- Home
- »
- Biotechnology
- »
-
Closed System Bioprocessing Market, Industry Report, 2033GVR Report cover
![Closed System Bioprocessing Market Size, Share & Trends Report]()
Closed System Bioprocessing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Bioreactors, Fermenters, Mixing Systems), By Application (Vaccine Manufacturing, Cell And Gene Therapy), By End Use (Pharmaceutical & Biotech Companies), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-868-7
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Closed System Bioprocessing Market Summary
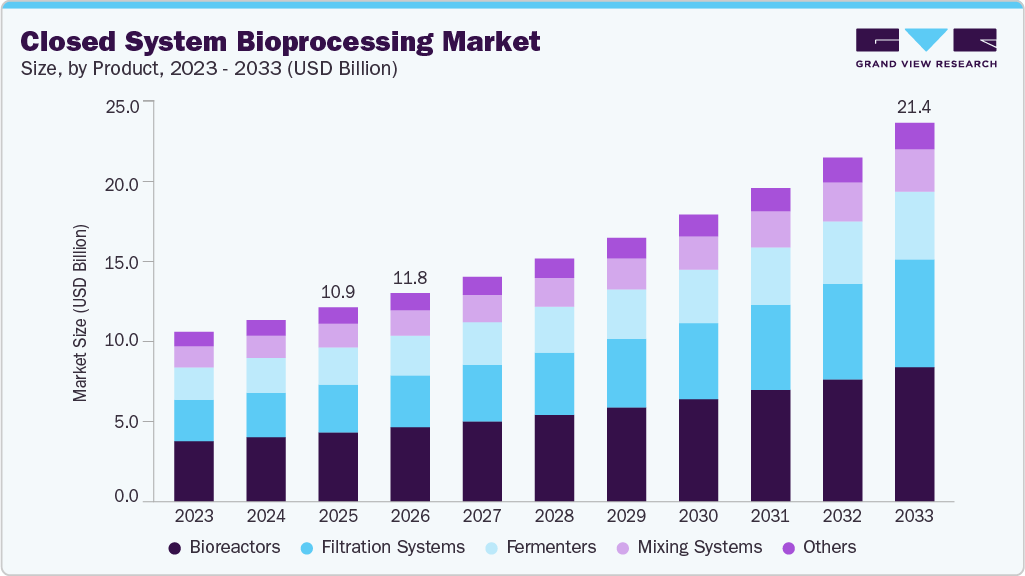
The global closed system bioprocessing market size was valued at USD 10.95 billion in 2025 and is projected to reach USD 21.37 billion by 2033, expanding at a CAGR of 8.90% from 2026 to 2033. This growth is primarily driven by the increasing adoption of single-use technologies, rising biologics and cell & gene therapy production, and the growing need to minimize contamination risks while improving operational efficiency in biomanufacturing processes.
Key Market Trends & Insights
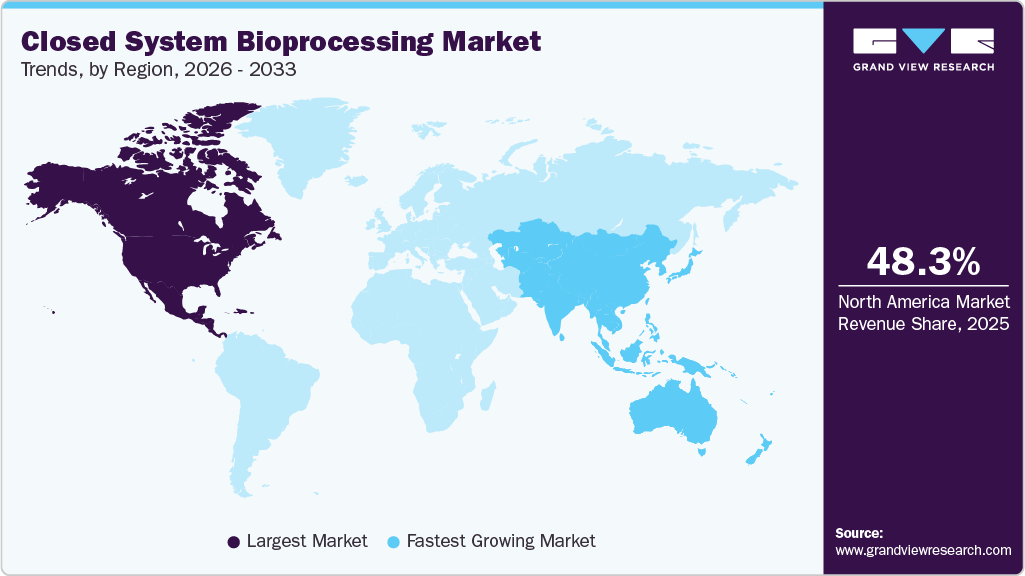
- The North America closed system bioprocessing industry held the largest global market share of 48.31% in 2025.
- The closed system bioprocessing industry in the U.S. is expected to grow over the forecast period due to the growing aging population.
- Based on product, the bioreactors segment dominated the market with a share of 35.67% in 2025.
- Based on application, the biopharmaceutical production segment dominated the market in 2025.
- Based on end use, the pharmaceutical & biotech companies segment dominated the market in 2025.
Market Size & Forecast
- 2025 Market Size: USD 10.95 Billion
- 2033 Projected Market Size: USD 21.37 Billion
- CAGR (2026-2033): 8.90%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Reduced Contamination Risk & Improved Product Quality
Closed system bioprocessing is witnessing strong demand due to its ability to significantly reduce contamination risks in biologics manufacturing. By operating within sealed, sterile pathways, these systems minimize human intervention and environmental exposure, which is critical for sensitive biologics such as monoclonal antibodies, vaccines, and cell therapies. This helps prevent batch failures, product recalls, and regulatory non-compliance while ensuring consistent sterility.
Summary Of Benefits Offered By Closed Processing
Critical Success Factor
Impact of Closed Processing
Flexibility
Enable production in lower classification environment
Enable multiproduct facilities with production of smaller batches run in parallel
Speed
Reduce new facility build times
Reduce lead time between product campaigns
Quality
Ensure the integrity of process steps
Operate process in controlled, closed environments
Reduce risk of contamination of product and protect operators
Costs
Reduce capital and operating expenditures
Reduce or minimize cleanroom classification level
Enable gray space or controlled-not-classified processing
Source: Parenteral Drug Association, Secondary Research, Primary Interviews, Grand View Research
In addition, closed systems enhance product quality and process reliability by reducing manual handling and enabling standardized, automated workflows. They support reproducibility, lower batch rejection rates, and align with GMP requirements for contamination control and traceability, driving increased adoption among biopharmaceutical manufacturers worldwide.
Rising Demand for Biologics, Vaccines & Advanced Therapies
The rising global demand for biologics, such as monoclonal antibodies, recombinant proteins, vaccines, and biosimilars, is a key factor driving the adoption of closed system bioprocessing. As biologics are highly sensitive to contamination and process variability, sealed and sterile processing environments are essential to maintain product quality, stability, and regulatory compliance, prompting increased investment from biopharmaceutical manufacturers.
In addition, the rapid growth of advanced therapies, including cell and gene therapies and personalized treatments, is further boosting demand for closed systems. These technologies support aseptic, automated, and flexible manufacturing workflows, enabling consistent product quality, faster production timelines, and reduced contamination risks in advanced therapeutic production.
Market Concentration & Characteristics
The closed system bioprocessing industry exhibits a high degree of innovation, driven by advancements in single-use technologies, automation, and digital integration. Innovations such as sensor-enabled disposable bioreactors, automated aseptic connectors, and real-time monitoring systems are improving sterility, efficiency, and scalability. For instance, in August 2023, in the United States, Sartorius and Repligen launched an integrated Biostat STR XCell ATF bioreactor system, streamlining perfusion-enabled, single-use upstream processing for intensified, closed-system biomanufacturing.
The market shows moderate to high M&A activity, driven by strategic efforts to expand single-use technology portfolios, strengthen biomanufacturing capabilities, and enhance global market presence. Leading life science companies are actively acquiring niche bioprocess equipment and consumables providers to integrate end-to-end closed processing solutions, accelerate innovation, and meet the growing demand for biologics and advanced therapy manufacturing.

The industry is strongly driven by stringent regulatory requirements focused on contamination control, product safety, and GMP compliance. Regulatory agencies favor closed systems as they reduce human intervention, improve sterility assurance, and enhance process traceability, thereby accelerating their adoption in biopharmaceutical manufacturing.
The closed system bioprocessing industry is experiencing strong product expansion, with companies introducing advanced single-use bioreactors, closed transfer systems, and automated processing solutions. These innovations aim to enhance sterility, scalability, and flexibility in biologics manufacturing.
The market is witnessing notable regional expansion, driven by increasing investments in biomanufacturing infrastructure across emerging economies, particularly in the Asia Pacific and Latin America. Growing biologics production, supportive government initiatives, and the expansion of contract manufacturing organizations are accelerating the adoption of closed processing technologies in these regions. For instance, in June 2025, in the United States, Europe, and the Asia Pacific, Cytiva expanded global manufacturing by USD 1.6 billion, increasing capacity for single-use technologies, filtration, chromatography, and cell culture media to support advanced therapeutics.
Product Insights
In 2025, the bioreactors segment led the closed system bioprocessing industry with a share of 35.67%, driven by their critical role in upstream biologics production, increasing adoption of single-use bioreactors, and rising demand for scalable, contamination-free cell culture systems. Their ability to support flexible manufacturing, reduce cleaning requirements, and enhance process efficiency further contributed to segment dominance.
The fermenters segment is expected to grow at a significant rate during the forecast period, as they play a vital role in microbial and cell culture processes used in biologics, vaccines, and recombinant protein production. Increasing demand for high-yield, contamination-free fermentation, along with the rising adoption of single-use and automated fermenter systems, is driving rapid segment growth.
Application Insights
The biopharmaceutical production segment dominated the market, accounting for a 41.55% share in 2025. The increasing adoption of single-use and automated closed processing systems to improve efficiency, ensure regulatory compliance, and reduce batch-failure risks further supported segment growth.
The cell and gene therapy segment is expected to register the fastest CAGR over the forecast period, driven by the rapid expansion of personalized medicine, increasing clinical pipeline of advanced therapies, and the critical need for highly sterile, closed processing environments. The growing adoption of automated, single-use closed systems to ensure contamination control, process consistency, and regulatory compliance is further accelerating segment growth.
End Use Insights
The pharmaceutical & biotech companies segment held the largest share of 53.78% in 2025, largely due to their extensive biologics production activities, strong investments in advanced manufacturing technologies, and high adoption of single-use closed systems to ensure sterility, regulatory compliance, and operational efficiency in large-scale biopharmaceutical manufacturing.
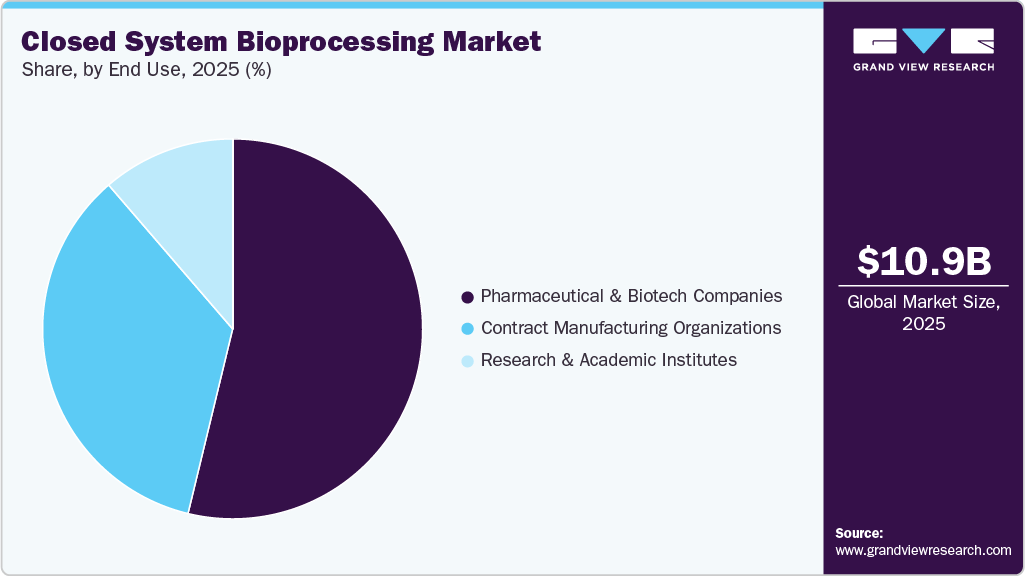
The contract manufacturing organizations (CMOs) segment is expected to register the fastest CAGR over the forecast period, driven by increasing outsourcing of biologics production, rising demand for flexible and cost-efficient manufacturing, and the growing adoption of closed, single-use systems to ensure sterility, scalability, and faster turnaround times.
Regional Insights
The closed system bioprocessing industry in North America accounted for the largest share of 48.31% in 2025, due to its mature biopharmaceutical manufacturing ecosystem, high concentration of biologics pipelines, and early adoption of advanced single-use and automated processing technologies. Strong funding for bioprocess innovation and the rapid commercialization of cell and gene therapies continue to support sustained demand for closed systems.
U.S. Closed System Bioprocessing Market Trends
The U.S. closed system bioprocessing industry represents the significant market, driven by its strong biologics pipeline, high concentration of biopharma manufacturers, and rapid adoption of advanced single-use and automated closed processing technologies.
Europe Closed System Bioprocessing Market Trends
The closed system bioprocessing industry in Europe is a well-established market, driven by expanding biosimilar production, strong academic-industry collaboration, and ongoing modernization of biologics manufacturing facilities. Regulatory focus on quality assurance and process standardization further encourages the transition toward closed processing environments.
The UK closed system bioprocessing industry is supported by a robust life sciences sector, expanding biologics research, and strong government funding for advanced therapy manufacturing. Growth is further driven by increasing investments in flexible, small-batch production capabilities.
The closed system bioprocessing industry in Germany is a key contributor due to its well-established biopharmaceutical production base, strong engineering capabilities, and rising biosimilar manufacturing. Continuous facility modernization is accelerating the shift toward closed processing systems.
Asia Pacific Closed System Bioprocessing Market Trends
The closed system bioprocessing industry in Asia Pacific is projected to grow at the fastest CAGR of 12.08% during the forecast period, supported by rising investments in biomanufacturing capacity, growth of regional CDMOs, and increasing domestic production of vaccines and biologics. Government incentives and cost-competitive production landscapes are accelerating technology adoption across emerging economies.
The China closed system bioprocessing industry is experiencing rapid expansion due to large-scale investments in biologics manufacturing, growing domestic biopharma companies, and increasing adoption of cost-efficient single-use closed technologies.
The closed system bioprocessing industry in Japan is driven by increasing demand for regenerative medicine, supportive regulatory frameworks for advanced therapies, and strong investments in high-quality biomanufacturing infrastructure.
Middle East and Africa Closed System Bioprocessing Market Trends
The closed system bioprocessing industry in the Middle East & Africa is growing steadily, fueled by initiatives to build local pharmaceutical manufacturing capabilities and reduce import dependence. Strategic partnerships, infrastructure upgrades, and gradual growth in biologics demand are contributing to the increasing uptake of closed bioprocessing solutions.
The Kuwait closed system bioprocessing industry is emerging gradually, supported by government initiatives to strengthen pharmaceutical self-sufficiency, rising healthcare investments, and efforts to develop local biomanufacturing capabilities.
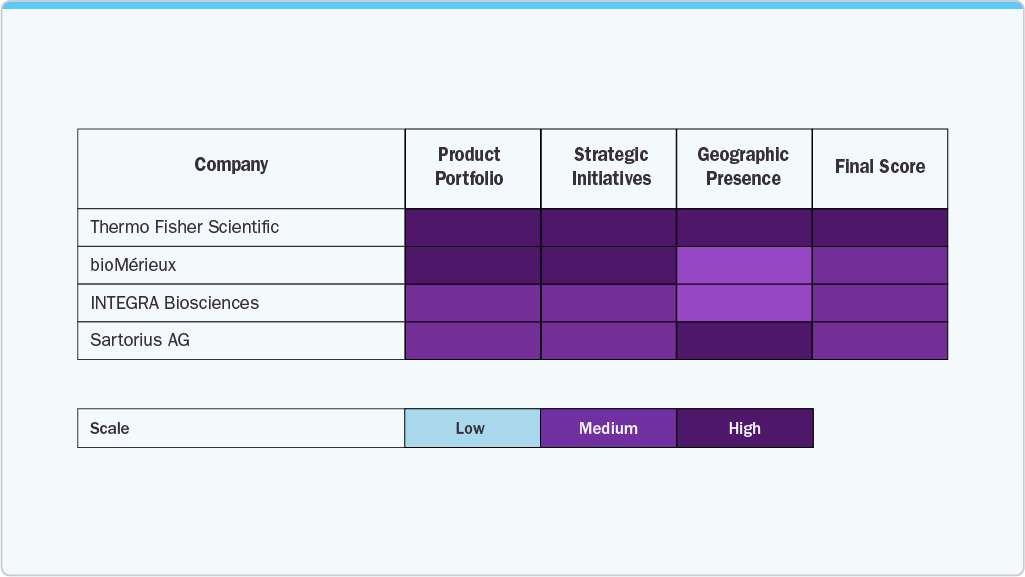
Key Closed System Bioprocessing Company Insights
Key players such as Thermo Fisher Scientific, Sartorius AG, Merck KGaA, GE Healthcare, and Danaher Corporation, among others, hold strong market positions due to extensive bioprocessing portfolios, global manufacturing presence, and continuous innovation in closed and single-use technologies.
Competition is driven by investments in integrated closed processing platforms, automation, digital monitoring, and scalable single-use technologies. Strategic partnerships, capacity expansion, and growing focus on cell and gene therapy manufacturing further intensify market competition.
Key Closed System Bioprocessing Companies:
The following key companies have been profiled for this study on the closed system bioprocessing market.
- Thermo Fisher Scientific
- Sartorius AG
- Merck KGaA
- GE Healthcare (Cytiva)
- Danaher Corporation
- Eppendorf AG
- Lonza Group
- Corning Incorporated
- Pall Corporation
- Applikon Biotechnology
- PBS Biotech
- Miltenyi Biotec
- Fujifilm Diosynth Biotechnologies
Recent Developments
-
In July 2025, Cytiva launched the ÄKTA readyflux TFF system 500, designed to improve low-volume bioprocessing efficiency, scalability, technology transfer, and batch consistency for advanced therapeutic manufacturing applications.
-
In March 2025, Cytiva expanded its Xcellerex portfolio by launching 500-L and 2000-L single-use bioreactors, enhancing scalable, closed, and efficient bioprocessing for advanced therapeutics.
Closed System Bioprocessing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 11.77 billion
Revenue forecast in 2033
USD 21.37 billion
Growth rate
CAGR of 8.90% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
Thermo Fisher Scientific; Sartorius AG; Merck KGaA; GE Healthcare (Cytiva); Danaher Corporation; Eppendorf AG; Lonza Group; Corning Incorporated; Pall Corporation; Applikon Biotechnology; PBS Biotech; Miltenyi Biotec; Fujifilm Diosynth Biotechnologies
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Closed System Bioprocessing Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global closed system bioprocessing market report on the basis of product, application, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Bioreactors
-
Fermenters
-
Mixing Systems
-
Filtration Systems
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopharmaceutical Production
-
Vaccine Manufacturing
-
Cell And Gene Therapy
-
Regenerative Medicine
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biotech Companies
-
Contract Manufacturing Organizations
-
Research & Academic Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The growth is primarily driven by the increasing adoption of single-use technologies, rising biologics and cell & gene therapy production, and the growing need to minimize contamination risks while improving operational efficiency in biomanufacturing processes
b. The global closed system bioprocessing market size was estimated at USD 10.95 billion in 2025 and is expected to reach USD 11.77 billion in 2026.
b. The global closed-system bioprocessing market is expected to grow at a compound annual growth rate of 8.90% from 2026 to 2033 to reach USD 21.37 billion by 2033.
b. North America led the closed system bioprocessing industry in 2025 with the largest share of 48.31%, due to its mature biopharmaceutical manufacturing ecosystem, high concentration of biologics pipelines, and early adoption of advanced single-use and automated processing technologies.
b. Some key players operating in the closed system bioprocessing market include Thermo Fisher Scientific; Sartorius AG; Merck KGaA; GE Healthcare (Cytiva); Danaher Corporation; Eppendorf AG; Lonza Group; Corning Incorporated; Pall Corporation; Applikon Biotechnology; PBS Biotech; Miltenyi Biotec; Fujifilm Diosynth Biotechnologies
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









