- Home
- »
- Pharmaceuticals
- »
-
Dasatinib Market Size & Share, Industry Report, 2033GVR Report cover
![Dasatinib Market Size, Share & Trends Report]()
Dasatinib Market (2026 - 2033) Size, Share & Trends Analysis Report By Drug Type, (Branded, Generic), By Indication (Chronic Myeloid Leukemia (CML), Acute Lymphoblastic Leukemia (ALL)), By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-865-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Dasatinib Market Summary
The global dasatinib market size was estimated at USD 5.59 billion in 2025 and is projected to reach USD 7.57 billion by 2033, growing at a CAGR of 4.27% from 2026 to 2033. The global dasatinib market is expanding due to the rising prevalence of hematologic malignancies, particularly chronic myeloid leukemia and Philadelphia chromosome positive acute lymphoblastic leukemia.
Key Market Trends & Insights
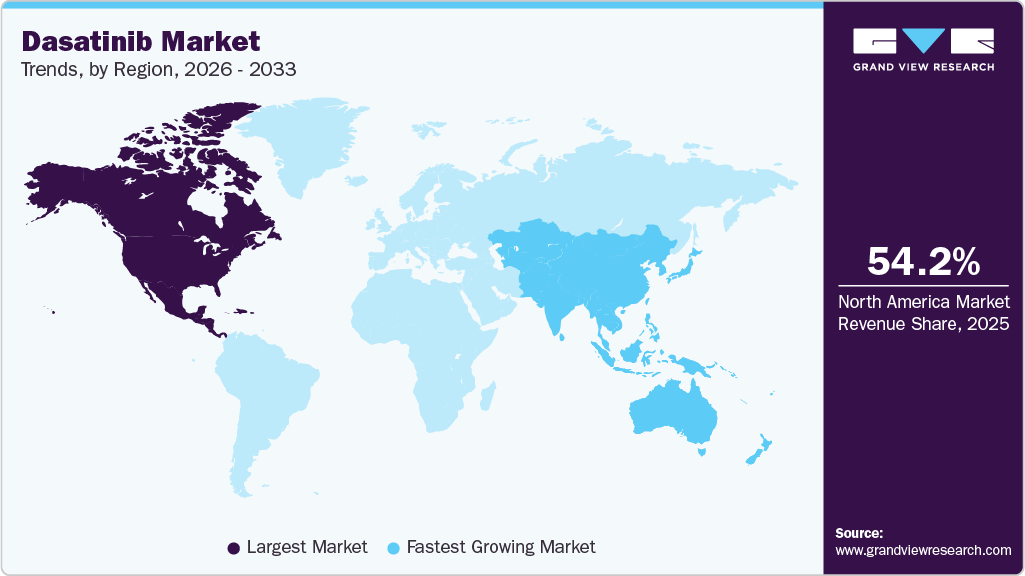
- North America dasatinib market held the largest share of 54.17% of the global market in 2025.
- The dasatinib industry in the U.S. is expected to grow significantly over the forecast period.
- By drug type, the generic segment held the highest market share of 69.24% in 2025.
- By indication, the Chronic Myeloid Leukemia (CML) segment held the highest market share in 2025.
- By distribution channel, the hospital pharmacies segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 5.59 Billion
- 2033 Projected Market Size: USD 7.57 Billion
- CAGR (2026-2033): 4.27%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Increasing awareness of targeted oncology therapies has improved diagnosis and early treatment initiation across developed and emerging regions. Physicians prefer tyrosine kinase inhibitors for sustained disease control, supporting long term demand. Longer survival has increased cumulative drug utilization, strengthening revenue visibility. For instance, November 2025, OncLive reported FDA approval of a generic dasatinib abbreviated new drug application in six strengths, 20 mg, 50 mg, 70 mg, 80 mg, 100 mg, and 140 mg. Initial clearance occurred in 2006, with expansion in January 2019 based on a phase 2 trial in 78 pediatric patients showing 64.1 percent 3 year event free survival, 95 percent confidence interval 52.4 percent to 74.7 percent, 96 percent below 5 percent marrow blasts at induction, 97 percent at consolidation, 4 percent fatal adverse effects among 81 patients, and 10 percent discontinuation.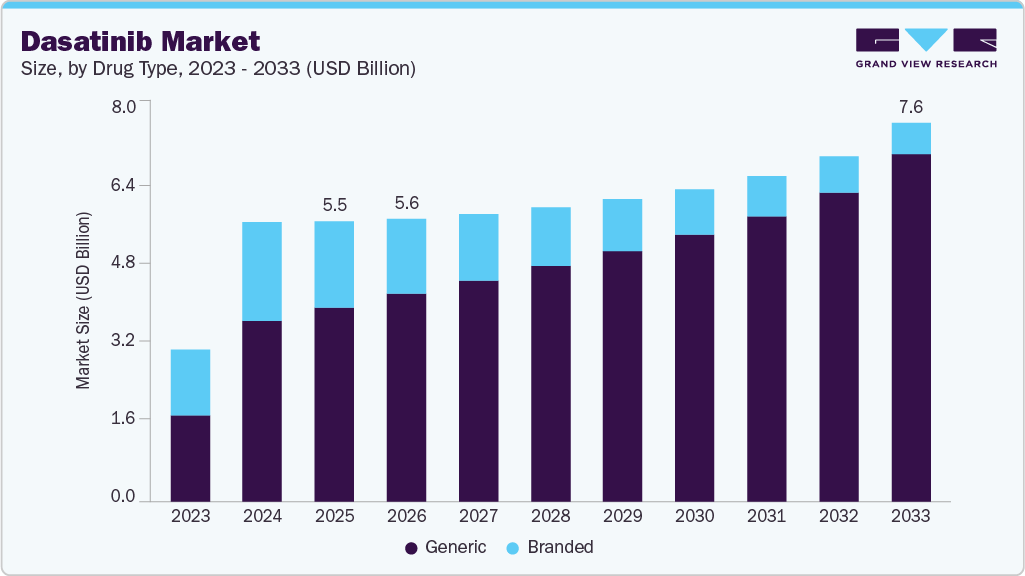
Advancements in precision medicine and molecular diagnostics have strengthened biomarker driven therapy selection, accelerating adoption of targeted treatments such as dasatinib. Clinical evidence in resistant or intolerant chronic myeloid leukemia has reinforced physician confidence and supported guideline recommendations for second generation tyrosine kinase inhibitors. For instance, January 2026, Cancer Reports published a retrospective analysis of 53 adults treated with low dose dasatinib 50 mg daily between 2002 and 2025, with a median age of 50 years. Molecular response outcomes included 41.5 percent achieving MR4.5, 20.8 percent MR4.0, and 15.1 percent major molecular response without deep response. Tyrosine kinase domain mutations were present in 32.1 percent, clinically significant adverse events in 49.1 percent, 22.6 percent required switching for inadequate response, and 7.5 percent for intolerance. Baseline BCR ABL1 greater than or equal to 100 percent, T315I mutation, and high ELTS risk predicted poorer outcomes.
Ongoing research into expanded indications and combination regimens is creating additional commercial opportunities beyond hematologic cancers. For instance, January 2025, Biomedical Reports published a meta analysis reviewing 55 trials through May 5, 2024 and including nine randomized studies, four phase II, three phase I, and two phase I or II, involving 234 patients, 107 on monotherapy and 127 on combination therapy. Among participants, 79.1 percent had non small cell lung cancer, with adenocarcinoma at 63.8 percent and squamous carcinoma at 22.1 percent. Progressive disease occurred in 52.4 percent of monotherapy versus 29.6 percent of combination therapy, while stable disease was 22.4 percent versus 38.3 percent. Adverse events included anemia in 42.2 percent, 50.6 percent with erlotinib versus 3.7 percent alone, diarrhea in 35.4 percent, and rash in 85.1 percent.
Expansion of generic manufacturing has improved affordability and accessibility across price sensitive markets, increasing patient reach and strengthening competitive dynamics. Broader distribution through hospital and specialty pharmacy networks has enhanced supply continuity and global penetration. Strategic collaborations between pharmaceutical companies and healthcare providers are supporting consistent product availability. Growth in oncology infrastructure across emerging economies is enabling wider adoption of advanced targeted therapies. Rising healthcare expenditure and expanding insurance coverage for cancer treatment are sustaining prescription volumes. Increasing physician familiarity with long term tyrosine kinase inhibitor management is reinforcing adherence and treatment persistence. Collectively, these structural drivers, supported by regulatory approvals and real world evidence, are expected to maintain steady growth in the global dasatinib market over the forecast period.
Market Concentration & Characteristics
The dasatinib market demonstrates a moderate to high degree of innovation, primarily driven by advancements in targeted oncology therapies and formulation improvements. Research efforts focus on optimizing treatment response, reducing adverse events, and improving patient adherence through refined dosing strategies. Pharmaceutical companies continue to explore combination therapies that enhance clinical outcomes in resistant leukemia cases. Innovations in diagnostic technologies support earlier detection and personalized therapy selection, which strengthens drug utilization. Lifecycle management strategies, including new indications and delivery approaches, also contribute to innovation intensity. Continuous clinical research activity maintains scientific interest and competitive differentiation within the market.
The market presents significant barriers to entry due to complex manufacturing processes and stringent quality requirements associated with oncology drugs. High capital investment is required for research, development, clinical validation, and regulatory approvals. Intellectual property considerations and patent landscapes have historically limited early market participation, though generic entry has increased following expirations. Established brand recognition and physician trust toward existing manufacturers create additional competitive challenges for new entrants. Strict pharmacovigilance and compliance standards further increase operational costs. Limited expertise in oncology drug commercialization also restricts smaller companies from entering the market effectively.
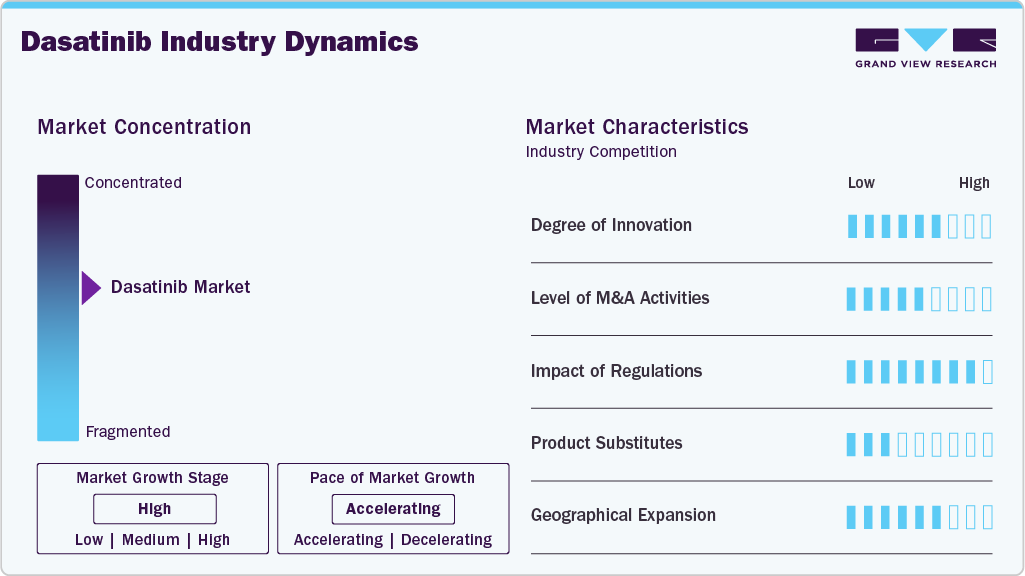
Regulatory frameworks strongly influence the dasatinib market through approval pathways, safety monitoring requirements, and manufacturing compliance standards. Oncology drugs undergo rigorous clinical evaluation to demonstrate efficacy and safety, which affects development timelines and costs. Post marketing surveillance requirements ensure ongoing assessment of adverse events and long-term treatment outcomes. Pricing regulations and reimbursement policies in various regions impact market accessibility and manufacturer profitability. Regulatory harmonization across certain regions has facilitated faster approvals and broader patient access. Compliance with evolving quality standards remains essential for maintaining market presence.
The presence of alternative tyrosine kinase inhibitors creates competitive pressure within the dasatinib market. Drugs such as imatinib, nilotinib, bosutinib, and ponatinib provide multiple therapeutic options for similar indications. Treatment selection often depends on patient tolerance, mutation profile, and disease progression status. Newer generation therapies with improved safety profiles may influence prescribing patterns over time. Physicians may switch therapies in cases of resistance or adverse reactions, affecting demand stability. Despite substitution risks, dasatinib maintains relevance due to its established efficacy in specific patient populations.
Geographical expansion plays a crucial role in shaping market concentration, with growth opportunities emerging across Asia Pacific, Latin America, and Middle East regions. Increasing oncology awareness and improving healthcare infrastructure are enabling broader adoption of targeted therapies. Pharmaceutical companies are expanding distribution partnerships to strengthen regional market penetration. Local manufacturing capabilities in certain countries are supporting supply efficiency and cost competitiveness. Rising patient populations and improved diagnostic capabilities contribute to regional demand growth. Expansion into underserved markets continues to diversify revenue streams and reduce dependence on mature regions.
Drug Type Insights
The generic segment dominated the market with the largest revenue share of 69.24% in 2025 and is the fastest growing segment over the forecast period due to patent expirations that enabled multiple generic manufacturers to enter core markets, increasing competition and lowering prices relative to the branded counterpart, Sprycel, thereby expanding volume and accessibility. Upon generic entry, dasatinib prices were projected to decline by 50‑60%, significantly broadening patient uptake and channel availability. According to BioPharmaDive, in July 2024, generic versions of Sprycel were expected to begin launching as early as September 2024, facilitating wider adoption and market penetration. Lower-cost generics align with broader oncology drug trends where off‑patent products rapidly capture share once exclusivity expires.
Indication Insights
The Chronic Myeloid Leukemia (CML) segment dominated the market with the largest revenue share of 59.93% in 2025 dasatinib is principally indicated for Philadelphia chromosome‑positive CML, resulting in consistently higher prescription volumes relative to other indications such as ALL. CML represents the most common adult leukemia subtype treated with BCR‑ABL tyrosine kinase inhibitors like dasatinib, making it the core clinical application. According to the U.S. FDA prescribing information, in September 2024, dasatinib is approved for adult patients with newly diagnosed Ph+ CML in chronic, accelerated, or blast phases, reinforcing its primary use in this disease category. Additionally, epidemiological data confirm CML’s predominance in adult leukemia cases, accounting for a substantial proportion of overall leukemia incidence.
The Acute Lymphoblastic Leukemia (ALL) segment is the fastest‑growing segment projected to grow at a CAGR of 5.66% over the forecast period owing to expanding clinical role in Philadelphia chromosome‑positive (Ph+) ALL and incorporation into combination regimens increases therapeutic demand beyond its historical CML focus. Rising ALL incidence, particularly in adult and pediatric populations with Ph+ disease, supports broader utilization of dasatinib in frontline and relapsed/refractory settings, improving clinical adoption. According to ClinicalTrials.gov, in February 2026, an interventional study (NCT06124157) was active testing dasatinib plus chemotherapy in ALL patients, indicating ongoing research expansion that underpins future use. Additionally, peer‑reviewed clinical data show dasatinib’s efficacy in Ph+ ALL with substantial hematologic response rates, supporting its growing role in this indication.
Distribution Channel Insights
The hospital pharmacies segment dominated the market with the largest revenue share of 58.62% in 2025, due to most oncology treatments, including targeted therapies such as dasatinib, are initiated and dispensed through hospital pharmacy departments where specialist oversight, infusion coordination, and patient monitoring occur. Hospital pharmacies typically handle high‑cost oncology drugs, ensuring compliance with treatment protocols and payer requirements, which funnels significant volumes through these channels. According to the U.S. FDA drug approval label, dasatinib’s administration and monitoring guidelines recommend management within clinical settings that align with hospital pharmacy distribution. In addition, a 2024 American Society of Health‑System Pharmacists report notes that hospital pharmacies accounted for over 60 % of oral oncology drug dispensing volume in institutional settings, corroborating dominant channel utilization.
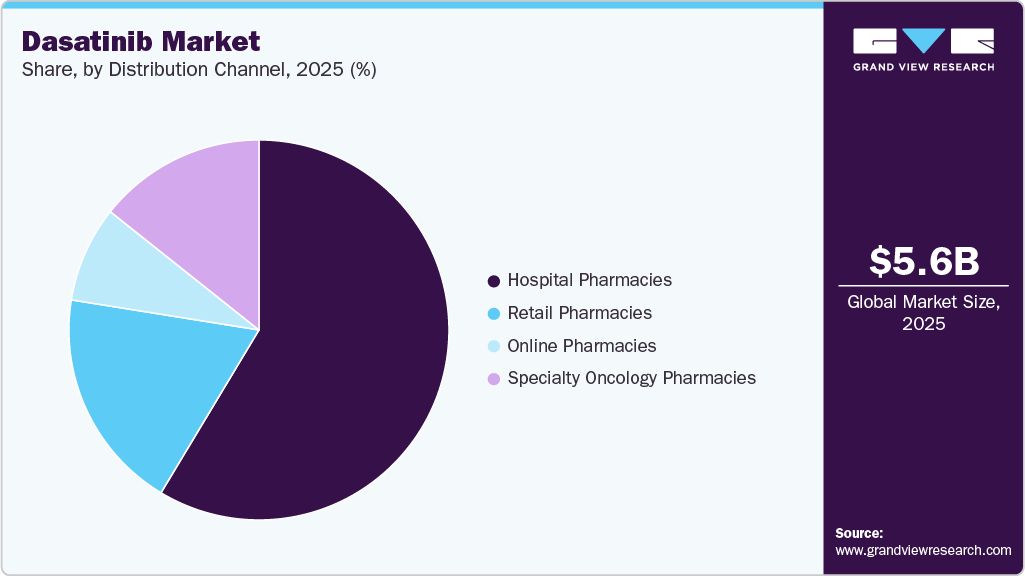
The online pharmacies segment is projected to grow at a CAGR of 7.42% over the forecast period due to increasing digital adoption and e‑health integration enable patients to fill specialty prescriptions remotely, reducing geographic and access barriers for long‑term therapies. Expanded telemedicine and e‑prescribing infrastructure facilitates secure electronic transmission of prescriptions directly to licensed online dispensaries, broadening patient reach. As documented in academic analyses, internet‑based pharmacy platforms are a rapidly developing pharmaceutical supply channel that complements traditional brick‑and‑mortar outlets and supports remote prescription fulfillment. In September 2024, the online pharmacy market was recognized globally as a rapidly developing channel of pharmaceutical supply, reflecting accelerated uptake of internet medicine distribution.
Regional Insights
North America Dasatinib Market Trends
North America held the largest dasatinib market share of 54.17% in 2025 due to advanced oncology treatment infrastructure and high adoption of targeted therapies. Strong clinical awareness among healthcare professionals supports early diagnosis and treatment initiation for leukemia patients. The presence of major pharmaceutical companies and established distribution systems strengthens product availability across hospitals and specialty pharmacies. Favorable reimbursement coverage for oncology treatments increases patient access to innovative therapies. Continuous clinical research activity and guideline driven treatment practices support consistent prescription volumes. High healthcare spending capacity further sustains regional market leadership.
U.S. Dasatinib Market Trends
The U.S. dasatinib market represents the largest share within North America due to a significant patient population receiving targeted cancer therapies. Physicians widely adopt tyrosine kinase inhibitors as standard treatment options for chronic myeloid leukemia management. Strong insurance coverage and specialty pharmacy networks improve treatment continuity for patients requiring long term therapy. Extensive clinical trials and academic research institutions contribute to ongoing therapeutic advancements. Early adoption of newly approved generics enhances accessibility across different income groups. High awareness regarding precision oncology strengthens market demand across the country.
Europe Dasatinib Market Trends
Europe holds a substantial share of the dasatinib market supported by well-established healthcare systems and standardized oncology treatment protocols. Increasing diagnosis rates for hematologic malignancies contribute to steady demand for targeted therapies. Access to multiple treatment options through hospital-based care models improves patient reach. Pharmaceutical companies maintain strong regional presence through collaborations with healthcare providers and distributors. Growing adoption of personalized medicine approaches enhances treatment outcomes and physician confidence. Stable reimbursement frameworks across several countries sustain consistent market utilization.
The UK shows steady growth in the dasatinib market driven by structured cancer care pathways and strong clinical adherence to treatment guidelines. Increased awareness of leukemia symptoms encourages timely medical intervention and therapy initiation. Hospital based oncology centers play a key role in prescribing targeted treatments for eligible patients. Access to specialized hematology services supports appropriate therapy selection for resistant or intolerant cases. Ongoing research collaborations between academic institutions and pharmaceutical companies contribute to clinical knowledge expansion. The presence of advanced diagnostic technologies further strengthens treatment adoption.
Germany represents a key European market due to its advanced healthcare infrastructure and strong emphasis on innovative cancer therapies. High physician expertise in hematology supports widespread adoption of targeted treatments for leukemia. Availability of comprehensive insurance coverage improves patient access to long term therapies. Pharmaceutical manufacturers maintain strong supply chains within the country, ensuring consistent drug availability. Clinical research initiatives and participation in multinational studies enhance treatment awareness. Rising focus on precision medicine continues to support demand for targeted oncology drugs.
France demonstrates stable demand for dasatinib supported by a structured healthcare delivery system and access to specialized oncology care. Physicians follow evidence-based treatment protocols that include tyrosine kinase inhibitors for leukemia management. Increased patient monitoring and follow up programs contribute to sustained therapy adherence. Hospital pharmacies serve as primary distribution channels, ensuring controlled access to oncology medications. Pharmaceutical companies maintain strong partnerships with healthcare providers to improve product availability. Continuous advancements in diagnostic technologies support earlier detection and treatment initiation.
Asia Pacific Dasatinib Market Trends
Asia Pacific dasatinib market is expected to register the fastest CAGR of 5.59% over the forecast period due to a large patient population and expanding access to oncology treatments. Rising awareness of targeted therapies is improving diagnosis and treatment rates across emerging economies. Growth in pharmaceutical manufacturing capabilities is enhancing regional supply and affordability of generic versions. Expanding healthcare infrastructure in urban areas supports broader therapy adoption. Increasing medical tourism activity in certain countries contributes to higher treatment volumes. Economic development and improving healthcare expenditure continue to drive rapid market expansion.
Japan represents a mature market within Asia Pacific due to advanced medical technology and strong clinical adoption of targeted therapies. Physicians emphasize precision treatment approaches supported by detailed diagnostic testing. Aging population demographics contribute to a steady incidence of hematologic malignancies requiring long term therapy. Pharmaceutical companies maintain strong distribution networks through hospital and specialty channels. High standards of clinical practice ensure consistent treatment adherence among patients. Continuous research initiatives support therapeutic innovation and optimized patient outcomes.
China is experiencing significant growth in the dasatinib market due to increasing cancer awareness and expanding healthcare access across urban regions. Domestic pharmaceutical companies are actively producing generic versions, improving affordability for a larger patient population. Rapid improvements in diagnostic capabilities are enabling earlier detection of leukemia cases. Expansion of hospital infrastructure supports greater availability of advanced oncology treatments. Strategic partnerships between manufacturers and healthcare providers enhance product distribution efficiency. Rising healthcare expenditure contributes to growing adoption of targeted therapies across the country.
Latin America Dasatinib Market Trends
Latin America shows moderate growth in the dasatinib market supported by improving access to oncology treatments and increasing awareness of leukemia management. Expansion of private healthcare facilities is enabling broader adoption of targeted therapies. Pharmaceutical companies are strengthening regional distribution channels to improve drug availability. Growing demand for cost effective treatment options is encouraging uptake of generic versions. Urban healthcare centers serve as primary hubs for specialized cancer care services. Economic improvements in certain countries are contributing to gradual market expansion.
Brazil leads the Latin American dasatinib market due to its large population base and expanding oncology treatment infrastructure. Increasing diagnosis rates for hematologic cancers are driving demand for targeted therapies. Availability of both branded and generic drugs improves treatment accessibility across different healthcare settings. Physicians are increasingly adopting tyrosine kinase inhibitors for long term disease management. Growth in private healthcare services supports patient access to advanced treatments. Rising awareness of cancer care options contributes to sustained market growth within the country.
Middle East & Africa Dasatinib Market Trends
The MEA region is witnessing gradual growth in the dasatinib market supported by improving healthcare infrastructure and increasing oncology awareness. Expansion of specialized cancer treatment centers is enabling access to advanced therapies. Pharmaceutical companies are focusing on regional partnerships to strengthen supply chains and distribution networks. Growing medical education initiatives are improving physician familiarity with targeted treatment options. Urban healthcare facilities are adopting modern diagnostic technologies for leukemia detection. Economic development in certain countries contributes to increasing healthcare spending and treatment adoption.
Saudi Arabia represents a key market within the MEA region due to expanding healthcare facilities and increasing access to advanced oncology treatments. Hospitals are adopting modern diagnostic and therapeutic technologies to manage complex cancer cases. Rising awareness of targeted therapies among healthcare professionals supports prescription growth. Pharmaceutical companies maintain active distribution partnerships to ensure consistent product availability. Increasing investment in specialized medical services strengthens treatment capacity across major cities. Growing patient awareness regarding early diagnosis contributes to improved therapy utilization.
Key Dasatinib Company Insights
Bristol-Myers Squibb Company maintains its presence in the dasatinib market through its branded therapy and ongoing clinical research in hematologic malignancies. Teva Pharmaceutical Industries Ltd. and Sandoz Group AG are contributing to market competition through the introduction of generic versions, improving treatment access across multiple regions. Sun Pharmaceutical Industries Limited, Hetero Drugs Limited, and Aurobindo Pharma Limited are expanding their oncology portfolios by increasing manufacturing capabilities and strengthening distribution networks. Dr. Reddy’s Laboratories Limited and Intas Pharmaceuticals Ltd. are focusing on cost-effective formulations and collaborative agreements to expand their reach in emerging markets. Cipla Limited and Lupin Limited are utilizing their experience in generic oncology medicines to improve affordability and patient availability. The market reflects evolving competition influenced by patent expirations, increasing demand for targeted leukemia treatments, and broader access to generic therapies worldwide.
Key Dasatinib Companies:
The following key companies have been profiled for this study on the dasatinib market.
- Bristol-Myers Squibb Company
- Teva Pharmaceutical Industries Ltd.
- Sandoz Group AG
- Sun Pharmaceutical Industries Limited
- Hetero Drugs Limited
- Aurobindo Pharma Limited
- Dr. Reddy’s Laboratories Limited
- Intas Pharmaceuticals Ltd.
- Cipla Limited
- Lupin Limited
Recent Developments
-
In February 2026, Lupin Limited announced the U.S. launch of Dasatinib Tablets after receiving Abbreviated New Drug Application approval from the U.S. Food and Drug Administration. The product was introduced in six strengths, 20 mg, 50 mg, 70 mg, 80 mg, 100 mg, and 140 mg, for adults and pediatric patients aged 1 year and older.
-
In November 2025, Alembic Pharmaceuticals Limited announced that it had received final approval from the U.S. Food and Drug Administration for its Abbreviated New Drug Application for Dasatinib Tablets in 20 mg, 50 mg, 70 mg, 80 mg, 100 mg, and 140 mg strengths.
-
In April 2025, Aurobindo Pharma’s wholly-owned arm Eugia Pharma Specialities Ltd had received final approval from the U.S. Food and Drug Administration to manufacture and market generic Dasatinib tablets in strengths of 20 mg, 50 mg, 70 mg, 80 mg, 100 mg, and 140 mg.
Dasatinib Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 5.65 billion
Revenue forecast in 2033
USD 7.57 billion
Growth rate
CAGR of 4.27% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Drug type, indication, distribution channel, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Bristol-Myers Squibb Company; Teva Pharmaceutical Industries Ltd.; Sandoz Group AG; Sun Pharmaceutical Industries Limited; Hetero Drugs Limited; Dr. Reddy’s Laboratories Limited; Intas Pharmaceuticals Ltd.; Cipla Limited; Lupin Limited
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Dasatinib Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global dasatinib market report based on drug type, indication, distribution channel, and region:
-
Drug Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Branded
-
Generic
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Chronic Myeloid Leukemia (CML)
-
Acute Lymphoblastic Leukemia (ALL)
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
Specialty Oncology Pharmacies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Based on drug type, the generic segment dominated the market with the largest revenue share of 69.24% in 2025, due to patent expirations that enabled multiple generic manufacturers to enter core markets, increasing competition and lowering prices relative to the branded counterpart.
b. Some of the key players in dasatinib market are Bristol-Myers Squibb Company, Teva Pharmaceutical Industries Ltd., Sandoz Group AG, Sun Pharmaceutical Industries Limited, Hetero Drugs Limited, Dr. Reddy’s Laboratories Limited, Intas Pharmaceuticals Ltd., Cipla Limited, Lupin Limited
b. The global dasatinib market is expanding due to the rising prevalence of hematologic malignancies, particularly chronic myeloid leukemia and Philadelphia chromosome positive acute lymphoblastic leukemia. Increasing awareness of targeted oncology therapies has improved diagnosis and early treatment initiation across developed and emerging regions.
b. The global dasatinib market size was valued at USD 5.59 billion in 2025 and is anticipated to reach USD 5.65 billion in 2026
b. The global dasatinib market is expected to witness a compound annual growth rate of 4.27% from 2026 to 2033 to reach USD 7.57 billion by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









