- Home
- »
- Pharmaceuticals
- »
-
High Potency Active Pharmaceutical Ingredients Market Report 2033GVR Report cover
![High Potency Active Pharmaceutical Ingredients Market Size, Share & Trends Report]()
High Potency Active Pharmaceutical Ingredients Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Synthetic, Biotech), By Manufacturer Type (In-house, Outsourced), By Drug Type (Innovative, Generic), By Application, By Region, And Segment Forecasts
- Report ID: 978-1-68038-563-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
HPAPI Market Summary
The global high potency active pharmaceutical ingredients (HPAPI) market size was estimated at USD 29.87 billion in 2025 and is projected to reach USD 48.29 billion by 2033, growing at a CAGR of 6.2% from 2026 to 2033. This growth is primarily driven by the rising prevalence of cancer and other chronic diseases, increasing adoption of targeted therapies and oncology drugs, and the expanding pipeline of highly potent molecules such as antibody drug conjugates and cytotoxic compounds.
Key Market Trends & Insights
- North America high potency active pharmaceutical ingredients market held the largest share of 38.08% of the global market in 2025.
- The high potency active pharmaceutical ingredients industry in the U.S. is expected to grow significantly over the forecast period.
- By product, the synthetic segment held the highest market share of 72.97% in 2025.
- By manufacturer type, the in-house segment held the highest market share in 2025.
- By drug type, the innovative segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 29.87 Billion
- 2033 Projected Market Size: USD 48.29 Billion
- CAGR (2026-2033): 6.2%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, growing outsourcing of HPAPI manufacturing to specialized CDMOs, continuous investments in high-containment manufacturing facilities, and stringent regulatory requirements that favor experienced players are further supporting market expansion.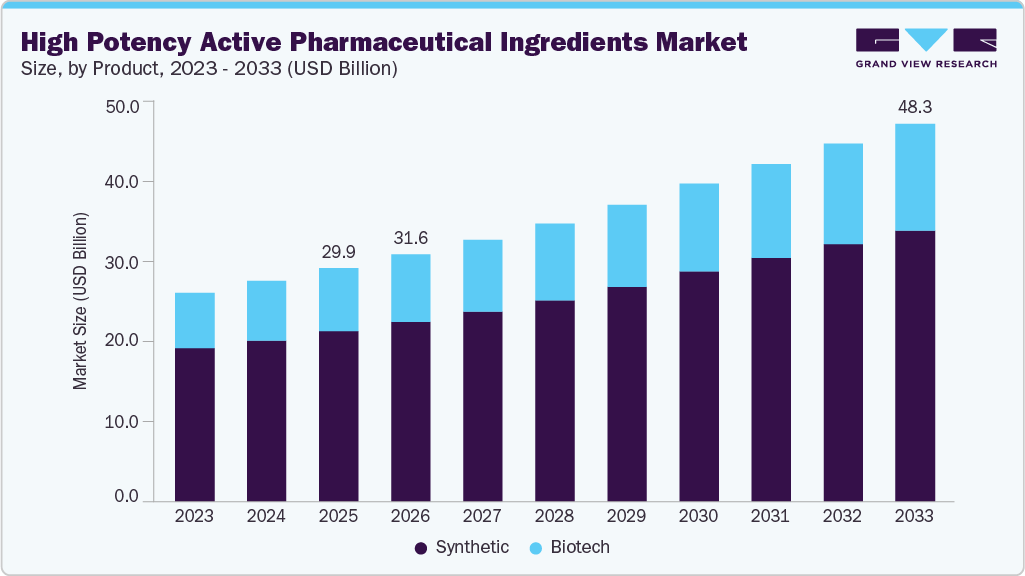
The rising incidence of cancer and other complex chronic conditions was a central factor driving the high potency active pharmaceutical ingredients market. Modern oncology treatment increasingly depended on highly potent molecules that achieved therapeutic effects at extremely low concentrations, supporting targeted drug action and improved patient outcomes. Pharmaceutical research pipelines continued to prioritize potent molecules to address unmet medical needs across multiple cancer types. HPAPIs were essential for reducing dosage volumes while maintaining clinical effectiveness, aligning with the growing emphasis on advanced therapies requiring precise biological interaction. For instance, January 2025, Polpharma API reported that global cancer cases were expected to increase by 61 percent by 2050, and that oncology accounted for 25 newly launched active substances in 2023, reinforcing sustained demand for highly potent active pharmaceutical ingredients across clinical development and commercial production.
The growing adoption of targeted therapies and advanced drug modalities significantly contributed to HPAPI market growth. Drug development strategies increasingly focused on precision medicine, which relied on potent compounds with specific mechanisms of action. Antibody drug conjugates, cytotoxic agents, and hormonal therapies depended on high potency ingredients to deliver controlled therapeutic effects while improving dosing efficiency and treatment consistency. Pharmaceutical innovators favored HPAPIs to enhance efficacy while managing side effects, accelerating the shift toward higher potency molecules in drug pipelines. For instance, November 2025, DCAT Value Chain Insights reported that approximately 70 percent of ADC-focused contract service providers offered manufacturing services for antibodies and or HPAPI cytotoxic payloads, and that HPAPI payloads represented about 20 percent of the ADC contract manufacturing value chain, underscoring the increasing role of potent ingredients in advanced therapeutic development.
The increasing dependence on contract development and manufacturing organizations was another major driver of market expansion. HPAPI manufacturing involved complex processes requiring advanced containment systems and specialized technical expertise, leading many pharmaceutical companies to outsource production to manage safety and operational complexity. CDMOs continued to invest in high containment infrastructure to improve scalability and accelerate development timelines while offering integrated synthesis, formulation, and commercial supply services. For instance, December 2025, Pharmaceutical Technology reported that multiple CDMOs expanded HPAPI capabilities through targeted investments, including a USD 90 million expansion focused on HPAPI and ADC manufacturing, the launch of new CGMP analytical testing laboratories in early 2025, and additional HPAPI capacity upgrades announced between January 2025 and May 2024, highlighting sustained infrastructure investment driven by growing outsourcing demand.
Market Concentration & Characteristics
The high potency active pharmaceutical ingredients market demonstrates a high degree of innovation driven by complex drug development needs. Manufacturers focus on advanced synthesis methods, containment technologies, and process optimization. Continuous innovation improves safety, yield, and scalability of highly potent compounds. Companies invest in novel conjugation techniques and specialized formulations to support targeted therapies. Process automation and digital monitoring enhance operational efficiency and quality control. Innovation remains a core competitive factor across the value chain.
Barriers to market entry are significantly high in the HPAPI market. Manufacturing requires substantial capital investment in high containment facilities and specialized equipment. Skilled workforce availability is limited and requires extensive training. Strict safety protocols increase operational complexity and cost. Long qualification timelines and customer audits further restrict new entrants. Established players benefit from scale, experience, and long-term client relationships.
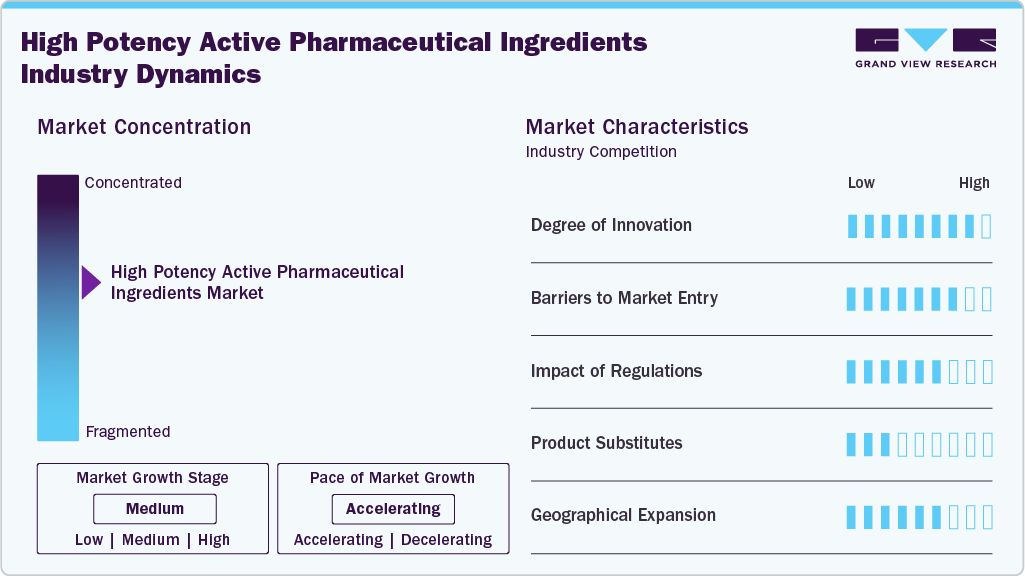
Regulatory oversight strongly influences market structure and operations. Manufacturers must comply with stringent safety, quality, and occupational exposure standards. Regulatory compliance demands extensive documentation, validation, and monitoring systems. Non compliance risks include production shutdowns and reputational damage. Regulatory expectations increase development timelines and manufacturing costs. These requirements favor experienced manufacturers with proven compliance records.
The threat of product substitutes remains relatively low in this market. High potency active ingredients are essential for specific therapeutic applications where alternatives are limited. Conventional APIs cannot easily replace HPAPIs in targeted and oncology treatments. Therapeutic performance depends on potency and precision of the active ingredient. Switching to substitutes may compromise efficacy and safety. This limited substitutability supports stable long-term demand.
Geographical expansion plays a key role in market growth strategies. Companies expand production facilities to diversify supply chains and serve regional demand. North America and Europe remain core manufacturing hubs with advanced capabilities. Asia Pacific attracts investment due to cost efficiency and expanding pharmaceutical activity. Regional expansion improves market access and capacity utilization. Global presence strengthens competitiveness and customer reach.
Product Insights
The synthetic segment dominated the market with the largest revenue share of 72.97% in 2025, driven by extensive use across oncology, hormonal, and central nervous system therapies. Synthetic high potency active pharmaceutical ingredients were widely adopted due to established manufacturing processes, strong reproducibility, and precise control over molecular structure, purity, and potency. Manufacturers favored synthetic HPAPIs for scalability, large-scale commercial production, and compatibility with advanced formulations. Continuous improvements in chemical synthesis and process optimization enhanced efficiency and cost control. For instance, December 2025, Pharmaceutical Technology reported that occupational exposure limits in synthetic HPAPI development were often below 1 microgram per cubic meter, requiring containment efficiencies above 99.99 percent. The article highlighted USD 90 million invested by Piramal Pharma Solutions in June 2025, USD 3 million by Sterling Pharma Solutions in May 2024, and facility expansions by SK pharmteco and AGC Pharma Chemicals, reinforcing the segment’s dominance.
The biotech segment is projected to grow at the fastest CAGR of 6.8% over the forecast period, due to the rapid expansion of biologics-based therapies and advanced drug modalities. Increasing development of antibody drug conjugates and highly targeted biologic treatments drives demand for potent biotech derived active ingredients. These products offer high specificity and improved therapeutic outcomes in complex disease areas. Growing focus on precision medicine supports wider adoption of biotech HPAPIs. Advances in bioprocessing technologies improve scalability and product consistency. Expanding clinical pipelines and commercial launches further accelerate growth of the biotech segment within the global market.
Manufacturer Type Insights
The in-house segment dominated the market with the largest revenue share of 72.06% in 2025, driven by large pharmaceutical companies’ preference to retain control over highly potent compound production. In-house manufacturing ensures greater oversight of quality, safety, and intellectual property for critical drug candidates, with established players investing heavily in dedicated high-containment facilities to support internal pipelines. This approach enables tighter integration between research, development, and commercial production while reducing dependency on external suppliers. For instance, in March 2023, Pfizer bought Seagen Inc. for about USD 43 billion, paying USD 229 per share, strengthening its in-house capabilities in developing high potency active pharmaceutical ingredients, particularly antibody-drug conjugates, and expanding its oncology portfolio. The deal, financed largely through USD 31 billion in long-term debt, added Seagen’s four marketed therapies and boosted Pfizer’s cancer drug revenue base of about USD 12 billion.
The outsourced segment is projected to grow at the fastest CAGR of 6.9% over the forecast period, supported by increasing reliance on specialized contract development and manufacturing organizations. HPAPI production requires advanced containment systems, skilled technical teams, and complex safety protocols, which many pharmaceutical companies prefer to access through outsourcing. Contract manufacturers offer flexible capacity and faster scale up for clinical and commercial needs. Growing drug development pipelines increase demand for external manufacturing support. Expansion of high containment facilities by CDMOs improves service availability. These factors collectively accelerate growth of the outsourced segment in the high potency active pharmaceutical ingredients market.
Drug Type Insights
The innovative segment dominated the market with the largest revenue share of 72.06% in 2025, due to the strong focus on novel drug development and high value therapeutic pipelines. Innovative HPAPIs are widely used in targeted therapies and oncology treatments that require precise and potent active ingredients. Pharmaceutical companies prioritize proprietary compounds to achieve clinical differentiation and improved efficacy. High investment in research and development supports continuous introduction of new molecules. These drugs often progress through advanced clinical stages and commercialization. This sustained innovation drives higher revenue contribution from the innovative segment in the global market.
The generic segment is projected to grow at the fastest CAGR of 7.9% over the forecast period. This growth is primarily driven by the increasing expiration of patents for high value drugs and the rising demand for cost effective treatment options. Generic manufacturers are expanding capabilities to produce highly potent compounds with consistent quality and safety. Growing adoption of generics in oncology and chronic disease management supports segment expansion. Improvements in manufacturing efficiency enable scalable production of potent molecules. Increasing participation of emerging market manufacturers further strengthens growth prospects for the generic segment.
Application Insights
The oncology segment held the largest revenue share in the high potency active pharmaceutical ingredients market in 2025, accounting for 76.53% of the total, supported by the high reliance on potent compounds in cancer treatment. Oncology therapies often require highly potent molecules to achieve targeted action at low doses. Increasing development of cytotoxic drugs and antibody‑drug conjugates drives sustained demand. For instance, in October 2025, Clinical Trials Arena noted oncology as the largest therapeutic use case for high potency active pharmaceutical ingredients, with cancer therapies such as antibody‑drug conjugates demanding advanced manufacturing solutions that address stringent containment and analytical control challenges, requiring robust processes to manage narrow therapeutic windows, control operator exposure, and ensure rigorous process validation amid global supply chain uncertainties. Pharmaceutical pipelines remain heavily focused on cancer indications across multiple stages, and high clinical success rates of targeted oncology drugs reinforce commercial adoption. These factors position oncology as the leading application segment in the global HPAPI market.
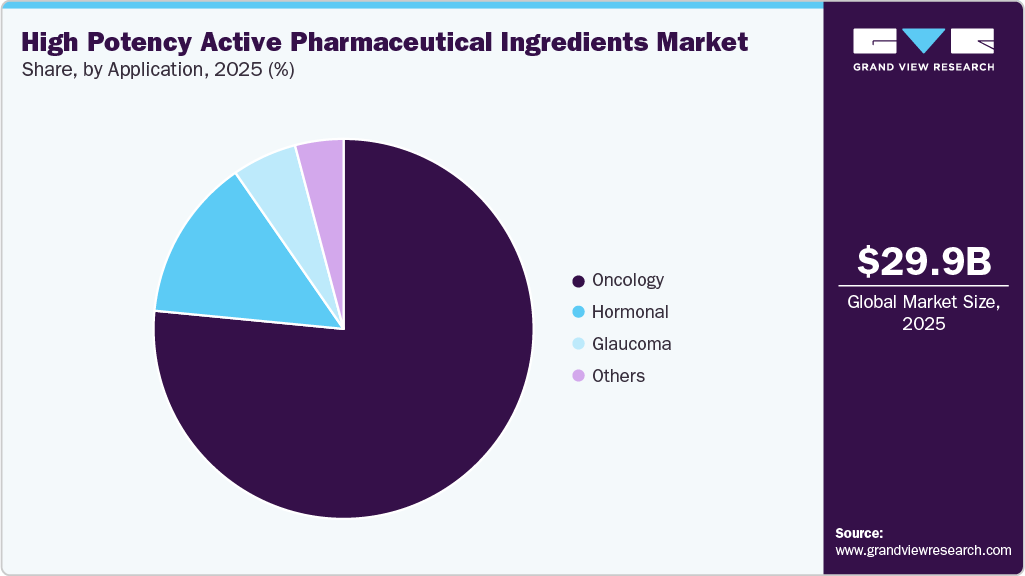
The glaucoma segment is projected to grow at the second fastest CAGR of 6.3% over the forecast period, driven by the rising prevalence of chronic eye disorders and the need for long‑term disease management. Treatments rely on highly potent compounds that deliver therapeutic effects at very low doses, while advances in ophthalmic drug formulations enhance delivery efficiency and patient adherence. Growing emphasis on precision dosing further supports the use of high‑potency ingredients. For instance, December 2025, Keensight Capital announced that Inke had acquired Pharmanoid, a specialized manufacturing site for ophthalmic highly potent active pharmaceutical ingredients, broadening its portfolio with prostaglandins used as first‑line treatment for glaucoma, the second leading cause of irreversible blindness globally, authorized in over 25 countries and produced at Pharmanoid’s 1,200 sqm EU GMP and ANVISA‑certified plant near Barcelona. Expansion of clinical research continues to support pipeline growth and steady segment expansion in the high‑potency API market.
Regional Insights
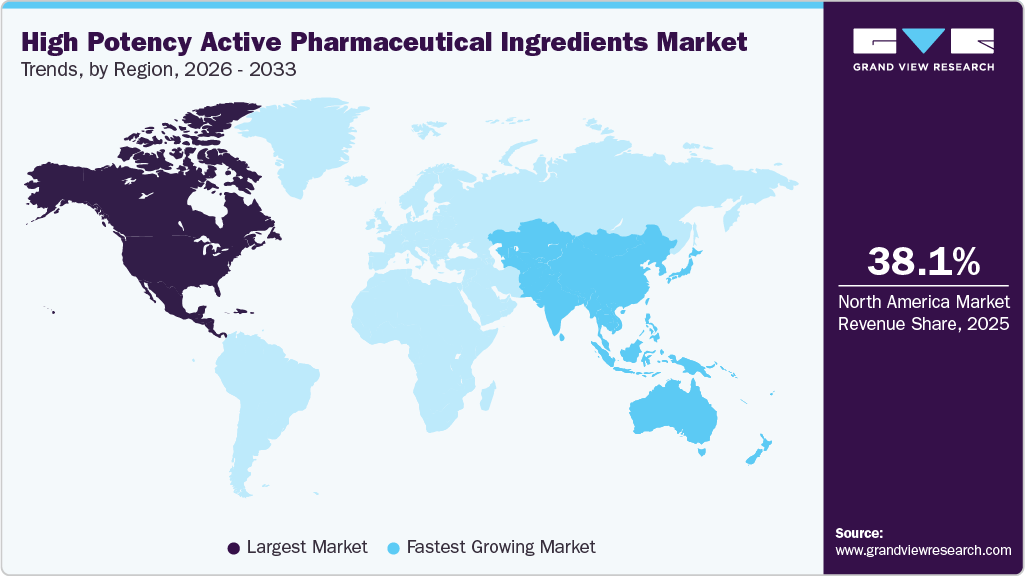
North America held the largest share of the high potency active pharmaceutical ingredients market in 2025, accounting for 38.08% of global revenue, due to its advanced pharmaceutical ecosystem. The region hosts a large concentration of innovator drug manufacturers and specialized HPAPI producers. Strong presence of oncology focused research pipelines drives sustained demand for potent compounds. Mature contract manufacturing networks support complex and large scale production needs. High adoption of targeted therapies increases reliance on low dose, high efficacy ingredients. Continuous capacity expansion strengthens the region’s leadership position.
U.S. High Potency Active Pharmaceutical Ingredients Market Trends
The U.S. represents the largest share within the North American HPAPI market. Extensive oncology drug development activity supports consistent demand for highly potent compounds. Pharmaceutical companies focus on precision therapies that require advanced HPAPI capabilities. Strong outsourcing trends benefit domestic CDMOs with high containment expertise. High investment in late stage clinical programs accelerates commercial scale production. The country remains a central hub for innovation driven HPAPI manufacturing.
Europe High Potency Active Pharmaceutical Ingredients Market Trends
Europe holds a significant share of the global HPAPI market due to its strong pharmaceutical manufacturing base. The region supports a wide network of specialty API producers and CDMOs. Increasing focus on targeted oncology and hormonal therapies fuels demand for potent ingredients. High quality manufacturing standards encourage long term client partnerships. Expansion of high containment facilities supports regional supply needs. Europe continues to attract global pharmaceutical collaborations.
The UK plays an important role in the European HPAPI market. Strong research driven pharmaceutical activity supports demand for potent molecules. The country specializes in early stage development and clinical scale manufacturing. Increasing collaboration between innovators and CDMOs strengthens production pipelines. Expertise in complex synthesis enhances market competitiveness. The UK remains attractive for high value, small volume HPAPI projects.
Germany stands as a key manufacturing hub within the HPAPI market. The country benefits from strong chemical engineering expertise and advanced infrastructure. High focus on quality and process efficiency supports consistent production output. Germany serves both domestic and export oriented pharmaceutical supply chains. Investment in containment technologies enhances manufacturing capabilities. The country maintains a strong position in commercial scale HPAPI production.
France contributes steadily to the HPAPI market through its established pharmaceutical sector. The country supports both innovative drug developers and contract manufacturers. Focus on oncology and endocrine therapies drives HPAPI utilization. Strong formulation and API integration capabilities enhance value chain efficiency. French manufacturers emphasize high quality and operational reliability. These factors support stable market participation.
Asia Pacific High Potency Active Pharmaceutical Ingredients Market Trends
Asia Pacific high potency active pharmaceutical ingredients market is expected to register the significant CAGR of 7.5% over the forecast period. Rapid expansion of pharmaceutical manufacturing capacity drives regional growth. Increasing participation in global supply chains boosts HPAPI demand. Cost efficient production attracts outsourcing from global drug developers. Rising clinical development activity accelerates local manufacturing needs. The region continues to strengthen its technical and containment capabilities.
Japan maintains a strong position in the HPAPI market through advanced pharmaceutical innovation. The country focuses on high quality, precision driven drug development. Growing demand for oncology and specialty therapies supports HPAPI usage. Japanese manufacturers emphasize process control and safety standards. Integration of API and formulation capabilities enhances efficiency. Stable domestic demand supports long term market growth.
China plays a growing role in the global HPAPI market. Rapid expansion of pharmaceutical manufacturing infrastructure supports increased production capacity. Rising investment in innovative drug development drives demand for potent compounds. Contract manufacturing activity continues to expand across multiple therapeutic areas. Improved technical expertise enhances competitiveness in complex API synthesis. China strengthens its position as a key global supplier.
Latin America High Potency Active Pharmaceutical Ingredients Market Trends
Latin America represents an emerging market for HPAPIs. Growing pharmaceutical production activity supports gradual demand growth. Regional manufacturers focus on expanding capabilities for specialized APIs. Increasing participation in generic and specialty drug production drives utilization. Strategic partnerships enhance access to advanced manufacturing knowledge. The region shows steady development potential.
Brazil leads the HPAPI market within Latin America. Expanding pharmaceutical manufacturing supports local demand for potent ingredients. Focus on oncology and chronic disease treatments increases HPAPI relevance. Domestic producers invest in upgrading production capabilities. Contract manufacturing activity continues to develop across the country. Brazil serves as a regional manufacturing center.
Middle East & Africa High Potency Active Pharmaceutical Ingredients Market Trends
The MEA region shows gradual development in the HPAPI market. Increasing pharmaceutical production activity supports limited but growing demand. Regional players focus on importing and secondary processing of APIs. Expansion of healthcare manufacturing drives interest in specialized ingredients. Infrastructure development improves long term market prospects. Growth remains moderate compared to developed regions.
Saudi Arabia represents a developing market within the MEA region. Expansion of pharmaceutical manufacturing facilities supports HPAPI demand growth. Focus on local drug production increases reliance on advanced APIs. Strategic collaborations enhance technical knowledge transfer. Emphasis on high value medicines supports potency driven formulations. The country shows increasing participation in regional supply chains.
Key High Potency Active Pharmaceutical Ingredients Company Insights
BASF SE and CordenPharma hold strong positions in the high potency active pharmaceutical ingredients market through advanced chemical synthesis expertise and dedicated high containment manufacturing capabilities. Bristol-Myers Squibb Company and Pfizer, Inc. maintain significant market shares supported by extensive oncology pipelines and long-standing experience in highly potent small molecule development. CARBOGEN AMCIS AG strengthens its presence through custom HPAPI synthesis and integrated development services. Boehringer Ingelheim International GmbH leverages in-house manufacturing expertise and a diversified high potency portfolio. Dr. Reddy’s Laboratories Ltd., Sun Pharmaceutical Industries, Ltd., Albany Molecular Research, Inc., Sanofi, and Cipla Inc. support market growth through expanding HPAPI capacity, contract manufacturing services, and increasing focus on potent and targeted therapies.
Key High Potency Active Pharmaceutical Ingredients Companies:
The following key companies have been profiled for this study on the high potency active pharmaceutical ingredients market.
- BASF SE
- CordenPharma
- Bristol-Myers Squibb Company
- CARBOGEN AMCIS AG
- Pfizer, Inc.
- Boehringer Ingelheim International GmbH
- Dr. Reddy’s Laboratories Ltd.
- Sun Pharmaceutical Industries, Ltd.
- Albany Molecular Research, Inc.
- Sanofi
- Cipla Inc.
Recent Developments
-
In June 2025, BASF stated that it had strengthened its commitment to the biopharma and pharmaceutical ingredients industries by opening a new Good Manufacturing Practice Solution Center in Wyandotte, Michigan. The company said the investment had expanded its pharmaceutical excipients and biopharma capabilities through advanced clean room packaging, high sensitivity analytical testing, and enhanced technical collaboration to support customized formulations and higher quality standards.
-
In November 2023, Bristol Myers Squibb had acquired its ORM-6151 program with an upfront payment of USD 100 million, and that Orum was eligible for additional milestone payments bringing the total deal value to USD 180 million, with ORM-6151 described as a first-in-class, anti-CD33 antibody-enabled GSPT1 degrader that had received FDA clearance for Phase 1 studies in patients with acute myeloid leukemia or high-risk myelodysplastic syndromes.
-
In July 2024, Pfizer had invested USD 1 billion to open a new pharmaceutical ingredient plant in Singapore’s Tuas Biomedical Park, which spanned 429,000 sq ft and was designed for highly automated production of active pharmaceutical ingredients, and that the project had created 250 new jobs to support global manufacturing of cancer, pain, and antibiotic medicines.
High Potency Active Pharmaceutical Ingredients Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 31.62 billion
Revenue forecast in 2033
USD 48.29 billion
Growth rate
CAGR of 6.2% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, manufacturer type, drug type, application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
BASF SE; CordenPharma; Bristol-Myers Squibb Company; CARBOGEN AMCIS AG; Pfizer, Inc.; Boehringer Ingelheim International GmbH; Dr. Reddy’s Laboratories Ltd.; Sun Pharmaceutical Industries, Ltd.; Albany Molecular Research, Inc.; Sanofi; Cipla Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global High Potency Active Pharmaceutical Ingredients Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global high potency active pharmaceutical ingredients market report based on product, manufacturer type, drug type, application, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Synthetic
-
Biotech
-
-
Manufacturer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
In-house
-
Outsourced
-
-
Drug Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Innovative
-
Generic
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Hormonal
-
Glaucoma
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global high potency active pharmaceutical ingredients market size was estimated at USD 25.17 billion in 2022 and is expected to reach USD 26.72 billion in 2023.
b. The global high potency active pharmaceutical ingredients market is expected to grow at a compound annual growth rate of 6.2% from 2023 to 2030 to reach USD 40.66 billion by 2030.
Which segment accounted for the largest high potency active pharmaceutical ingredients market share?b. Synthetic molecules dominated the HPAPIs market with a share of 72.3% in 2022 owing to the easier protocols for the synthesis of these molecules coupled with the higher availability of raw material.
b. The key companies of high potency APIs industry include Lonza, BASF SE; CordenPharma; Dr. Reddy’s Laboratories Ltd.; CARBOGEN AMCIS AG; Pfizer, Inc.; Sun Pharmaceutical Industries, Ltd.; Teva Pharmaceutical Industries Ltd.; Albany Molecular Research, Inc.; Sanofi S.A.; Merck & Co., Inc.; Novartis AG; F. Hoffmann-La Roche Ltd.; Bristol-Myers Squibb Company; Boehringer Ingelheim International GmbH; Lonza; Cipla Inc.
b. Key factors that are driving the high potency APIs market growth include a rise in prevalence of cancer and the global anti-cancer market, growth of targeted therapy approaches, and increasing application of high potency active pharmaceutical ingredients.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









