- Home
- »
- Clinical Diagnostics
- »
-
Kidney Function Tests Market Size, Industry Report, 2033GVR Report cover
![Kidney Function Tests Market Size, Share & Trends Report]()
Kidney Function Tests Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Clearance Tests, Urine Tests, Blood Tests, Dilution And Concentration Tests), By End Use (Hospitals, Diagnostic Laboratories), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-721-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Kidney Function Tests Market Summary
The global kidney function tests market size was valued at USD 1.12 billion in 2025 and is projected to reach USD 1.77 billion by 2033, growing at a CAGR of 5.87% from 2026 to 2033. Several factors are anticipated to drive market growth, including increased investments by industry players, growing research and development efforts, a high prevalence of renal diseases, government initiatives, and advancements in technology.
Key Market Trends & Insights
- North America kidney function tests industry dominated and accounted for a 36.16% share in 2025.
- The U.S. kidney function tests market is projected to grow significantly during the forecast period.
- By product, the clearance tests segment held the highest market share of 27.53% in 2025.
- Based on end use, the hospitals segment held the highest market share of 39.50% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.12 Billion
- 2033 Projected Market Size: USD 1.77 Billion
- CAGR (2026-2033): 5.87%
- North America: Largest Market in 2025
For instance, in February 2024, Simple HealthKit launched a new renal evaluation test for diabetes. The test is available for clinics and at home settings. Such developments are likely to propel the growth of kidney function tests industry.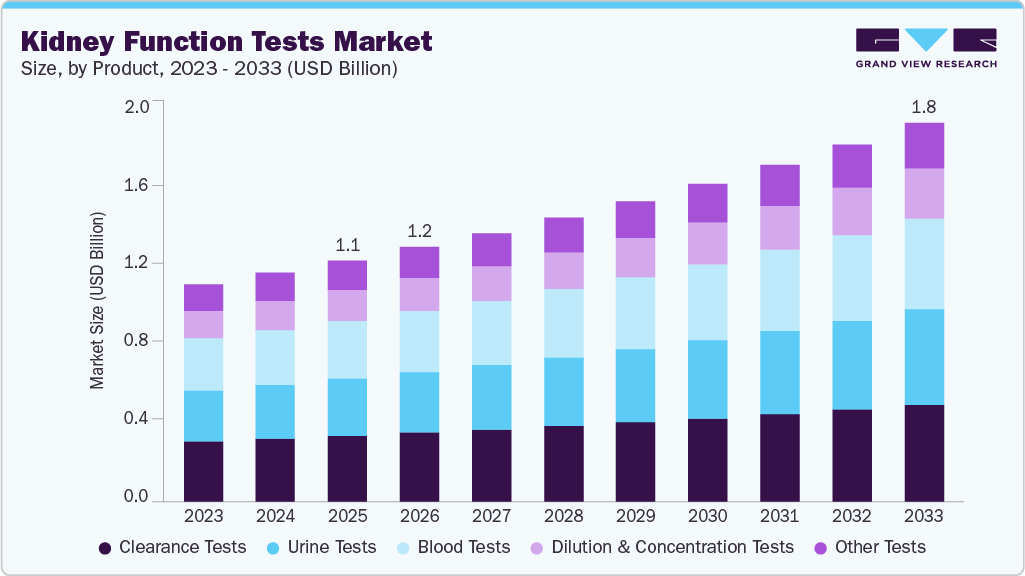
During the COVID-19 pandemic, the incidence of renal injury increased significantly. Research highlighted that the individuals with pre-existing renal conditions faced a higher risk of severe complications and poorer outcomes upon contracting COVID-19. Ongoing studies on the long-term effects of COVID-19 have also revealed its potential lasting impact on renal health. However, the pandemic disrupted healthcare services for renal disease patients, as lockdowns and social distancing mandates led to temporary closures of non-emergency medical services. This resulted in delays in diagnosing renal diseases due to the suspension of routine visits and diagnostic tests during peak pandemic phases.
The rising prevalence of acute kidney injury (AKI) and its progression to CKD is expected to propel the kidney function tests industry in the forecast period. CKD refers to persistent structural and functional abnormalities in the kidneys lasting at least three months. Conditions such as lifestyle-related diseases and kidney injuries are major contributors to CKD, which remains a global health burden and a leading cause of morbidity and mortality worldwide. According to the World Health Organization (WHO), renal disease has risen to become one of the top 10 leading causes of death globally over the past decade. Additionally, the National Kidney Foundation reports that approximately 37 million people in the U.S., or over one in seven adults, are affected by some form of renal disease.
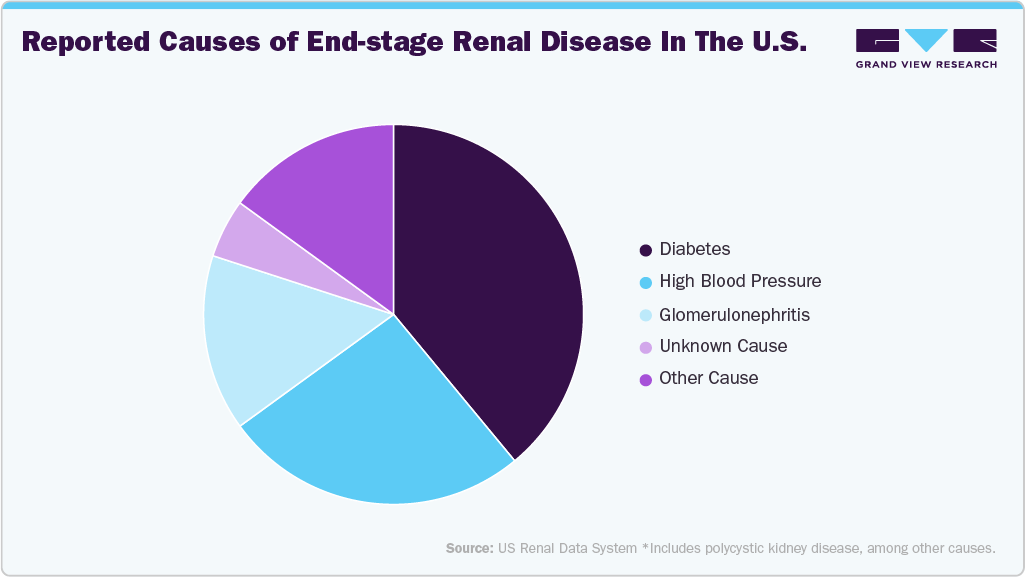
The growing prevalence of diabetes and hypertension remains the primary driver of CKD incidence. Nearly one-third of individuals with diabetes develop kidney-related complications, largely due to prolonged hyperglycemia and uncontrolled blood pressure. According to the Centers for Disease Control and Prevention, as of August 2023, the prevalence of total diagnosed and undiagnosed diabetes among U.S. adults stood at 15.8%, with 4.5% remaining undiagnosed, further increasing the pool of individuals at risk for kidney damage. On a global scale, the International Diabetes Federation projects that 783 million adults, nearly 1 in 8 people will be living with diabetes by 2045, representing a 46% increase, with type 2 diabetes accounting for over 90% of cases
The growing adoption of digital urinalysis emphasizes the increasing demand for home-based diagnostics, which is contributing significantly to market growth. For instance, Healthy.io, an Israel-based digital health start-up, developed an FDA-approved at-home smartphone urinalysis test designed to detect kidney-related issues such as proteinuria. Early detection through this test can prevent complications arising from renal diseases. Their urinalysis product, Dip.io, combines disposable cups and strips with smartphone cameras to analyze and interpret results. A trial conducted with Geisinger Health and the National Kidney Foundation (NKF) demonstrated a 71% adherence rate among patients with hypertension who had never undergone prior testing. This innovation is fueled by advancements in smartphone technology.
Additionally, increased R&D efforts to develop innovative renal care solutions are anticipated to accelerate the growth of the renal function tests industry during the forecast period. KidneyX, a collaboration between the U.S. Department of Health and Human Services and the American Society of Nephrology, is focused on advancing the diagnosis, treatment, and prevention of renal diseases. This initiative aims to improve outcomes for the 850 million people affected globally by driving innovation in renal devices, drugs, biologics, and other therapies. Similarly, Canada’s largest kidney research initiative, the Can-SOLVE Chronic Kidney Disease Network, received USD 11.8 million in funding to support its work through 2027. This partnership between researchers, healthcare providers, and patients is dedicated to finding solutions for nearly 4 million Canadians living with CKD
Recent Launches and Technological Advancements:
-
In October 2025, Roche launched a CE-marked, AI-based Kidney KlinRisk Algorithm designed to predict the risk of kidney function decline in patients with diabetes or hypertension, even before a formal diagnosis of chronic kidney disease (CKD). The algorithm supports early risk stratification and clinical decision-making, indirectly driving demand for confirmatory kidney function tests by enabling earlier screening and monitoring.
-
In September 2025, Renalytix, in collaboration with Tempus AI, expanded access to KidneyIntelX.dkd, an FDA-approved, AI-enabled blood test for diabetic kidney disease. The test combines biomarker analysis with machine learning to assess disease progression risk years before clinical symptoms appear, strengthening the role of advanced blood-based kidney diagnostics in early intervention strategies.
Technological advancements in renal function tests industry are expected to create significant growth opportunities during the forecast period. For instance, in August 2022, Bloom Diagnostics introduced a smart diagnostic system capable of detecting various medical conditions, including its latest addition, the Bloom Kidney Test. This test measures cystatin C levels in the blood to assess the glomerular filtration rate (GFR), an essential indicator of renal function. The system classifies results into three categories: mildly decreased function, normal function, and decreased function. Such innovative developments are likely to enhance diagnostic accuracy and accessibility, further driving market growth.
Market Concentration & Characteristics
The degree of innovation in the kidney function test market is high, as multi-biomarker panels represent a significant opportunity for advancement in early detection and disease monitoring. Beyond traditional serum creatinine and blood urea nitrogen (BUN) tests, companies are increasingly developing assays incorporating cystatin C, NGAL, and other novel biomarkers to improve diagnostic accuracy, particularly for early-stage chronic kidney disease (CKD) and acute kidney injury (AKI). In May 2024, Thermo Fisher Scientific launched a CXCL10-based urine testing service for kidney transplant recipients, enabling early detection of transplant rejection within 24 hours. The non-invasive test enhances post-transplant kidney monitoring and highlights ongoing innovation in urine-based diagnostics for specialized nephrology applications.
The level of mergers and acquisitions in the kidney function test market is moderate. Major diagnostic companies have pursued targeted acquisitions and partnerships to strengthen their renal testing portfolios and expand into specialized biomarker segments. For instance,in April 2025, Healthy.io acquired Labrador Health to advance smartphone-enabled, at-home urine testing solutions in the U.S., reflecting growing competitive focus on improving screening access and patient engagement.
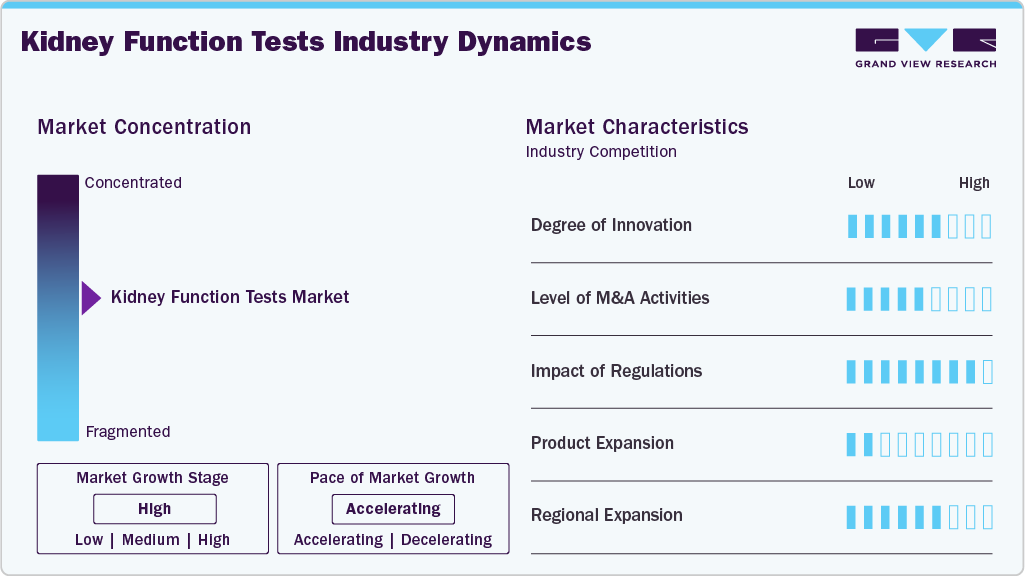
The impact of regulation on the kidney function test market is high, as products must comply with stringent in vitro diagnostic (IVD) standards, clinical validation requirements, and quality management systems. Regulatory authorities require strong evidence of analytical performance, reproducibility, and clinical relevance, particularly for novel biomarkers intended for early CKD detection.
Product expansion remains a key growth factor, with manufacturers increasingly launching advanced renal panels, home-based testing kits, and point-of-care solutions designed to support early diagnosis and continuous monitoring of kidney diseases. Expansion into preventive screening programs, diabetic nephropathy monitoring, and hospital-based acute care settings is broadening the application scope of kidney function testing.
Regional expansion in the kidney function test market is accelerating as emerging economies invest in improving access to diagnostic infrastructure and chronic disease management services. Growth in Asia Pacific, Latin America, and parts of the Middle East & Africa is supported by rising awareness of CKD, increasing diabetes and hypertension prevalence, and expanding laboratory networks.
Product insights
The clearance tests segment accounted for the largest share of the market at 27.53% in 2025. Clearance tests are considered the gold standard for assessing and monitoring glomerular filtration rate (GFR). These include creatinine and inulin tests, which serve as valuable supplements to traditional diagnostic methods, offering high accuracy in results. Their optimal sensitivity and precision minimize performance variability, ensuring reliable outcomes. Additionally, the affordability and widespread availability of these tests in the market have contributed to their growing adoption.
The urine tests is anticipated to experience the fastest growth of 6.75% in the forecast period. Urea quantification is one of the most commonly employed methods for evaluating renal function. Analyzing urea levels in urine, plasma, and serum is essential for identifying renal disorders and dysfunction. Urine tests, often paired with creatinine determination, play a pivotal role in diagnosing pre-renal, renal, and post-renal uremia. Additionally, increasing investments in renal function testing are expected to drive market growth. For instance, in May 2024, Thermo Fisher Scientific launched a CXCL10-based urine testing service for kidney transplant recipients, enabling early detection of transplant rejection within 24 hours and strengthening its presence in specialty nephrology diagnostics.
End Use Insights
The hospitals segment captured the highest share of 39.50% in 2025. Large number of tests being performed and processed in the hospitals is the major reason for the segment dominance. Diagnostic centers often operate in collaboration with hospitals, many of which have their in-house diagnostic setups. The continuous improvement of healthcare infrastructure is expected to enhance hospital facilities, allowing them to expand their diagnostic services. This expansion is likely to increase the demand for products like renal function tests, thereby driving market growth. Furthermore, favorable reimbursement policies and global initiatives by healthcare organizations to encourage the adoption of advanced diagnostic tests are expected to further accelerate growth of the kidney function tests industry.
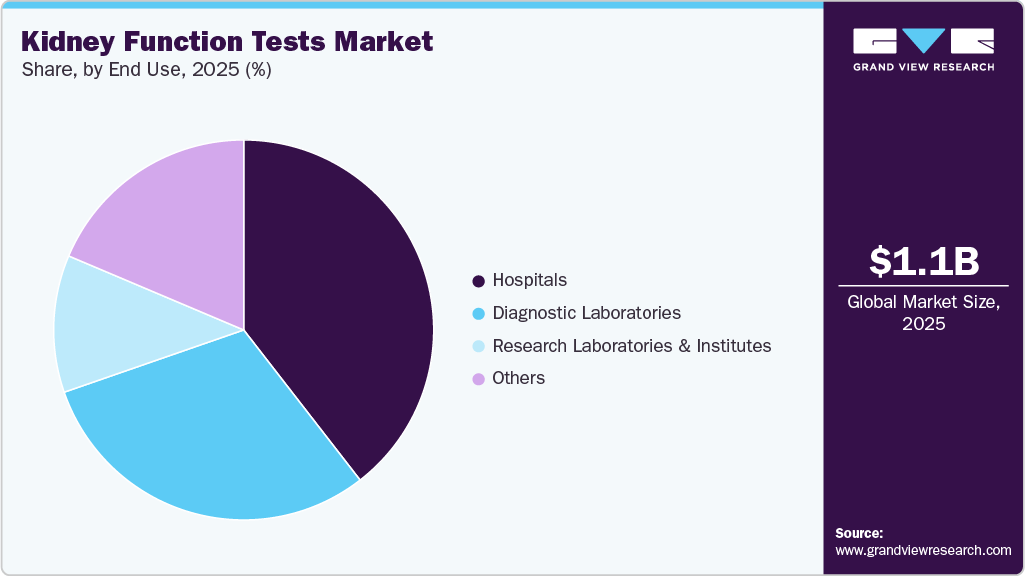
The diagnostic laboratories segment is predicted to grow at an exponential CAGR of 6.66% in the kidney function tests industry. The rising awareness of CKD, increasing demand for affordable diagnostic services, and rapid technological advancements are key factors anticipated to drive segment growth. Additionally, government initiatives aimed at improving access to diagnostic facilities, including reimbursement for renal function tests, further contribute to market expansion. These efforts are expected to enhance the availability and affordability of advanced diagnostic solutions, fueling growth in the segment.
Regional Insights
North America kidney function tests industry dominated and accounted for a 36.16% share in 2025. This market dominance can be attributed to well-established healthcare infrastructure, heightened awareness of CKD, and supportive reimbursement policies coupled with government initiatives. Additionally, key manufacturers in the region are emphasizing innovation. Furthermore, in September 2025, Renalytix announced a strategic collaboration with Tempus AI to advance and scale KidneyIntelX.dkd, an AI-enabled prognostic blood test for diabetic kidney disease, across the North American healthcare ecosystem. The collaboration integrates KidneyIntelX.dkd with Tempus’s extensive clinical data, genomics, and AI infrastructure, enabling more precise risk stratification and embedding renal risk prediction into routine clinical decision-making workflows. This initiative is positioned to support earlier identification of patients at high risk of kidney function decline and to guide targeted intervention strategies within large health systems.
U.S. Kidney Function Tests Market Trends
The U.S. kidney function tests market is projected to grow significantly during the forecast period, driven by several factors, including the rising prevalence of CKD and related conditions like diabetes and hypertension, which are major risk factors. Increasing awareness of early diagnosis and management of renal diseases has led to a higher demand for advanced diagnostic tools. renal diseases are a major cause of mortality in the U.S., with an estimated 37 million people, or about 1 in 7 Americans, living with CKD. However, nearly 40% of individuals with impaired renal function are unaware of their condition. In parallel, companies are expanding capabilities through service launches and strategic investments aimed at specialized and decentralized testing. In May 2024, Thermo Fisher Scientific launched a CXCL10-based urine testing service for kidney transplant recipients, enabling early detection of transplant rejection within 24 hours and strengthening its presence in specialty nephrology diagnostics.
Europe Kidney Function Tests Market Trends
The Europe kidney function tests market is likely to emerge as a lucrative region. Europe has high prevalence of geriatric population, and aging is a significant risk factor for renal disease. As the elderly population grows, there is a higher demand for renal function tests to monitor and manage age-related renal functions.
The UK kidney function tests market is projected to grow during the forecast period. CKD is becoming more widespread in the UK, primarily driven by the rising incidences of diabetes, hypertension, and obesity. According to Kidney Research UK, more than 7 million people are living with CKD which is likely to grow to 8.3 million by 2032. This high prevalence is fueling demand for renal function tests for early detection and better disease management. The UK KFT market operates under NHS commissioning and regional pathology networks; large suppliers Roche, Abbott, Siemens Healthineers and Danaher dominate instrumentation and reagent supply and in April 2025, Healthy.io’s strategic acquisition of Labrador Health expands capabilities for quantitative at-home testing and accelerates UK deployment potential. KDIGO guideline updates and inclusion of prognostic assays have also raised clinician interest in biomarker-driven testing.
The France kidney function tests market is expected to show steady growth over the forecast period. Well-developed healthcare infrastructure, including robust diagnostic capabilities, facilitates the widespread availability of renal function tests across the country.
The Germany kidney function tests market is projected to expand during the forecast period. Germany’s KFT market is shaped by a dense cluster of global and domestic diagnostics capabilities that combine manufacturing scale with deep hospital penetration. Siemens Healthineers (headquartered in Germany), Roche, Abbott, Danaher and Sysmex compete on reagent menus, high-throughput automation, and hospital IT integration. In May 2025, Boditech Med and Germany-based SphingoTec GmbH announced the commercial launch of the AFIAS sphingotest penKid assay. This IVDR-marked test measures Proenkephalin A 119-159 (penKid) in whole blood/plasma to provide real-time assessment of kidney function in critically ill patients, especially those with sepsis or acute kidney injury (AKI). It is designed to act as an early biomarker, detecting changes up to 48 hours before conventional markers like serum creatinine
Asia Pacific Kidney Function Tests Market Trends
The Asia Pacific kidney function tests market is expected to experience the highest growth rate of 7.6% CAGR during the forecast period. Factors such as rising rates of diabetes, hypertension, and obesity, which are common in the region, are contributing to a higher incidence of CKD. Countries like China, India, and Japan are seeing a significant rise in kidney-related diseases, which is fueling demand for renal function tests.
The China kidney function tests market is projected to expand throughout the forecast period. The Chinese government has launched several initiatives to raise awareness about kidney disease and promote early detection. National health campaigns aimed at reducing the prevalence of chronic diseases, including kidney disease, and improving access to healthcare services are likely to fuel market growth.
The Japan kidney function tests market is anticipated to grow during the forecast period, driven by its large geriatric population. As kidney function naturally declines with age, the demand for regular kidney function testing is increasing among the elderly. With an aging demographic, there is a rising need for early diagnosis and monitoring of CKD, making kidney function tests more crucial in the country.
Latin America Kidney Function Tests Market Trends
The Latin America kidney function tests marketis likely to show significant growth over the forecast period. The aging population, particularly in countries like Brazil and Argentina, is further contributing to this growth.
The Brazil kidney function tests market is anticipated to grow during the forecast period. Technological advancements are also helping to drive market growth. The adoption of point-of-care (POC) testing devices and home-based monitoring kits is increasing in Brazil, allowing individuals to track their kidney function at home and reducing the need for hospital visits.
Saudi Arabia Kidney Function Tests Market Trends
The Saudi Arabia kidney function tests market is anticipated to experience substantial growth in forecast period. The government has been actively addressing the rising burden of kidney disease through various initiatives. The Ministry of Health (MOH) has implemented national programs aimed at improving awareness of kidney diseases and encouraging early detection, especially among high-risk populations.
Key Kidney Function Tests Company Insights
Key players in this market are implementing various strategies, including new product launches, geographical expansion, partnerships, and collaborations, to expand their market presence. For instance, in November 2024, a group of researchers at Chung-Ang University in South Korea has developed a reliable test for the detection of SDMA in urine, which acts as an accurate indicator for kidney health detection.
Key Kidney Function Tests Companies:
The following key companies have been profiled for this study on the kidney function tests market
- Abbott
- Danaher
- F. Hoffmann-La Roche Ltd.
- Sysmex Corporation
- Siemens Healthineers
- Randox Laboratories Ltd
- Quest Diagnostics
- ACON Laboratories, Inc.
- Nova Biomedical Corporation
- Laboratory Corporation of America Holdings
Recent Developments
-
In December 2025, Roche Diagnostics Oy and Sysmex Nordic agreed to transfer distribution of Sysmex’s urine analysis product portfolio in Finland impacting availability and support of urinalysis solutions used in renal screening.
-
In December 2025, Sysmex signed an MOU to build its first diagnostic reagent factory in Saudi Arabia, strengthening regional supply of urinalysis and other diagnostic reagents that labs use in tests relevant to kidney health.
-
In October 2025, Quest completed the acquisition of the Spectra Laboratories clinical testing assets, positioning it to provide dialysis-related clinical testing services to independent dialysis clinics and broaden its chronic kidney disease testing leadership.
-
In February 2024, Simple HealthKit launched kidney test program to study kidney function at home and at a medical center. The Kidney Health Evaluation for Patients with Diabetes initiative focuses on a new kidney health test aimed at helping insurers enhance patient outcomes. This initiative can contribute to improved performance in key metrics, such as Healthcare Effectiveness Data and Information Set and Medicare Advantage star ratings.
Kidney Function Tests Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.18 billion
Revenue forecast in 2033
USD 1.77 billion
Growth rate
CAGR of 5.87% from 2026 to 2033
Base year for estimation
2025
Historical data
2018 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Spain; Italy; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; Kuwait; UAE; South Africa
Key companies profiled
Abbott; Danaher; F. Hoffmann-La Roche Ltd.; Sysmex Corporation; Siemens Healthineers; Randox Laboratories Ltd; Quest Diagnostics; ACON Laboratories, Inc.; Nova Biomedical Corporation; Laboratory Corporation of America Holdings
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Kidney Function Tests Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the global kidney function tests on the basis of product, end use, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Clearance Tests
-
Urine Tests
-
Blood Tests
-
Dilution And Concentration Tests
-
Other Tests
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Diagnostic Laboratories
-
Research Laboratories and Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global kidney function tests market size was estimated at USD 1.12 billion in 2025 and is expected to reach USD 1.18 billion in 2026.
b. The global kidney function tests market is expected to grow at a compound annual growth rate of 5.87% from 2026 to 2033 to reach USD 1.77 billion by 20333.
b. Some key players operating in the kidney function tests market include Abbott, Danaher, F. Hoffmann-La Roche Ltd., Sysmex Corporation, Siemens Healthcare GmbH, Randox Laboratories, Quest Diagnostics, and Laboratory Corporation of America Holdings.
b. Key factors that are driving the kidney function tests market growth include the growing prevalence of chronic kidney diseases and technological advancements in renal function tests.
b. The clearance tests segment dominated the market for kidney function tests and accounted for the largest revenue share of 27.53% in 2025.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









