- Home
- »
- Pharmaceuticals
- »
-
Peptide Therapeutics Market Size, Industry Report, 2033GVR Report cover
![Peptide Therapeutics Market Size, Share & Trends Report]()
Peptide Therapeutics Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Innovative, Generic), By Application (Metabolic Disorders, Cancer), By Type Of Manufacturers, By Route Of Administration, By Synthesis Technology, By Region, And Segment Forecasts
- Report ID: 978-1-68038-179-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
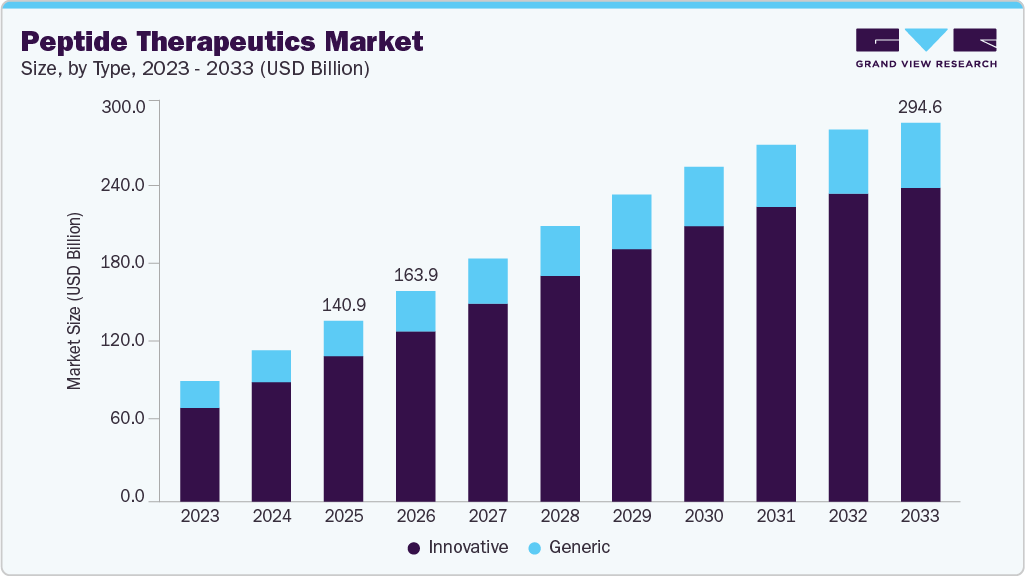
Peptide Therapeutics Market Summary
The global peptide therapeutics market size was estimated at USD 140.86 billion in 2025 and is projected to reach USD 294.58 billion by 2033, growing at a CAGR of 8.73% from 2026 to 2033. The market is expanding steadily, supported by rising demand for targeted and high efficacy treatment options across major disease areas.
Key Market Trends & Insights
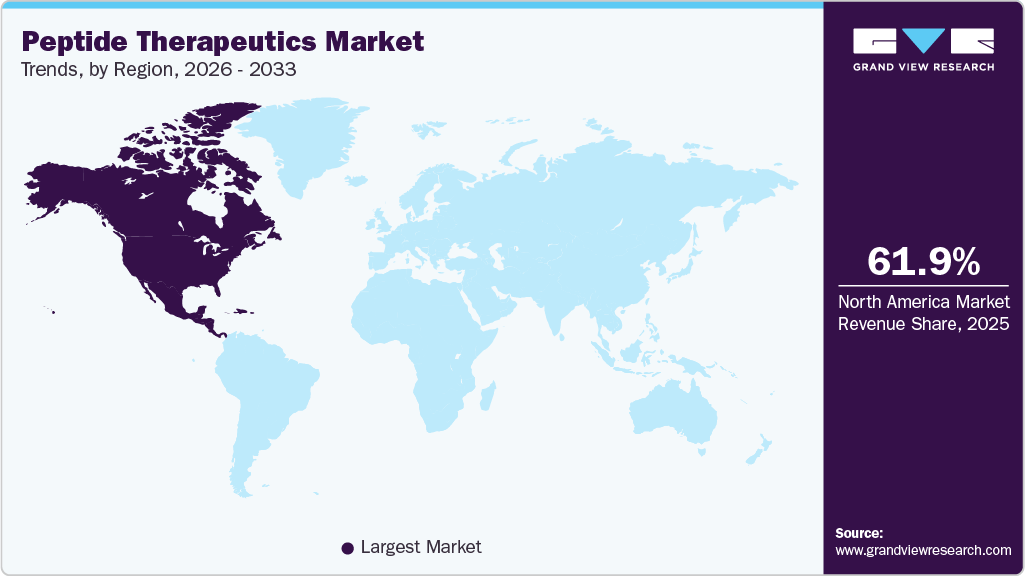
- North America peptide therapeutics industry held the largest share of 61.99% of the global market in 2025.
- The peptide therapeutics industry in the U.S. is expected to grow significantly over the forecast period.
- By type, the innovative segment held the largest market share of 80.08% in 2025.
- By application, the metabolic disorders segment held the largest market share in 2025.
- By type of manufacturers, the in-house segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 140.86 Billion
- 2033 Projected Market Size: USD 294.58 Billion
- CAGR (2026-2033): 8.73%
- North America: Largest market in 2025
The increasing prevalence of metabolic disorders, particularly diabetes and obesity, continues to generate substantial demand for peptide-based drugs across developed and emerging markets. Strong commercial performance of GLP-1 receptor agonists has reinforced confidence in peptide platforms and accelerated research investments. Pharmaceutical companies are prioritizing peptide innovation to address unmet clinical needs with improved safety, selectivity, and tolerability profiles. Advances in peptide engineering have enhanced molecular stability, receptor specificity, and extended half-life, strengthening overall therapeutic performance. Growing physician preference for precision therapies is further elevating clinical adoption. Expanding biologics acceptance across healthcare systems is creating favorable conditions for peptide commercialization. These combined factors are contributing to sustained revenue expansion and long-term market growth.Growth momentum is further supported by expanding applications in oncology, gastrointestinal disorders, cardiovascular conditions, and rare endocrine diseases. Peptide receptor targeting strategies are improving therapeutic precision and minimizing systemic toxicity in cancer treatment, while continuous innovation in long-acting formulations has reduced dosing frequency and improved patient adherence across chronic disease segments. Oral peptide delivery technologies are advancing through absorption enhancers and formulation optimization, increasing patient convenience and broadening eligible populations.
Companies are investing in differentiated pipeline candidates with dual or multi receptor activity to improve clinical outcomes, and strategic collaborations between biotechnology firms and major pharmaceutical companies are accelerating development timelines and strengthening commercialization capabilities. For instance, in September 2025, GlobeNewswire reported that once weekly injectable semaglutide was associated with a 23% reduced risk of major adverse cardiovascular events compared to dulaglutide in nearly 60,000 US Medicare patients aged 66 years or older, along with a 26% lower risk of death and a 25% lower risk of heart attack, stroke, hospitalization for unstable angina or heart failure and death from any cause, with results presented at the 2025 European Association for the Study of Diabetes Annual Meeting.
Manufacturing advancements are contributing to greater scalability, consistency, and cost efficiency across the peptide value chain. Improvements in solid phase peptide synthesis and hybrid production technologies are enhancing yield, purity, and batch reproducibility, while expanded capacity among contract development and manufacturing organizations is supporting global supply requirements. For instance, in May 2025, Pharma Manufacturing reported that Swiss based Bachem expanded multisite production capacities across Switzerland, the United States, and the United Kingdom, commenced construction of a new large scale facility known as Building K in Bubendorf, increased annual peptide production capacity at its Vista, California site to nearly one metric ton, doubled diagnostic peptide output at its St Helens site in the UK, and initiated concept design work for an additional facility in Sisseln, Switzerland, further strengthening global peptide manufacturing infrastructure.
Market Concentration & Characteristics
The peptide therapeutics industry demonstrates a high degree of innovation driven by continuous advancements in molecular design, stability enhancement, and receptor specificity. Companies are developing long-acting analogs, multi receptor agonists, and peptide conjugates to improve efficacy and treatment durability. Novel drug delivery platforms, including oral and depot formulations, are expanding clinical applicability. Research efforts are focused on improving bioavailability and reducing degradation during systemic circulation. Integration of computational modeling and structure-based design is accelerating discovery timelines. Strong pipeline activity across metabolic, oncology, and rare disease segments reflects sustained innovation intensity.
Barriers to entry in the peptide therapeutics industry remain moderate to high due to complex development requirements and capital-intensive manufacturing infrastructure. Specialized expertise in peptide synthesis, purification, and analytical validation is essential for commercialization. Clinical development timelines are lengthy, with substantial investment required for safety and efficacy evaluation. Intellectual property protection around peptide sequences and formulations limits early-stage competition. Established players benefit from integrated supply chains and global distribution networks. These structural challenges restrict rapid entry of smaller participants without strategic partnerships
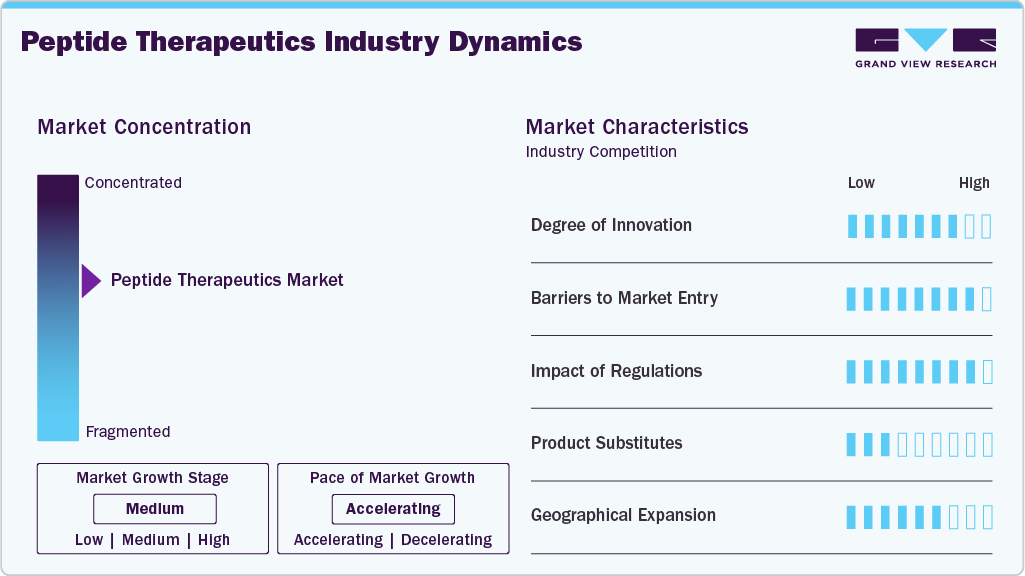
Regulatory frameworks play a critical role in shaping product development, approval timelines, and commercialization strategies in the peptide therapeutics industry. Stringent quality standards govern peptide synthesis, purity levels, and batch consistency. Comprehensive clinical trial data are required to demonstrate safety, pharmacokinetics, and therapeutic efficacy. Regulatory scrutiny is particularly rigorous for innovative peptides with novel mechanisms of action. Post marketing surveillance requirements add ongoing compliance responsibilities for manufacturers. Clear regulatory pathways for generics and follow on peptides influence competitive dynamics and pricing strategies.
Peptide therapeutics face competition from small molecule drugs, monoclonal antibodies, and emerging gene-based therapies across multiple indications. Small molecules often offer oral convenience and lower manufacturing costs in certain disease areas. Monoclonal antibodies provide high specificity and extended half-life for chronic conditions. Biosimilars are increasing competitive pressure in established biologics segments. Advanced cell and gene therapies are expanding treatment options for rare and complex disorders. Competitive positioning of peptides depends on clinical differentiation, safety profile, and dosing advantages.
Geographical expansion in the peptide therapeutics industry is supported by increasing healthcare infrastructure development and rising chronic disease prevalence across emerging economies. North America and Europe maintain strong market leadership through advanced research capabilities and high biological adoption. Asia Pacific is experiencing rapid growth driven by expanding pharmaceutical manufacturing capacity and improving access to specialty care. Multinational companies are strengthening regional partnerships to enhance distribution and local production capabilities. Market penetration strategies include localized clinical trials and region-specific product launches. Broader global reach is reinforcing long term revenue diversification.
Type Insights
The innovative segment dominated the market with the largest revenue share of 80.08% in 2025, supported by rapid clinical advancement of next generation peptide candidates and broader integration across oncology, metabolic, and rare disease treatments. Continuous improvements in peptide stability, receptor selectivity, and half-life enhanced therapeutic precision and strengthened commercial viability. Pharmaceutical companies increased investments in engineered peptide platforms to deliver higher efficacy with optimized safety profiles, reinforcing competitive differentiation. For instance, in July 2025, Health reported findings from a phase 2 trial of the investigational monthly glucagon like peptide 1 drug MariTide, in which 592 people with obesity were enrolled. Of these, 72 percent completed the 52-week study while 28 percent discontinued. Participants without diabetes lost up to 16.2 percent of body weight, or 19.9 percent when excluding those who discontinued, while participants with both obesity and type 2 diabetes lost up to 12.3 percent, or 17 percent when excluding those who discontinued. The therapy was also associated with up to a 2.2 percentage point improvement in A1C levels, along with reductions in blood pressure, inflammation markers, and lipid levels compared with placebo, which demonstrated comparatively smaller changes.
The generic segment is projected to grow at a CAGR of 6.95% over the forecast period, fueled by increasing availability of cost-effective peptide drug alternatives that expand patient access and widen treatment uptake. Generic peptide products reduce treatment costs for chronic conditions including diabetes and obesity, which continues to attract broader prescribing patterns across diverse healthcare settings. For instance, in August 2025, Investor’s Business Daily reported that the FDA approved the first-ever generic version of the GLP-1 weight-loss drug Saxenda, allowing Teva to market the lower-cost liraglutide alternative, which directly challenged the original brand’s pricing and reinforced market demand for affordable peptides. Growing competition in off-patent peptide classes will encourage more manufacturers to enter the segment, enhancing supply diversity and driving overall market growth.
Application Insights
The metabolic disorders segment dominated the market with the largest revenue share of 63.63% in 2025, driven by the increasing global prevalence of obesity and diabetes and the strong clinical adoption of peptide-based therapies such as GLP 1 receptor agonists. Rising cases of type 2 diabetes and associated cardiovascular complications accelerated demand for targeted and long-acting peptide therapeutics with improved safety and efficacy profiles. Expanding regulatory approvals, advancements in formulation technologies, and growing healthcare expenditure across developed and emerging economies further strengthened segment growth. Pharmaceutical companies also intensified research and development efforts to introduce next generation metabolic peptide drugs. For instance, in June 2025, Cell Reports reported that identifying corticotropin releasing hormone required extraction of 490,000 ovine hypothalamus fragments, and over 95% to 99% of peptides detected in complex samples were degradation fragments, highlighting significant research complexity.
The pain segment is projected to grow at a CAGR of 7.47% over the forecast period, fueled by rising demand for effective non-opioid and targeted analgesic therapies that reduce reliance on traditional painkillers while offering safer treatment profiles. Enhanced research into peptide and non-peptide pain targets has expanded clinical options for acute and chronic pain conditions, increasing physician confidence in alternative pain management approaches. For instance, in January, 2025, Reuters reported that the U.S. FDA approved Journavx (suzetrigine), a first-in-class non-opioid pain medication that significantly reduced surgical pain compared to placebo, marking a major step forward in non-opioid pain treatment development and reinforcing focus on advanced analgesic options for patients.
Type Of Manufacturers Insights
The in-house segment dominated the market with the largest revenue share of 64.86% in 2025, driven by expanding internal manufacturing investments and the need for tighter quality and supply chain control. Major pharmaceutical companies strengthened proprietary peptide synthesis capabilities to support high demand therapies in metabolic and oncology indications, while integrated production facilities enabled faster scale up, improved batch consistency, and stronger protection of intellectual property. For instance, in December 2025, Reuters reported that Eli Lilly and Company invested more than USD 6 billion to build a new active drug ingredient manufacturing facility in Huntsville, Alabama, which was the third such U.S. site announced and was planned to produce small molecule synthetic and peptide medicines including its oral GLP 1 drug orforglipron. Construction was slated to begin in 2026 and targeted for completion in 2032, creating about 3,000 construction jobs and 450 permanent roles, with each dollar invested expected to generate up to four dollars in local economic activity from over 300 applications.
The outsourced segment is projected to grow at a CAGR of 8.90% over the forecast period, fueled by rising demand for specialized peptide synthesis expertise and flexible manufacturing capacity. Small and mid sized biotechnology companies increasingly rely on contract development and manufacturing organizations to reduce capital expenditure and accelerate clinical timelines. Outsourcing enables access to advanced solid phase peptide synthesis platforms, high purity purification systems, and regulatory compliant production without large infrastructure investments. For instance, Sep 12, 2024, BioPharm International reported that SK pharmteco expanded its California peptide manufacturing facility to address growing global demand and overcome purification bottlenecks, highlighting strong momentum in contract peptide production services. Expanding clinical pipelines and increasing complexity of peptide molecules are expected to further drive reliance on experienced outsourced partners.
Route Of Administration Insights
The parenteral route segment dominated the market with the largest revenue share of 83.99% in 2025, driven by the high clinical preference for injectable peptide therapies that deliver consistent systemic exposure and optimal therapeutic outcomes. Many peptide drugs are inherently unstable in the digestive system, making injections the preferred administration method for sustained efficacy in chronic conditions. For instance, August, 2025, Reuters reported that Novo Nordisk launched its injectable obesity drug Wegovy in South Africa, expanding its global reach and underscoring strong demand for parenteral peptide formulations in emerging markets. Such launches highlight continued reliance on injectable peptide delivery as a key driver of revenue within the market’s dominant segment.
The others segment is projected to grow at a CAGR of 9.47% over the forecast period, fueled by increasing development of alternative peptide delivery systems such as transdermal patches, implantable depots, and sustained release biodegradable implants. These innovative approaches aim to enhance patient convenience, improve adherence, and maintain stable therapeutic drug levels over extended durations. Long acting implant technologies are gaining attention for chronic disease management, particularly in endocrine and metabolic disorders. Advances in polymer based drug delivery platforms are enabling controlled peptide release with reduced dosing frequency. Research efforts are also exploring microneedle arrays and localized delivery systems for targeted therapeutic action. Expanding clinical validation of these novel administration formats is expected to support steady growth within this emerging route segment.
Synthesis Technology Insights
The recombinant DNA technology segment dominated the market with the largest revenue share of 80.18% in 2025, due to its widespread use in the large-scale production of complex therapeutic peptides with high purity, consistency, and scalability. This technology enables precise genetic manipulation to express peptide sequences in host systems such as bacteria, yeast, and mammalian cells, ensuring efficient and cost-effective manufacturing. The increasing demand for insulin analogs, growth hormones, and other recombinant peptide drugs for chronic conditions such as diabetes and hormonal disorders has significantly supported segment growth. In addition, advancements in expression systems, fermentation technologies, and downstream purification processes have improved yield and reduced production timelines. Strong regulatory acceptance and the growing pipeline of biologics and biosimilars further reinforced the dominance of recombinant DNA technology in peptide therapeutic development.
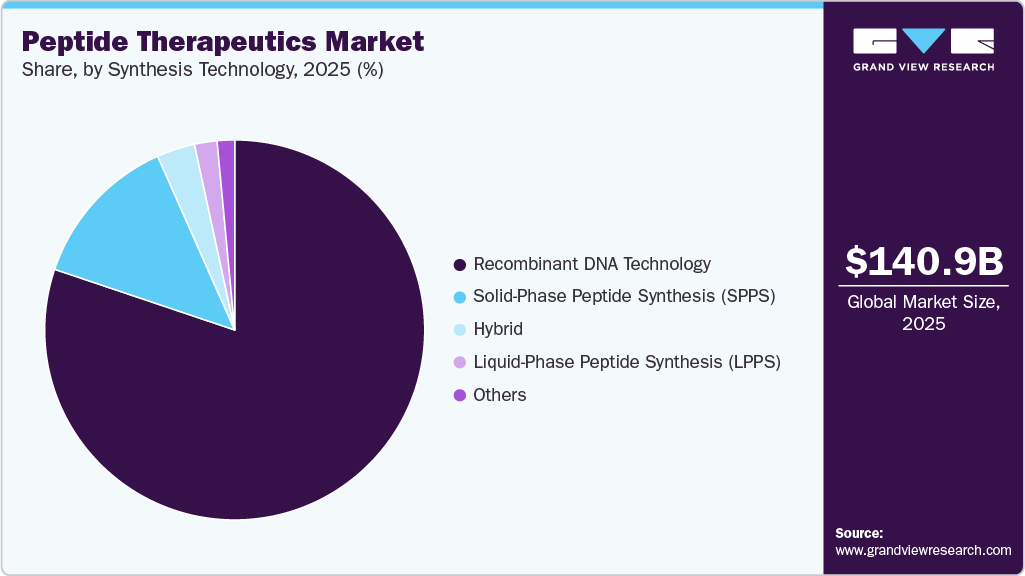
The Liquid Phase Peptide Synthesis, LPPS, segment is projected to grow at a CAGR of 7.62% over the forecast period due to its strong suitability for large scale industrial peptide production, particularly for short to medium length peptides. LPPS offers advantages such as cost effectiveness for bulk manufacturing, easier purification processes, and established scalability compared to solid phase techniques in certain applications. It is widely adopted for the synthesis of active pharmaceutical ingredients used in metabolic disorders, oncology, and cardiovascular therapies. In addition, increasing demand for high purity therapeutic peptides, expanding contract manufacturing activities, and technological refinements in solution phase chemistry are further supporting segment growth.
Regional Insights
North America peptide therapeutics market held the largest global share of 61.99% in 2025, supported by advanced biopharmaceutical research infrastructure and strong commercialization capabilities. High prevalence of metabolic disorders and cancer continues to generate sustained demand for innovative peptide drugs. Leading pharmaceutical companies maintain extensive peptide pipelines and manufacturing facilities across the region. Strong adoption of GLP-1 receptor agonists and endocrine therapies supports revenue leadership. Sophisticated healthcare systems enable rapid integration of newly approved peptide treatments. Strategic collaborations between biotechnology firms and major pharmaceutical companies further strengthen market expansion.
U.S. Peptide Therapeutics Market Trends
The U.S. peptide therapeutics industry represents the largest contributor within North America, driven by high specialty drug utilization and rapid uptake of novel therapies. Robust clinical trial activity supports continuous peptide innovation across metabolic and oncology segments. Presence of major market players enhances product availability and commercialization efficiency. Advanced diagnostic capabilities improve early disease detection, increasing treatment volumes. Strong investment in research and development accelerates pipeline progression. High awareness among healthcare providers supports broad prescription of peptide based medications.
Europe Peptide Therapeutics Market Trends
Europe peptide therapeutics industry held a significant share in 2025, supported by established pharmaceutical manufacturing capabilities and expanding biologics adoption. Increasing incidence of diabetes, obesity, and gastrointestinal disorders sustains demand for peptide treatments. Regional companies are investing in advanced synthesis technologies to enhance production efficiency. Growth in oncology focused peptide therapies contributes to market stability. Collaborative research networks support innovation across academic and commercial sectors. Consistent introduction of long acting peptide formulations strengthens therapeutic portfolios
The UK peptide therapeutics industry is expanding steadily, supported by a strong life sciences ecosystem and advanced clinical research infrastructure. Rising cases of metabolic and cardiovascular conditions are increasing demand for targeted therapies. Pharmaceutical firms are focusing on specialty peptides for oncology and rare endocrine diseases. Growth in contract manufacturing services enhances regional production capabilities. Integration of novel drug delivery technologies supports improved treatment adherence. Increasing participation in multinational clinical studies strengthens pipeline development.
Germany peptide therapeutics industry represents a key market in Europe, supported by strong pharmaceutical engineering expertise and advanced manufacturing standards. Demand for peptide therapies is rising across metabolic and oncology applications. Local companies emphasize high quality synthesis processes and analytical precision. Expansion of specialty care centers is improving access to advanced biologic treatments. Increased collaboration between biotech startups and established firms supports innovation. Export oriented pharmaceutical operations further reinforce Germany’s strategic position.
France peptide therapeutics industry maintains steady growth, driven by expanding biologics utilization and rising chronic disease prevalence. Demand for endocrine and gastrointestinal peptide drugs remains strong. Domestic pharmaceutical companies are investing in formulation improvements and long acting peptide development. Clinical research initiatives are enhancing early phase peptide discovery. Adoption of precision medicine approaches is strengthening oncology related applications. Increasing partnerships with international firms are expanding commercial reach.
Asia Pacific Peptide Therapeutics Market Trends
Asia-Pacific peptide therapeutics industry is expected to register a CAGR of 7.98% over the forecast period due to rapid growth in diabetes and obesity prevalence is expanding the target patient population. Pharmaceutical manufacturing capacity is increasing across major economies in the region. Rising healthcare expenditure and improved access to specialty treatments are supporting market expansion. Global companies are establishing regional production and distribution partnerships. Expanding clinical research activities are strengthening local innovation capabilities.
Japan peptide therapeutics industry represents a mature market supported by advanced pharmaceutical technologies and strong demand for metabolic treatments. High aging population contributes to increased incidence of chronic diseases. Domestic firms focus on high purity synthesis and specialized peptide formulations. Adoption of innovative drug delivery systems is improving patient compliance. Strong collaboration between research institutions and industry supports pipeline development. Continuous product refinement enhances competitiveness within the region.
China peptide therapeutics industry is emerging as a high growth market for peptide therapeutics, supported by expanding pharmaceutical manufacturing infrastructure. Rising prevalence of diabetes and cardiovascular disorders is increasing treatment demand. Domestic companies are scaling up solid phase peptide synthesis capabilities. Growing participation in global clinical trials strengthens innovation exposure. Increasing availability of generic peptide drugs enhances affordability. Expansion of hospital networks improves access to specialty biologic therapies.
Latin America Peptide Therapeutics Market Trends
Latin America peptide therapeutics industry is experiencing gradual growth driven by rising awareness of advanced biologic treatments. Increasing incidence of metabolic and gastrointestinal diseases supports therapeutic demand. Regional pharmaceutical distributors are expanding access to branded peptide drugs. Growth in private healthcare facilities enhances specialty treatment adoption. Multinational companies are strengthening distribution networks across major economies. Market expansion remains steady with improving healthcare infrastructure.
Brazil peptide therapeutics industry leads the Latin American market, supported by a large patient base and expanding pharmaceutical distribution channels. Rising prevalence of obesity and endocrine disorders is increasing prescription volumes. Urban healthcare centers are adopting innovative peptide therapies at a faster pace. Partnerships between domestic and international firms enhance product availability. Investment in local manufacturing capabilities supports supply chain efficiency. Growing physician familiarity with biologics contributes to sustained market demand.
Middle East & Africa Peptide Therapeutics Market Trends
The Middle East & Africa peptide therapeutics industry is gradually expanding, supported by improving healthcare infrastructure and specialty care services. Increasing prevalence of diabetes is a major contributor to treatment demand. Multinational pharmaceutical companies are expanding distribution agreements across key countries. Adoption of advanced metabolic and oncology therapies is rising in urban centers. Growing investment in hospital facilities enhances access to peptide medications. Market development remains moderate with long term growth potential.
Saudi Arabia peptide therapeutics industry represents a leading market within Middle East & Africa, driven by high diabetes prevalence and strong demand for metabolic therapies. Expansion of specialty clinics is supporting increased biologic drug utilization. International pharmaceutical firms are strengthening their regional presence through partnerships. Rising awareness of advanced endocrine treatments is improving prescription rates. Modern hospital infrastructure supports administration of injectable peptide drugs. Continued focus on expanding pharmaceutical supply chains reinforces market growth prospects.
Key Peptide Therapeutics Company Insights
Eli Lilly and Company and Novo Nordisk A/S dominate the peptide therapeutics industry through strong leadership in GLP-1 based treatments for diabetes and obesity. Sanofi and Pfizer Inc. maintain broad specialty portfolios across endocrine and inflammatory disorders. Amgen Inc. and F. Hoffmann-La Roche Ltd focus on oncology and rare disease peptide innovation. Takeda Pharmaceutical Company Limited, AstraZeneca, and Novartis AG expand targeted biologic strategies. Teva Pharmaceutical Industries Ltd., GSK plc, Ironwood Pharmaceuticals, Inc., Radius Health, Inc., and Ipsen Pharma strengthen competition through specialty and gastrointestinal peptide offerings.
Key Peptide Therapeutics Companies:
The following key companies have been profiled for this study on the peptide therapeutics market.
- Eli Lilly and Company
- Pfizer Inc.
- Amgen Inc.
- Takeda Pharmaceutical Company Limited
- AstraZeneca
- Teva Pharmaceutical Industries Ltd.
- Sanofi
- F. Hoffmann-La Roche Ltd
- Novartis AG
- Novo Nordisk A/S
- GSK plc
- Ironwood Pharmaceuticals, Inc.
- Radius Health, Inc.
- Ipsen Pharma
Recent Developments
-
In February 2026, Zonsen PepLib Biotech Inc. and Eli Lilly entered into a global research and development collaboration to advance novel peptide based drug candidates, under which PepLib would utilize its proprietary peptide libraries to identify optimized active molecules while Lilly would lead Investigational New Drug enabling studies, clinical development, regulatory approvals, and commercialization activities.
-
In December 2025, Eli Lilly and Company announced it planned to invest more than USD 6 billion to build a new active pharmaceutical ingredient manufacturing facility in Huntsville, Alabama, which was expected to start construction in 2026 and be completed by 2032, generate about 3,000 construction jobs, create 450 permanent jobs including engineers, scientists, operations personnel and lab technicians, and produce small-molecule synthetic and peptide medicines including orforglipron, with the site chosen from over 300 applications and each dollar invested estimated to generate up to four dollars in local economic activity.
-
In December 2025, Pfizer Inc. entered into an exclusive global collaboration and license agreement with YaoPharma under which YaoPharma completed an ongoing Phase 1 trial of YP05002, granted Pfizer exclusive global development, manufacturing and commercialization rights, received a USD 150 million upfront payment, and was eligible for up to USD 1.935 billion in development, regulatory and commercial milestone payments plus tiered royalties, while Pfizer planned combination studies with its PF-07976016 in Phase 2 development.
Peptide Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 163.98 billion
Revenue forecast in 2033
USD 294.58 billion
Growth rate
CAGR of 8.73% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type, application, type of manufacturers, route of administration, synthesis technology, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Eli Lilly and Company; Pfizer Inc.; Amgen Inc.; Takeda Pharmaceutical Company Limited; AstraZeneca; Teva Pharmaceutical Industries Ltd.; Sanofi; F. Hoffmann-La Roche Ltd; Novartis AG; Novo Nordisk A/S; GSK plc; Ironwood Pharmaceuticals, Inc.; Radius Health, Inc.; Ipsen Pharma
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Peptide Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global peptide therapeutics market report based on application, type, type of manufacturer, route of administration, synthesis technology, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Innovative
-
Generic
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Metabolic Disorders
-
Cancer
-
Neurological Disorders
-
Gastrointestinal Disorders
-
Cardiovascular Disorders
-
Pain
-
Infectious Disease
-
Renal Disorders
-
Dermatological Disorders
-
Respiratory Disorders
-
Others
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Parenteral Route
-
Oral Route
-
Mucosal
-
Pulmonary
-
Others
-
-
Synthesis Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Recombinant DNA Technology
-
Solid-Phase Peptide Synthesis (SPPS)
-
Hybrid
-
Liquid-Phase Peptide Synthesis (LPPS)
-
Others
-
-
Type of Manufacturers Outlook (Revenue, USD Million, 2021 - 2033)
-
Inhouse
-
Outsourced
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some of the key players in peptide therapeutics market are Eli Lilly and Company, Pfizer Inc., Amgen Inc., Takeda Pharmaceutical Company Limited, AstraZeneca, Teva Pharmaceutical Industries Ltd., Sanofi, F. Hoffmann-La Roche Ltd, Novartis AG, Novo Nordisk A/S, GSK plc, Ironwood Pharmaceuticals, Inc., Radius Health, Inc. (Subsidiary of Gurnet Point Capital and Patient Square Capital), Ipsen Pharma
b. The global peptide therapeutics market size was valued at USD 140.86 billion in 2025 and is anticipated to reach USD 163.98 billion in 2026.
b. The global peptide therapeutics market is expected to witness a compound annual growth rate of 8.73% from 2026 to 2033 to reach USD 294.58 billion by 2033.
b. Based on type, the innovative segment dominated the market with the largest revenue share of 80.08% in 2025, supported by rapid clinical advancement of next generation peptide candidates and broader integration across oncology, metabolic, and rare disease treatments.
b. North America held the largest peptide therapeutics market share of 61.99% in 2025, supported by advanced biopharmaceutical research infrastructure and strong commercialization capabilities.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









