contract manufacturing industry in India is expected to experience significant growth at a significant CAGR during the forecast period. This growth is driven by an increasing focus on research and development (R&D) to support the pipeline of generics, biologics, biosimilars, and personalized medicines, along with a rising demand for cost-efficient production services.
Key Pharmaceutical Contract Manufacturing Company Insights
Market players are undertaking various strategic initiatives, such as the launch of new product innovations, collaborations, partnerships, and mergers & acquisitions, to strengthen their service offering and provide a competitive advantage. For instance, in March 2024, Tonix Pharmaceuticals Holding Corp. mentioned that it has selected two CMOs including Almac Pharma Services, to potentially produce Tonmya for commercial use in the U.S. The company is excited to move its first internally developed program toward NDA submission & to collaborate with CMOs for commercial supply and potential launch of Tonmya.
Key Pharmaceutical Contract Manufacturing Companies:
The following key companies have been profiled for this study on the pharmaceutical contract manufacturing market.
- AbbVie Contract Manufacturing
- Almac Group
- Boehringer Ingelheim BioXcellence
- Catalent
- Dr. Reddy's Laboratories
- Fareva
- Fujifilm Diosynth Biotechnologies
- Jubilant HollisterStier
- Lonza Group
- Patheon (Thermo Fisher Scientific)
- Pfizer CentreOne
- Prakruti Life Science
- Recipharm
- Samsung Biologics
- Siegfried Holding AG
- Vetter Pharma
- WuXi AppTec
Recent Developments
-
In October 2024, Samsung Biologics mentioned a contract manufacturing partnership with an Asian pharmaceutical company. This partnership is valued at USD 1.24 billion, making it one of the most significant contracts signed by a single client to date. In addition, the production will take place in South Korea at Samsung Biologics' biomanufacturing facility, and the partnership will run through December 2037. With the agreement, Samsung Biologics' total contracts for 2024 have exceeded USD 3.3 billion.
-
In March 2023, Pfizer mentioned a USD 183 million contract manufacturing partnership with Samsung Biologics. In addition, after two years, the Samsung Biologics mentioned the agreement to manufacture COVID-19 vaccines for Moderna, as well as for other leading mRNA vaccine producers, including Pfizer. This is the first time Korea's top conglomerate Samsung Group's biotechnology unit unveiled its CMO contract with Pfizer.
Pharmaceutical Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 155.11 billion
Revenue forecast in 2033
USD 310.97 billion
Growth rate
CAGR of 10.45% from 2026 to 2033
Historical Year
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, product, drug, scale of operation, workflow, application, end use, company size, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
AbbVie Contract Manufacturing; Almac Group; Boehringer Ingelheim BioXcellence; Catalent; Dr. Reddy's Laboratories; Fareva; Fujifilm Diosynth Biotechnologies; Jubilant HollisterStier; Lonza Group; Patheon (Thermo Fisher Scientific); Pfizer CentreOne; Prakruti Life Science; Recipharm; Samsung Biologics; Siegfried Holding AG; Vetter Pharma; WuXi AppTec
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Pharmaceutical Contract Manufacturing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global pharmaceutical contract manufacturing market report based on service, product, scale of operation, workflow, application, end use, company size, and region:
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
API Manufacturing
-
Finished Drug Product Manufacturing
-
Oral Solids
-
Liquids
-
Topical
-
Others
-
-
Packaging Services
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecule
-
Biologics
-
Monoclonal Antibodies
-
Vaccines
-
Recombinant Proteins
-
Others
-
-
-
Drug Outlook (Revenue, USD Million, 2021 - 2033)
-
Generics
-
Branded
-
-
Scale of Operation Outlook (Revenue, USD Million, 2021 - 2033)
-
Batch
-
Continuous
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Commercial
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Infectious Diseases
-
Neurological Disorders
-
Cardiovascular Disease
-
Metabolic Disorders
-
Autoimmune Diseases
-
Respiratory Diseases
-
Ophthalmology
-
Gastrointestinal Disorders
-
Hormonal Disorders
-
Hematological Disorders
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotechnology companies
-
-
Company Size Outlook (Revenue, USD Million, 2021 - 2033)
-
Small
-
Medium
-
Large
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global pharmaceutical contract manufacturing market size was estimated at USD 141.74 billion in 2025 and is expected to reach USD 155.11 billion in 2026.
b. The global pharmaceutical contract manufacturing market is expected to grow at a compound annual growth rate of 10.45% from 2026 to 2033 to reach USD 310.97 billion by 2033.
b. North America dominated the pharmaceutical contract manufacturing market with a share of 38.44% in 2025. This is attributable to several factors, including technological advancements, growing demand for pharmaceutical products, and the increasing outsourcing of pharmaceutical production to third-party manufacturers.
b. Some key players operating in the pharmaceutical contract manufacturing market include AbbVie Contract Manufacturing, Almac Group, Boehringer Ingelheim BioXcellence, Catalent, Dr. Reddy's Laboratories, Fareva, Fujifilm Diosynth Biotechnologies, Jubilant HollisterStier, Lonza Group, Patheon (Thermo Fisher Scientific), Pfizer CentreOne, Prakruti Life Science, Recipharm, Samsung Biologics, Siegfried Holding AG, Vetter Pharma, WuXi AppTec
b. Key factors driving the pharmaceutical contract manufacturing market growth include the surge in clinical and commercial workflow realignment among pharmaceutical manufacturers. The pharmaceutical industry is witnessing a shift toward outsourcing complex manufacturing processes to specialized partners, such as contract manufacturing organizations (CMOs), which is driving market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









