- Home
- »
- Medical Devices
- »
-
Flow Diverters Market Size & Share, Industry Report, 2033GVR Report cover
![Flow Diverters Market Size, Share & Trends Report]()
Flow Diverters Market (2026 - 2033) Size, Share & Trends Analysis Report By Diameter Size (2-3 mm, 3-4 mm, 4-5 mm, >5 mm), By Region (North America, Europe, Asia Pacific, Middle East & Africa, Latin America), And Segment Forecasts
- Report ID: GVR-4-68039-184-2
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Flow Diverters Market Summary
The global flow diverters market size was estimated at USD 385.60 million in 2025 and is projected to reach USD 988.81 million by 2033, growing at a CAGR of 12.43% from 2026 to 2033. The market is driven by the increasing incidence of intracranial aneurysms, which is raising the demand for advanced endovascular treatment options.
Key Market Trends & Insights
- The North America led the market with the largest revenue share in 2025.
- By diameter size, the >5mm size segment led the market with the largest revenue share in 2025.
- By diameter size, the 2-3 mm segment is expected to grow at the fastest rate during the forecast period
Market Size & Forecast
- 2025 Market Size: USD 385.60 million
- 2033 Projected Market Size: USD 988.81 million
- CAGR (2026-2033): 12.43%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, ongoing mergers and acquisitions among medical device companies are strengthening product portfolios and accelerating technological innovation in flow diversion therapies. The rising incidence of intracranial aneurysms is a key driver of demand for flow-diverter devices. The increasing prevalence of risk factors such as hypertension, smoking, and aging populations has led to a higher number of aneurysm diagnoses globally.
As flow diverters provide an effective treatment option for complex and wide-neck aneurysms, their adoption is increasing among neurointerventional specialists. For instance, according to studies cited by the Radiological Society of North America (RSNA) in 2025, intracranial aneurysms affect an estimated 3.2% of the global population. A significant proportion of cases remain undetected until rupture, with nearly 50% of aneurysms being identified only after rupture, which is associated with high morbidity and mortality rates.
Brain Aneurysm Statistics (2025)
Category
Statistic
Prevalence in U.S.
Approximately 6.8 million people in the United States have an unruptured brain aneurysm (about 1 in 50 people)
Annual rupture rate
Around 8-10 per 100,000 people experience aneurysm rupture each year
Annual rupture cases (U.S.)
About 30,000 people suffer a brain aneurysm rupture annually
Frequency of rupture
A brain aneurysm ruptures every 18 minutes in the United States
Global mortality
Nearly 500,000 deaths worldwide each year are caused by brain aneurysms
Stroke contribution
Ruptured aneurysms account for 3-5% of all new stroke cases
Source: Brain Aneurysm Foundation
Surgical clipping, flow diverters, and endovascular coiling are notable modalities for treating intracranial aneurysms. Some key players operating in this market are Stryker Corporation, Medtronic, MicroPort Corporation, and Johnson & Johnson. Key players constantly try to introduce technologically advanced flow diverters to gain a competitive advantage. The creation of flow diverters that are more durable and more efficient at controlling blood flow within blood vessels results from the integration of innovative materials and manufacturing processes.
Furthermore, advancements in imaging technologies have improved the placement of flow diverters with greater precision and accuracy, thereby reducing the risk of complications during treatment. For instance, in July 2025, Stryker announced the commercial launch of the Surpass Elite Flow Diverter in the U.S., South Korea, and Europe. The next-generation device features an upgraded implant design and proprietary surface modification technology to improve device performance and reduce thrombin generation during aneurysm treatment.
“Surpass Elite Flow Diverter captures the essence of a cobalt chromium device, delivering outstanding performance and exceptional ease of use,” said Dr. Alejandro Tomasello, Vall d'Hebron University Hospital, Barcelona, Spain.
“I have been fortunate to be part of the development of the Surpass Elite Flow Diverter, Stryker’s most sophisticated flow-diverting stent to date,” said Dr. Alex Coon, Carondelet Neurological Institute, Tucson, Arizona. “My early experience suggests that this device gives physicians the exact blend of device opening, vessel-wall apposition, and surface treatment for which we have all been waiting a decade.”
“This cadenced launch strategy reflects our commitment to clinical excellence,” said Jim Marucci, president of Stryker’s Neurovascular division. “After carefully validating performance across a diverse set of early cases, we’re pleased to move to this next step and help address unmet customer needs.”
Furthermore, in July 2023, MicroVention, Inc., collaborated with U.S. physicians, and over 1,000 patients were effectively treated using the FRED X flow diverter. Specifically designed for redirecting blood flow in small vessels, FRED X is the only FDA PMA-approved option featuring a 0.021" delivery system for smaller sizes and without a distal lead wire. Enhanced with X Tech, the FRED device maintains precise and predictable placement characteristics. Such initiatives boost market growth in the near future.
Moreover, acquisitions and mergers among industry key players are anticipated to propel market growth over the forecast period. For instance, in February 2025, Stryker announced the completion of its acquisition of Inari Medical, Inc., a company known for developing innovative solutions for the removal of venous thromboembolism (VTE) clots without the use of thrombolytic drugs. The acquisition strengthens Stryker’s presence in the peripheral vascular space and expands its portfolio within the rapidly growing VTE treatment segment.
“The acquisition of Inari Medical marks a significant milestone in expanding our interventional endovascular portfolio,” said Kevin Lobo, Chair and Chief Executive Officer, Stryker. “We look forward to welcoming the talented Inari team to Stryker and working together to improve outcomes for patients worldwide.”
Moreover, in May 2023, Stryker acquired Cerus Endovascular Ltd., a company specializing in the development and production of medical devices for treating intracranial aneurysms. Cerus Endovascular's products, including the Neqstent coil-assisted flow diverter and Contour neurovascular system, which are CE Marked, will complement Stryker's current product line of treatments for brain aneurysms.
“The acquisition of Cerus Endovascular is highly complementary to Stryker’s Neurovascular business, and strengthens our hemorrhagic portfolio globally,” stated Jim Marucci, president of Stryker’s Neurovascular division. “We look forward to working with the Cerus team to deliver on our mission of making healthcare better. Together, we will enable physicians to treat a broader range of aneurysms.”
With mergers and acquisitions, the market’s key players integrate their assets, expertise, and technological abilities. The act of merger often leads to improved research and development capacity, making it easier to develop more advanced, effective flow diverters, thereby propelling market growth in the near future.
Market Concentration & Characteristics
The flow diverters market demonstrates a high degree of innovation driven by continuous advancements in device design, materials, and delivery systems. Manufacturers are focusing on improving mesh density, surface coatings, and flexibility to enhance aneurysm occlusion rates and reduce procedural complications. These innovations are enabling physicians to treat complex intracranial aneurysms more effectively. For instance, Stryker announced the commercial launch of the Surpass Elite Flow Diverter across the U.S., South Korea, and Europe. This next-generation device features an upgraded implant design and proprietary surface modification technology to enhance device performance and reduce thrombin generation during aneurysm treatment.
The industry is witnessing a growing level of merger and acquisition activities as medical device companies aim to strengthen their neurovascular treatment portfolios and expand technological capabilities. Through acquisitions and strategic partnerships, firms are gaining access to advanced aneurysm treatment technologies while enhancing their presence in the neurointerventional device segment. For instance, in January 2026, Zylox-Tonbridge announced a definitive agreement to acquire Optimed Medizinische Instrumente GmbH, a Germany-based company specializing in minimally invasive medical devices, with the option to acquire full equity through a multi-step transaction.
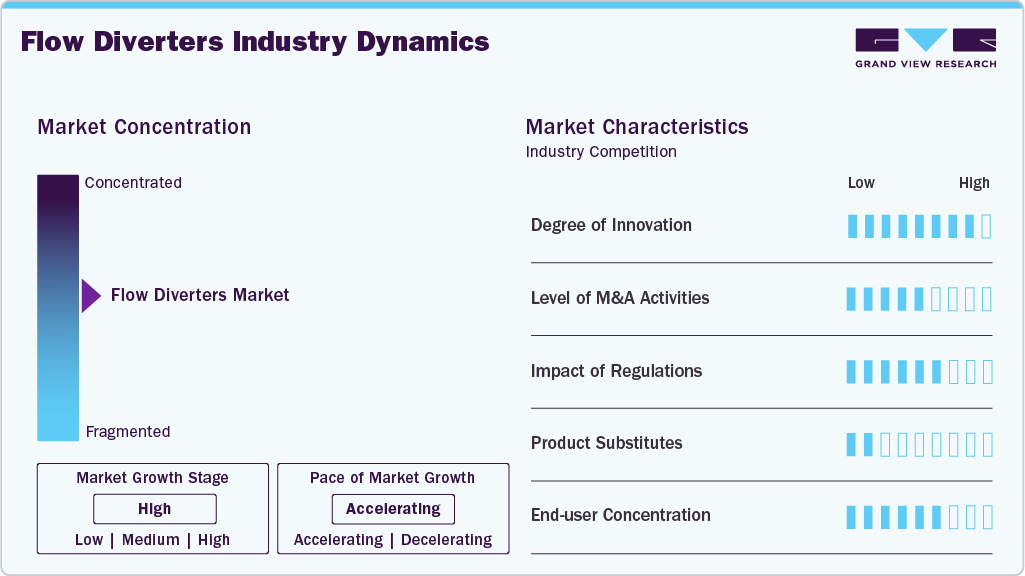
Regulatory frameworks play a significant role in shaping the flow diverters market, as these devices require rigorous clinical evaluation and regulatory approvals before commercialization. Strict approval processes ensure safety and effectiveness, but can also extend product development timelines and increase compliance costs for manufacturers. Regulatory clearances enable companies to expand their product availability across global markets.
The end-use concentration is primarily centered in hospitals and specialized neurovascular centers, where advanced imaging systems and skilled neurointerventional specialists are available. These facilities handle most intracranial aneurysm treatments because they can perform complex endovascular procedures. As a result, large tertiary care hospitals and specialized stroke centers account for a significant share of flow-diverter device use.
Diameter Size Insights
The >5 mm segment dominated the flow diverters market in 2025, primarily due to the higher risk of rupture associated with larger intracranial aneurysms, which often require advanced endovascular treatment. Flow diverters are widely used for aneurysms larger than 5 mm as they provide effective vessel reconstruction and long-term aneurysm occlusion. In addition, increasing diagnosis of medium-to-large aneurysms through advanced imaging techniques has further supported the growth of this segment.
The 2-3 mm segment is expected to grow at the fastest rate during the forecast period, driven by earlier detection of small intracranial aneurysms using advanced imaging technologies such as CT and MR angiography. Growing awareness among clinicians about preventive treatment and improved device designs suitable for small vessels is further supporting the adoption of flow diverters for smaller aneurysms.
Regional Insights
The flow diverters industry in North America accounted for 35.82% of the revenue share in the global market in 2025, owing to the presence of major manufacturers, increased spending on R&D, and an increase in government initiatives. Besides, a robust reimbursement network and strong government funding are expected to contribute to market growth. Moreover, demand for flow diverters may be positively affected by greater public awareness among patients and healthcare professionals of the importance of early detection and treatment for chronic illnesses.
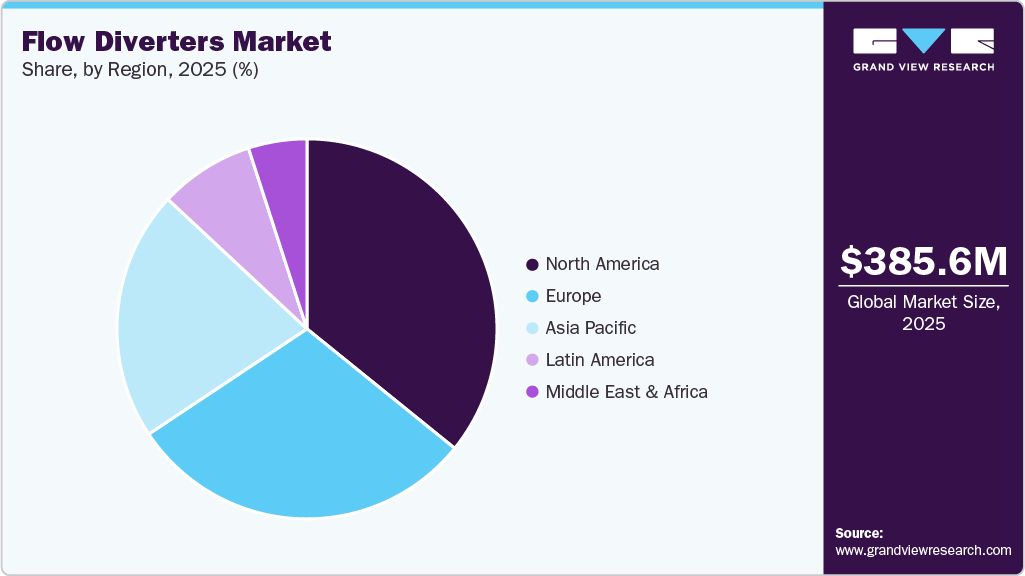
Increased awareness within the healthcare community and the patients regarding the importance of early diagnosis & treatment for chronic disorders may positively influence the demand for flow diverters. For instance, according to the Centers for Medicare & Medicaid Services (CMS), 7.5% of U.S. health expenditures were anticipated to be spent in 2022. It was expected to reach $1 trillion for the first time in 2023. Moreover, a substantial portion of the nation's economy was accounted for by skilled workers and high private-sector earnings in developed economies. As a result, the flow diverter market in North America is expected to grow due to improved healthcare facilities and developing infrastructure.
U.S. Flow Diverters Market Trends
The flow diverters industry in the U.S. is expected to grow over the forecast period. The main factors propelling the expansion are the rising rates of hospitalization, traffic accidents, and the prevalence of chronic illnesses. For instance, according to the annual report released by America's Health Rankings 2023, 133 million people, approximately 43% of the U.S. population, suffered from one or more chronic illnesses in 2022. Moreover, a significant driver of market expansion in this area is the growing elderly population.
Europe Flow Diverters Market Trends
The flow diverters industry in Europe is witnessing significant growth driven by increasing diagnoses of intracranial aneurysms and the rising use of minimally invasive neurovascular procedures. The presence of advanced healthcare infrastructure and specialized neurointerventional centers is supporting the adoption of flow diversion technologies. In addition, ongoing clinical research and early adoption of innovative neurovascular devices in several European countries are contributing to market expansion.
The UK flow diverters industry is witnessing gradual growth driven by increasing awareness and diagnosis of intracranial aneurysms. The availability of specialized neurointerventional centers and advanced imaging technologies is supporting the adoption of endovascular treatments. In addition, ongoing clinical studies and the introduction of newer-generation flow-diverter devices are contributing to the expansion of the market in the country.
Asia Pacific Flow Diverters Market Trends
The flow diverters industry in the Asia Pacific is experiencing notable growth driven by the rising burden of neurovascular disorders and improved access to specialized medical care. A rising number of road accidents in the region, which can lead to traumatic brain injuries and vascular complications, is also contributing to the demand for advanced neurovascular treatments. In addition, expanding healthcare infrastructure and the growing number of neurointerventional centers are supporting the adoption of flow diverter devices across the region. In addition, evolving healthcare infrastructure, supportive government initiatives, and increasing awareness regarding early diagnosis and treatment have driven the market in the Asia Pacific to grow at a substantial rate.
The China flow diverters industryis expected to grow over the forecast period owing to the need for advanced medical equipment and the increasing number of hospitals and healthcare centers in the country. In addition, the government's initiatives to improve healthcare infrastructure and provide better medical facilities to the citizens are also contributing to the growth in China.
Latin America Flow Diverters Market Trends
The flow diverters industry in Latin America is gradually expanding due to increasing awareness of neurovascular diseases and improved access to advanced endovascular treatments. Growing investments in healthcare infrastructure and the development of specialized neurology and stroke centers are driving the adoption of flow-diversion technologies. In addition, the rising availability of modern imaging systems is enabling earlier diagnosis and treatment of intracranial aneurysms in the region.
Middle East and Africa Flow Diverters Market Trends
The flow diverters industry in the Middle East and Africa is expected to witness significant growth in the coming years due to a rise in the prevalence of chronic illness, growing expenditure on healthcare infrastructure, and rising demand for cutting-edge medical technology. The market is anticipated to be propelled by the adoption of innovative, cutting-edge flow diverters that improve patient comfort and safety.
Key Flow Diverters Company Insights
The flow diverters market is characterized by a few major medical device companies holding significant shares, driven by strong product portfolios and global distribution networks. Key players include Medtronic, Stryker, and Terumo (through its subsidiary MicroVention), which dominate the competitive landscape through continuous product innovation and strategic collaborations. These companies maintain strong market positions by investing in research, clinical studies, and next-generation neurovascular devices.
Key Flow Diverters Companies:
The following key companies have been profiled for this study on the flow diverters market.
- Stryker
- Medtronic
- MicroVention, Inc. (Terumo Corporation)
- Balt (Bridgepoint Group plc)
- MicroPort Scientific Corporation
- CERENOVUS (Johnson & Johnson Services, Inc.)
- phenox GmbH (Wallaby Medical)
- Acandis GmbH
- InspireMD, Inc.
- ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD
- AccuMedical
Recent Developments
-
In May 2025, AccuMedical announced the global launch of its next-generation Lattice NEXT, a “tipless” mechanical balloon-assisted flow-diverter system, at the WLNC x OCIN2025 International Neurointervention Conference. The launch highlighted the company’s continued innovation in neurointerventional therapy, showcasing advancements in flow diverter design and access technologies for the treatment of complex intracranial aneurysms.
-
In February 2025, Stryker announced that it had completed the acquisition of Inari Medical, Inc., a company known for developing innovative devices to remove venous thromboembolism (VTE) clots without the use of thrombolytic drugs. The acquisition strengthens Stryker’s position in the peripheral vascular segment and expands its capabilities in the rapidly growing VTE treatment market.
-
In February 2025, AccuMedical announced that its Lattice Flow Diverter received the 2024 Patent-Intensive Product Recognition from the Patent Protection Association of China. The recognition highlights AccuMedical’s strong innovation capabilities in the medical device sector and reflects the advanced technology and distinctive design of the Lattice product.
-
In July 2025, Stryker announced the commercial launch of the Surpass Elite Flow Diverter across the United States, South Korea, and Europe. This next-generation device features an upgraded implant design and proprietary surface modification technology to improve performance and reduce thrombin generation during flow-diversion procedures.
-
In September 2024, MicroVention, Inc., a wholly owned subsidiary of Terumo Corporation, announced its official rebranding to Terumo Neuro. The rebranding reflects a new phase in the company’s development while continuing its focus on advancing innovation and commercialization of neurovascular treatment technologies.
Flow Diverters Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 435.43 million
Revenue forecast in 2033
USD 988.81 million
Growth rate
CAGR of 12.43% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Diameter size, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East and Africa
Country Scope
U.S.; Canada; Mexico; UK; Germany; Italy; France; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Stryker; Medtronic; MicroVention, Inc. (Terumo Corporation); Balt (Bridgepoint Group plc); MicroPort Scientific Corporation; CERENOVUS (Johnson & Johnson Services, Inc.); phenox GmbH (Wallaby Medical); Acandis GmbH; InspireMD, Inc.; ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD; AccuMedical
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
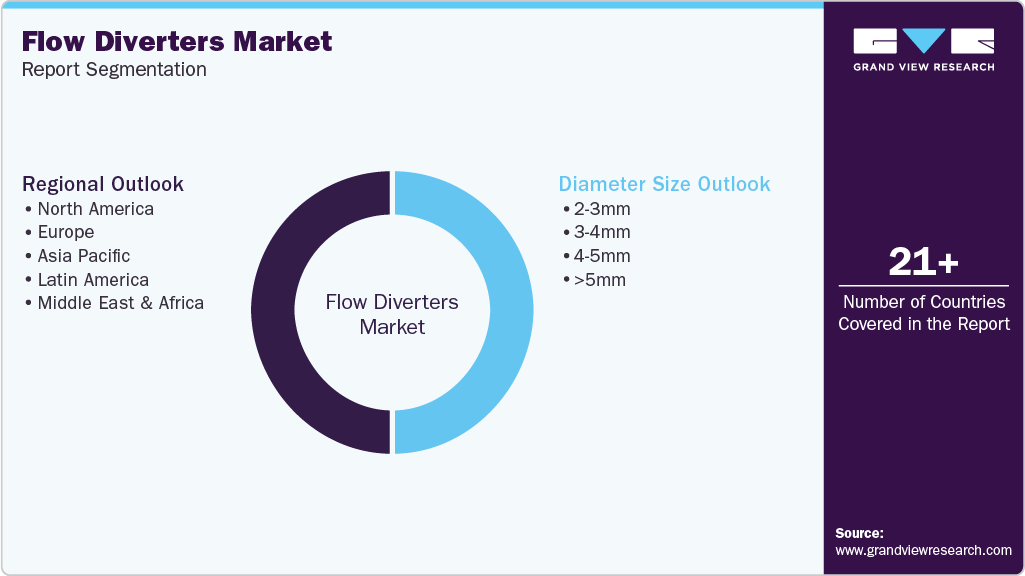
Global Flow Diverters Market Report Segmentation
This report forecasts revenue growth at the global, regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global flow diverters market report based on diameter size and region.
-
Diameter Size Outlook (Revenue, USD Million, 2021 - 2033)
-
2-3mm
-
3-4mm
-
4-5mm
-
>5mm
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global flow diverters market size was estimated at USD 303.01 million in 2023 and is expected to reach USD 341.70 million in 2024.
b. The global flow diverters market is expected to grow at a compound annual growth rate of 13.29% from 2024 to 2030 to reach USD 722.59 million by 2030.
b. North America dominated the flow diverters market with the highest share of 35.98% in 2023. This is attributable to rising healthcare awareness coupled with the introduction of technologically advanced products and constant research and development initiatives.
b. Some key players operating in the flow diverters market include Medtronic plc, Stryker Corporation, MicroVention, Inc. (Terumo Corporation), Balt, MicroPort, Scientific Corporation, phenox GmbH, Acandis GmbH, Cerus Endovascular Inc., CERENOVUS (Johnson & Johnson Services, Inc.), InspireMD, Inc., and others.
b. Key factors driving the flow diverters market growth include rising demand for minimally invasive surgeries; increasing government initiative, favorable government policies, and rising incidence of intracranial aneurysms.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









