- Home
- »
- Clinical Diagnostics
- »
-
Colorectal Cancer Diagnostics Market, Industry Report, 2033GVR Report cover
![Colorectal Cancer Diagnostics Market Size, Share & Trends Report]()
Colorectal Cancer Diagnostics Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Diagnostic Kits & Assays, Instruments & Systems, Reagents & Consumables), By Test Type, By Cancer Type, By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-877-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Colorectal Cancer Diagnostics Market Summary
The global colorectal cancer diagnostics market size was estimated at USD 9.53 billion in 2025 and is projected to reach USD 14.39 billion by 2033, growing at a CAGR of 5.34% from 2026 to 2033. The industry is driven by the increasing incidence of colorectal cancer, rising demand for early and accurate diagnostic solutions, growing adoption of non-invasive screening methods, and advancements in molecular and genomic diagnostic technologies.
Key Market Trends & Insights
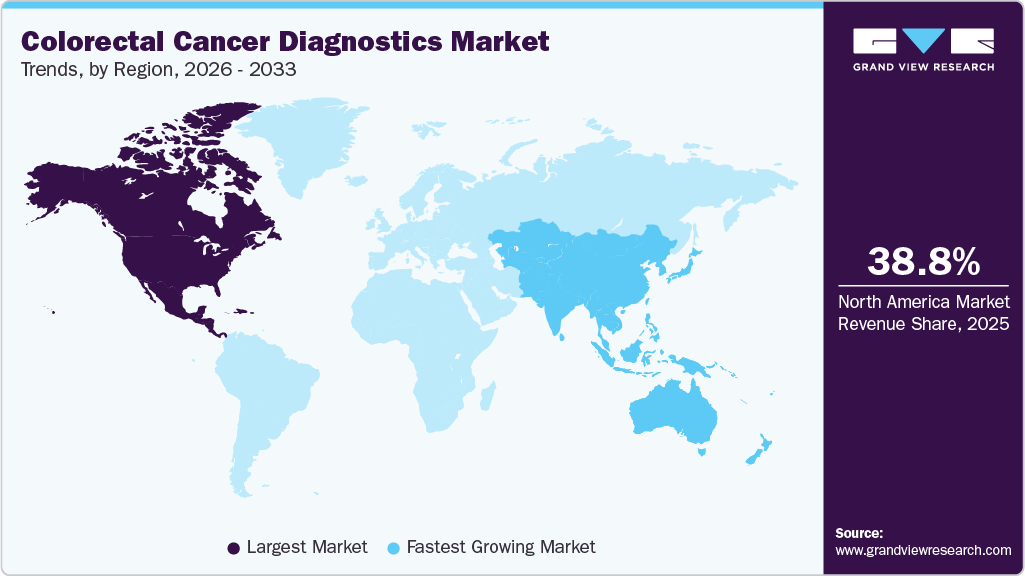
- North America colorectal cancer diagnostics market dominated the market and accounted for the largest revenue share of 38.82% in 2025.
- The U.S. led the North America market and held the largest revenue share in 2025.
- Based on product, the diagnostic kits & assays segment dominated the global market and accounted for the largest revenue share of 41.96% 2025.
- Based on test type, the stool based tests segment held the largest revenue share of 60.30% in 2025.
- Based on cancer type, the adenocarcinoma segment held the largest revenue share of 92.80% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 9.53 Billion
- 2033 Projected Market Size: USD 14.39 Billion
- CAGR (2026-2033): 5.34%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The rising prevalence of colorectal cancer, particularly among younger adults, is further increasing the need for effective screening and diagnostic approaches. In addition, the growing financial burden associated with colorectal cancer treatment is driving demand for more cost-effective and efficient diagnostic solutions. Technological advancements in genomics and molecular diagnostics are also improving the accuracy and personalization of colorectal cancer detection. Moreover, the increasing emphasis on early disease detection and preventive healthcare is encouraging the adoption of innovative colorectal cancer diagnostic technologies.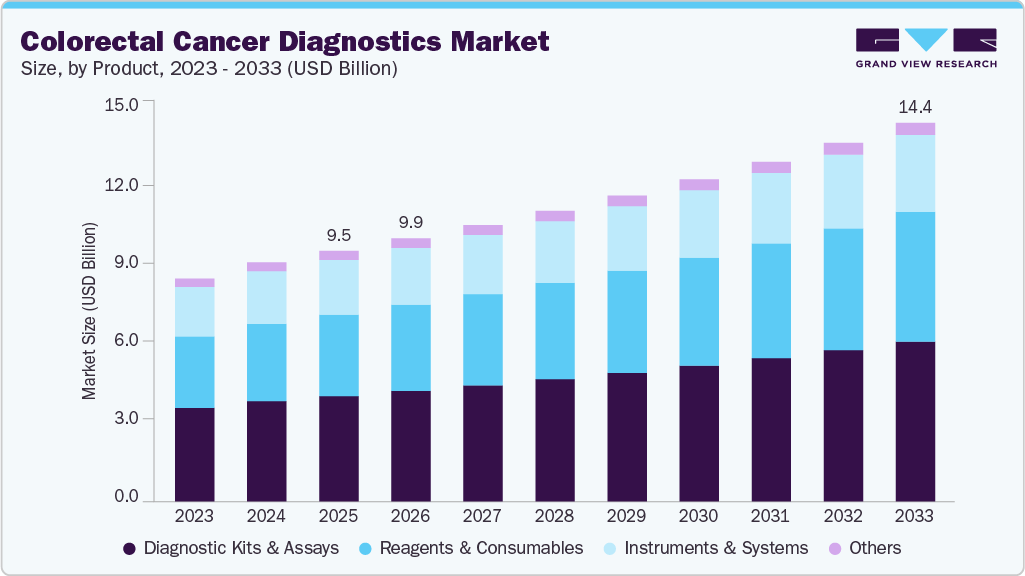
The global burden of colorectal cancer (CRC) continues to increase, making it among the most commonly diagnosed cancers worldwide. According to estimates from the International Agency for Research on Cancer (IARC) and the GLOBOCAN study published in Gut, approximately 1.9 million new CRC cases and over 930,000 deaths were reported globally in 2020. Projections indicate that the global burden could rise to around 3.2 million new cases and 1.6 million deaths annually by 2040. This increasing disease prevalence is a significant factor driving demand for improved screening, diagnostic, and treatment solutions. CRC is considered one of the most preventable cancers when detected early, particularly through the identification and removal of precancerous lesions. However, the growing incidence in transitioning economies and among younger adults highlights the need for improved awareness, preventive strategies, and broader screening initiatives to reduce future disease burden and mortality.
Early detection plays a critical role in improving survival outcomes, as colorectal cancer is highly treatable when diagnosed at an early stage. Timely diagnosis not only improves patient survival rates but also significantly reduces treatment costs compared to advanced-stage disease management. Consequently, the increasing emphasis on early detection is accelerating the adoption of advanced screening technologies. Non-invasive, at-home stool-based screening tests such as fecal immunochemical tests (FIT) and stool DNA tests (e.g., Cologuard) are gaining widespread acceptance due to their convenience and effectiveness, thereby improving patient compliance with screening programs. From an economic perspective, the treatment of late-stage colorectal cancer can cost up to three times more than early-stage treatment, strengthening the value of large-scale screening programs and preventive diagnostics.
Government-led screening initiatives also play a pivotal role in improving public awareness and increasing access to colorectal cancer screening. Many countries have implemented population-based screening programs aimed at reducing CRC-related mortality through early diagnosis and timely intervention. These initiatives typically target specific age groups and utilize screening modalities such as fecal immunochemical tests (FIT) and fecal occult blood tests (FOBT). For instance, in the U.S., the Centers for Disease Control and Prevention (CDC) Colorectal Cancer Control Program (CRCCP) serves as a key public health initiative designed to increase screening rates and reduce deaths associated with colorectal cancer.
The CRCCP primarily focuses on adults aged 45 to 75 years, particularly individuals from low-income populations or those with limited access to healthcare services. Prior to launching the full program, the CDC implemented a four-year colorectal cancer screening pilot program (2005-2009) across five locations in the U.S. This pilot provided screening services recommended by the U.S. Preventive Services Task Force (USPSTF). The program demonstrated that structured public health initiatives could significantly improve screening accessibility and facilitate early detection. Following its success, the U.S. Congress approved additional funding, leading to the official launch of the CRCCP in 2009. Over time, the program has evolved from simply providing screening services to collaborating with healthcare systems, clinics, and community organizations to implement evidence-based strategies that increase screening uptake among high-risk populations.
The growth of the colorectal cancer diagnostics market is also supported by the rising adoption of non-invasive screening methods. Non-invasive diagnostic tests, including stool-based and blood-based screening solutions, are gaining significant attention due to their ease of use and improved patient acceptance. Recent clinical advancements include highly sensitive stool-based RNA and DNA tests, which have demonstrated sensitivity levels exceeding 93% for colorectal cancer detection, as well as the development of blood-based cell-free DNA screening tests. These technologies enhance diagnostic accuracy while addressing patient compliance barriers, making colorectal cancer screening more accessible and effective.
Furthermore, the increasing preference for non-invasive diagnostic approaches has accelerated market adoption. Screening methods such as FIT, FOBT, and stool-based DNA tests provide convenient alternatives to traditional diagnostic procedures and support large-scale population screening initiatives. These solutions are particularly valuable in countries implementing national colorectal cancer screening programs, as they enable cost-effective and scalable diagnostic strategies.
Technological advancements in genomic and molecular diagnostics are also transforming colorectal cancer detection. Innovations such as next-generation sequencing (NGS), biomarker-based assays, and liquid biopsy technologies allow for the identification of genetic mutations and molecular alterations associated with colorectal cancer. These approaches facilitate earlier and more accurate diagnosis, enable personalized treatment strategies, and improve disease monitoring. Biomarker-based detection methods represent a promising area of innovation, offering less invasive and more patient-friendly screening solutions. For example, blood-based diagnostic tests require only a simple blood sample, eliminating the need for invasive procedures and complex preparation, while stool-based tests can be performed conveniently at home, further reducing barriers to screening participation.
Market Concentration & Characteristics
The degree of innovation in the CRC diagnostics market is high, driven by rapid advancements in genomics, molecular diagnostics, and artificial intelligence (AI)-enabled screening technologies. Emerging innovations such as liquid biopsies utilizing circulating tumor DNA (ctDNA), RNA-based biomarkers, and advanced molecular assays enable more precise and personalized detection approaches. However, artificial intelligence is transforming screening and diagnostic workflows through technologies such as Computer-Aided Detection (CADe) and Computer-Aided Diagnosis (CADx), which enhance the accuracy and efficiency of imaging-based screening procedures. Collectively, these advancements demonstrate a strong innovation pipeline centered on non-invasive, genomic, and AI-driven diagnostic solutions, significantly improving diagnostic accuracy, operational efficiency, and accessibility of colorectal cancer screening.
Mergers and acquisitions (M&A) activity in the industry remains moderate but strategically significant. Leading companies such as Abbott Laboratories, Exact Sciences, Guardant Health, and Siemens Healthineers are actively following acquisitions and partnerships to strengthen their diagnostic portfolios and expand their molecular testing capabilities. The market is witnessing increasing consolidation as companies focus on accelerating innovation in early cancer detection technologies. For instance, in 2023, Exact Sciences acquired the rights to a blood-based colorectal cancer screening test developed by Freenome, enhancing its capabilities in liquid biopsy–based screening for early CRC detection. This strategic move also intensified competition with Guardant Health, which offers similar liquid biopsy–based screening solutions. Furthermore, in 2025, Abbott Laboratories announced plans to acquire Exact Sciences in a transaction valued at approximately USD 23 billion. The acquisition is expected to integrate Exact Sciences’ flagship colorectal cancer screening test, Cologuard, along with other oncology diagnostic solutions into Abbott’s diagnostics portfolio, significantly strengthening its position in the global cancer screening market.
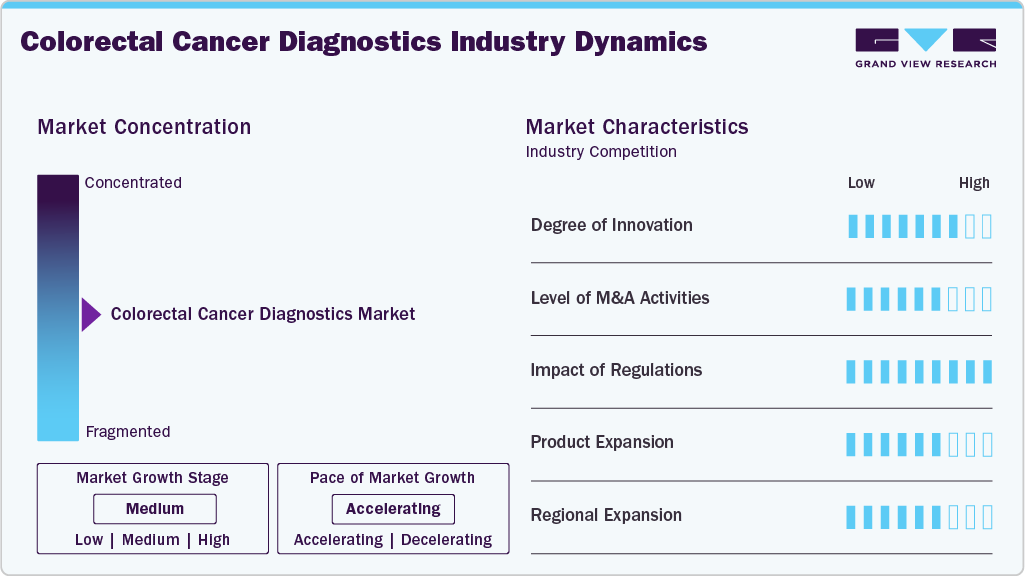
Regulatory frameworks and reimbursement policies play a critical role in shaping the colorectal cancer diagnostics landscape by influencing screening accessibility, adoption rates, and technological innovation. Coverage policies from organizations such as the Centers for Medicare & Medicaid Services (CMS) establish performance thresholds for non-invasive screening tests, for example, requiring blood-based screening tests to achieve at least 74% sensitivity and 90% specificity. These requirements encourage the development and commercialization of higher-accuracy diagnostic technologies. Additionally, regulatory approvals from the U.S. Food and Drug Administration (FDA) for diagnostic tests such as Cologuard and Epi proColon have supported their adoption in clinical practice, particularly among patients seeking non-invasive screening options. Overall, supportive regulatory and reimbursement frameworks are accelerating the transition toward advanced molecular and non-invasive colorectal diagnostic cancer technologies.
The industry is experiencing significant product expansion, driven by a strategic shift from invasive procedures toward high-precision, patient-friendly diagnostic technologies. The molecular diagnostics segment, valued at approximately USD 2.6 billion in 2024, is increasingly incorporating next-generation sequencing (NGS) and PCR-based assays into routine clinical practice to identify genetic mutations such as KRAS, thereby enabling more personalized treatment approaches. Liquid biopsy technologies represent the fastest-growing segment, with a projected CAGR of approximately 13.1%, as they provide a minimally invasive method for detecting circulating tumor DNA (ctDNA). Recent regulatory milestones include the U.S. FDA approvals of Guardant Health’s Shield and Geneoscopy’s ColoSense in 2024, further expanding the availability of advanced screening options. In addition, at-home stool DNA tests continue to dominate the market, accounting for approximately 60.30% of revenue share, as these tests significantly improve patient compliance with screening programs. The approval of next-generation screening solutions such as Cologuard Plus in 2024 further highlights the industry’s shift toward highly sensitive and patient-centric diagnostic technologies.
Regional expansion of the market is influenced by variations in healthcare infrastructure, awareness levels, and technology adoption across key geographic regions. North America currently dominates the market, accounting for approximately 38.87% of the global market share in 2025, supported by well-established screening programs, high awareness levels, and the widespread adoption of advanced technologies such as AI-enabled imaging and molecular diagnostics. The Asia-Pacific region is expected to witness the fastest growth, with a projected CAGR of approximately 6.23%, driven by rapid urbanization, rising disposable incomes, and increasing government-led screening initiatives in countries such as China and India. Europe continues to experience steady growth due to strong healthcare systems and established national screening programs in countries including the UK, Germany, and France. Meanwhile, the LAMEA region (Latin America, the Middle East, and Africa) is expected to demonstrate gradual long-term growth as governments and healthcare organizations continue to invest in healthcare infrastructure development, public awareness campaigns, and early cancer detection programs.
Product Insights
Diagnostic kits & assays dominated the market and accounted for the largest revenue share of 41.96% in 2025 due to the increasing demand for non-invasive, home-based, and highly accurate screening solutions, such as fecal occult blood tests (FOBT). This dominance is primarily driven by the rising global incidence of colorectal cancer, growing awareness of early detection, and technological advancements in liquid biopsy and biomarker-based diagnostics. The development of molecular diagnostic technologies targeting biomarkers such as KRAS, NRAS, and BRAF mutations enables more precise and early detection while supporting personalized treatment strategies. Additionally, innovative screening solutions, including stool DNA tests and liquid biopsy assays, are strengthening the segment’s growth by providing convenient and highly sensitive methods for detecting colorectal cancer at earlier stages.
The reagents and consumables segment is anticipated to grow at the fastest rate over the forecast period. The growth of this segment is driven by the high volume usage of testing materials, including antibodies, probes, and diagnostic kits required for routine screening and laboratory testing. increasing demand for screening tools such as fecal immunochemical tests (FIT), stool-based DNA assays, and molecular diagnostic reagents is further supporting segment expansion. Overall, the segment is benefiting from the rising focus on early cancer detection and the increasing adoption of non-invasive screening technologies across healthcare systems.
Test Type Insights
Stool based testing segment dominated the market and accounted for the largest revenue share of 60.30% in 2025. The dominance of this segment is primarily attributed to the widespread use of fecal occult blood tests (FOBT), fecal immunochemical tests (FIT), and stool DNA tests, which detect blood or genetic biomarkers associated with colorectal cancer. The increasing incidence of CRC, particularly among adults aged 45 years and above, along with advancements in high-sensitivity and non-invasive screening technologies such as next-generation FIT tests, has significantly increased the adoption of stool-based screening methods. In addition, government-supported screening initiatives and population-based screening programs have further accelerated the demand for stool-based testing due to its convenience, cost-effectiveness, and suitability for large-scale screening.
Blood-based tests are anticipated to grow at the fastest rate over the forecast period. The growth of this segment is primarily driven by increasing demand for minimally invasive screening methods that require only a simple blood sample, improving patient comfort and screening compliance. Blood-based diagnostic technologies, particularly those based on circulating tumor DNA (ctDNA), cell-free DNA, and biomarker detection, are gaining significant attention as they enable early detection of colorectal cancer with improved convenience compared to traditional screening approaches.
Cancer Type Insights
The adenocarcinoma cancer type segment dominated the market, accounting for a revenue share of 92.80% in 2025, and is expected to grow at the fastest rate during the forecast period. Adenocarcinoma is the most prevalent form of colorectal cancer, representing over 90% of all colon cancer cases worldwide. A study conducted by Ji et al., which analyzed clinical data from 1,092 primary colorectal cancer patients, also confirmed adenocarcinoma as the predominant cancer type among CRC patients. The high prevalence of this cancer type is largely attributed to the fact that most colorectal cancers originate from adenomatous polyps, which are common precancerous lesions found in the colon and rectum. Additionally, current clinical screening guidelines, diagnostic protocols, and reimbursement frameworks are largely designed to support the detection and management of adenocarcinoma compared with less common colorectal cancer subtypes such as lymphomas or sarcomas. As a result, many of the major diagnostic innovations in the colorectal cancer diagnostics market, including liquid biopsy technologies, AI-assisted colonoscopy systems, and molecular residual disease (MRD) tests, have primarily been developed to enhance the early detection, diagnosis, and monitoring of adenocarcinoma, further supporting the strong market dominance of this segment.
The other cancer type segment is anticipated to grow at a lucrative rate over the forecast period. This segment primarily includes rare histological subtypes of colorectal cancer, such as lymphomas, neuroendocrine tumors, and sarcomas, which require specialized diagnostic approaches. Although the patient population associated with these cancer types is relatively small compared to adenocarcinoma, the segment presents significant growth potential due to the complexity of diagnosis and the need for advanced diagnostic technologies. Detecting these rare subtypes often requires sophisticated diagnostic tools, including advanced imaging techniques, molecular diagnostics, and specialized pathology testing, which contribute to higher diagnostic costs and increased revenue generation. Additionally, growing research efforts focused on understanding rare colorectal cancer subtypes and improving diagnostic accuracy are expected to further support the growth of this segment.
End Use Insights
Hospitals led the market, accounting for 38.13% of revenue share in 2025.This dominance is primarily attributed to the presence of advanced medical infrastructure and comprehensive care capabilities within hospital settings. Hospitals are equipped with sophisticated diagnostic technologies, including advanced imaging systems, gastroenterology laboratories, and specialized oncology departments, which enable accurate and efficient diagnosis of conditions such as colorectal cancer. In addition, hospitals can perform diagnostic procedures such as colonoscopy and biopsy, along with subsequent therapeutic interventions within the same facility, ensuring integrated patient care. The availability of highly skilled specialists, including gastroenterologists, oncologists, and surgeons, further supports effective patient evaluation, diagnosis, and treatment management.
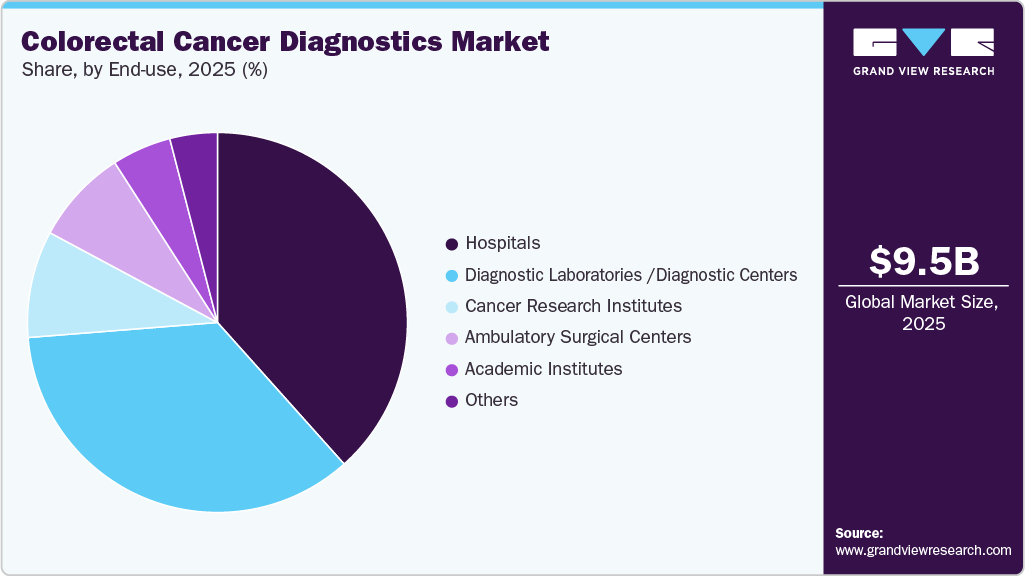
The diagnostic centers segment is anticipated to grow at the fastest rate over the forecast period. This growth is primarily driven by the increasing preference for convenient and specialized outpatient diagnostic services. Diagnostic centers play a key role in conducting high-volume non-invasive screening tests, including fecal occult blood tests (FOBT), fecal immunochemical tests (FIT), and specialized molecular diagnostic analyses for colorectal cancer detection. These facilities are increasingly favored by patients due to their shorter turnaround times, streamlined scheduling processes, and greater accessibility, allowing individuals to avoid the longer waiting periods often associated with large hospital systems.
Regional Insights
North America colorectal cancer diagnostics market led the global industry and accounted for the largest revenue share of 38.82% in 2025. The region represents one of the most developed markets for colorectal cancer diagnostics, supported by well-established screening programs, advanced healthcare infrastructure, and high awareness regarding early cancer detection. In addition, the rapid adoption of molecular diagnostic technologies and advanced screening tools has further strengthened the region’s diagnostic capabilities. Organized screening strategies, including colonoscopy, fecal immunochemical tests (FIT), and stool-based molecular tests, are widely implemented across the U.S. and Canada as part of national and regional screening initiatives.
U.S. Colorectal Cancer Diagnostics Market Trends
The colorectal cancer diagnostics market in the U.S. is supported by high disease awareness, increasing incidence rates, advanced healthcare infrastructure, and well-established early screening initiatives. The market is largely driven by the adoption of non-invasive stool-based screening tests such as Cologuard, alongside traditional diagnostic procedures like colonoscopy. In addition, the expansion of home-based testing and outpatient diagnostic facilities is further supporting market growth by improving accessibility and patient compliance with screening programs.
Europe Colorectal Cancer Diagnostics Market Trends
The colorectal cancer diagnostics market in Europe represents a significant market due to the high disease burden and well-structured population-based screening programs implemented across many countries. Incidence rates are among the highest globally, particularly in countries such as Norway, Denmark, and the Netherlands. While Western Europe benefits from strong screening initiatives using fecal occult blood tests and colonoscopy, parts of Central and Eastern Europe continue to report higher mortality rates, highlighting the need for improved early detection and diagnostic technologies.
Asia Pacific Colorectal Cancer Diagnostics Market Trends
The colorectal cancer diagnostics market in Asia Pacific is driven by a large population base, rising cancer incidence, and improving healthcare infrastructure. Eastern Asia carries a significant disease burden, reporting one of the highest numbers of colorectal cancer cases globally. Although prevalence rates are generally lower than in Western countries, increasing adoption of screening programs and preventive healthcare initiatives is supporting market growth across the region.
Latin America Colorectal Cancer Diagnostics Market Trends
The colorectal cancer diagnostics market in Latin America is growing, with countries such as Brazil and Argentina witnessing rising colorectal cancer incidence due to aging populations and lifestyle changes. However, limited access to screening programs and advanced diagnostic technologies continues to affect early detection rates. Ongoing improvements in healthcare infrastructure and cancer control initiatives are expected to gradually increase diagnostic adoption across the region.
Middle East & Africa Colorectal Cancer Diagnostic Market Trends
The colorectal cancer diagnostics market in the Middle East & Africa is expected to grow over the forecast period. While overall incidence rates remain lower compared to other regions, diagnostic demand is increasing as healthcare infrastructure and screening awareness improve. Countries such as Israel and several Middle Eastern nations report relatively higher incidence rates, which is encouraging the development and adoption of colorectal cancer screening and diagnostic services.
Key Colorectal Cancer Diagnostic Company Insights
The competitive landscape of the colorectal cancer diagnostics market is characterized by the presence of several global diagnostic companies that focus on molecular diagnostics, screening technologies, and advanced imaging solutions. Market participants compete primarily through product innovation, regulatory approvals, strategic collaborations, and the expansion of their diagnostic portfolios. The market remains relatively consolidated, with the top ten industry leaders, including companies such as Exact Sciences and Roche, accounting for nearly 50% of the global market revenue.
Competitive dynamics are increasingly being shaped by the industry’s shift toward liquid biopsy technologies and non-invasive molecular screening solutions, as leading companies continue to invest significantly in research and development to maintain their market position. To strengthen their long-term leadership, these companies are actively pursuing strategic mergers and acquisitions, technological innovation, and precision diagnostics initiatives. Their efforts are largely focused on integrating high-sensitivity genomic profiling and advanced molecular diagnostics into routine clinical practice, addressing the growing demand for early cancer detection and personalized treatment strategies.
Key Colorectal Cancer Diagnostic Companies:
The following key companies have been profiled for this study on the colorectal cancer diagnostic market.
- Exact Sciences Corporation
- F. Hoffmann-La Roche Ltd
- Abbott
- Danaher Corporation
- Guardant Health, Inc.
- Quest Diagnostics Incorporated
- Fujifilm Holdings Corporation
- Olympus Corporation
- Epigenomics AG
- Eiken Chemical Co., Ltd.
Recent Developments
-
In March 2026, Guardant Health, Inc. announced that its blood-based colorectal cancer screening test, Shield, became accessible through the national test ordering and collection network of Quest Diagnostics. Through this collaboration, physicians can order the Shield test via Quest’s electronic health record systems, and patients can complete screening through Quest’s nationwide network of patient service centers and phlebotomists, expanding access to non-invasive blood-based colorectal cancer screening.
-
In July 2025, Exact Sciences Corporation expanded its colorectal cancer screening partnership with Humana Inc. to improve access to the Cologuard Plus test. Under the agreement, the non-invasive stool-based screening test will be available as an in-network service for eligible Humana Medicare Advantage members across the U.S. starting August 2025, helping increase patient access to convenient at-home colorectal cancer screening
-
In May 2024, Geneoscopy Inc. received U.S. FDA approval for ColoSense, a non-invasive multi-target stool RNA (mt-sRNA) test designed for colorectal cancer screening in adults aged 45 years and older at average risk. The test detects colorectal cancer and advanced adenomas using RNA biomarkers from stool samples, providing a convenient and non-invasive screening alternative. This approval marks the introduction of the first FDA-approved stool RNA-based colorectal cancer screening test, expanding the availability of molecular diagnostic solutions for early cancer detection.
Colorectal Cancer Diagnostic Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 9.99 billion
Revenue forecast in 2033
USD 14.39 billion
Growth rate
CAGR of 5.34% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, test type, cancer type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; Rest of Europe; China; Japan; India; Australia; Thailand; South Korea; Rest of APAC; Brazil; Argentina; Rest of Latin America; South Africa; Saudi Arabia; UAE; Kuwait; Rest of MEA
Key companies profiled
Exact Sciences Corporation; F. Hoffmann-La Roche Ltd (Roche Diagnostics); Abbott; Danaher Corporation;Guardant Health, Inc.;Quest Diagnostics Incorporated;Fujifilm Holdings Corporation;Olympus Corporation;Epigenomics AG;Eiken Chemical Co., Ltd
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Colorectal Cancer Diagnostic Market Report Segmentation
This report forecasts revenue growth at the global, regional and country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global colorectal cancer diagnostics market report on the basis of product, test type, cancer type, end-use, and region:
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Diagnostic Kits & Assays
-
Instruments & Systems
-
Reagent & Consumables
-
Others
-
-
Test Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Stool-Based Tests
-
Endoscopy-Based Tests
-
Imaging Tests
-
Blood-Based Tests
-
Others
-
-
Cancer Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Adenocarcinoma
-
Others
-
-
End Use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Hospitals Laboratories
-
Diagnostic Laboratories / Diagnostic Centers
-
Cancer Research Institutes
-
Ambulatory Surgical Centers
-
Academic Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
Norway
-
-
Rest of Europe
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Rest of Middle East
-
-
Frequently Asked Questions About This Report
b. The global colorectal cancer diagnostics market size was estimated at USD 9.53 billion in 2025 and is expected to reach USD 10.00 billion in 2026.
b. The global colorectal cancer diagnostics market is expected to grow at a compound annual growth rate of 5.34% from 2026 to 2033 to reach USD 14.39 billion by 2033.
b. North America dominated the colorectal cancer diagnostics market with a share of 38.82% in 2025. This is attributable to widespread screening programs, advanced healthcare infrastructure, and strong awareness initiatives and the rapid adoption of molecular diagnostic technologies.
b. Some key players operating in the colorectal cancer diagnostics market include Exact Sciences Corporation, F. Hoffmann-La Roche Ltd (Roche Diagnostics), Abbott, Danaher Corporation, Guardant Health, Inc., Quest Diagnostics Incorporated, Fujifilm Holdings Corporation, Olympus Corporation, Epigenomics AG, Eiken Chemical Co., Ltd
b. Key factors that are driving the market growth include increasing incidence of colorectal cancer, rising demand for early and accurate diagnostic solutions, growing adoption of non-invasive screening methods, and advancements in molecular and genomic diagnostic technologies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









