- Home
- »
- Biotechnology
- »
-
Cell Therapy Human Raw Materials Market Size Report, 2033GVR Report cover
![Cell Therapy Human Raw Materials Market Size, Share & Trends Report]()
Cell Therapy Human Raw Materials Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Cell Culture Media, Cell Culture Sera, Cell Culture Supplements, Reagents & Buffers), By End Use (CROs & CMOs, Academic & Research Institutions), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-185-7
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
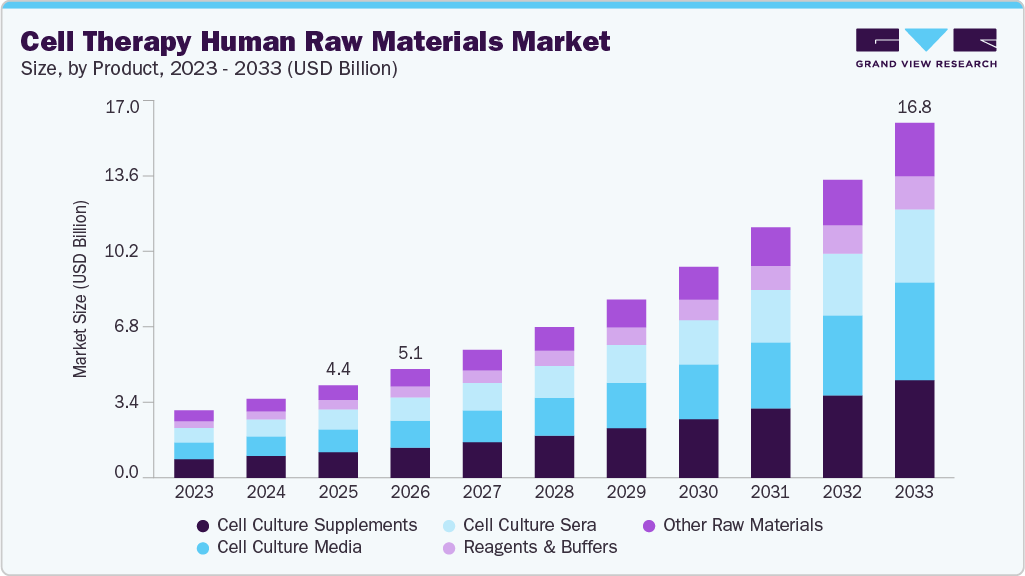
Cell Therapy Human Raw Materials Market Summary
The global cell therapy human raw materials market was size valued at USD 4.37 billion in 2025 and is projected to reach USD 16.79 billion by 2033, expanding at a CAGR of 18.44% from 2026 to 2033. The increasing prevalence of chronic diseases such as cancer, diabetes, & autoimmune disorders, the growing demand for regenerative and personalized medicines, rising R&D activities, and increasing investments are anticipated to boost the cell therapy human raw materials market over the forecast period.
Key Market Trends & Insights
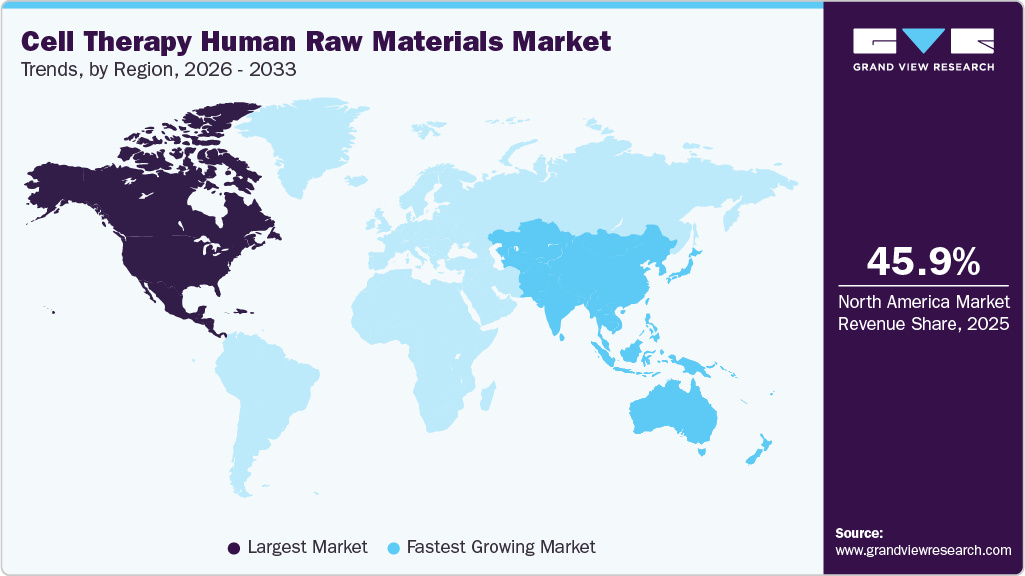
- North America cell therapy human raw materials industry held the largest global market share of 45.98% in 2025.
- The cell therapy human raw materials industry in the U.S. is expected to grow over the forecast period due to the expanding aging population.
- Based on product, the cell culture supplements segment dominated the market with a share of 27.79% in 2025.
- Based on end use, the biopharmaceutical & pharmaceutical companies segment dominated the market in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.37 Billion
- 2033 Projected Market Size: USD 16.79 Billion
- CAGR (2026-2033): 18.44%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Rapid Growth Of The Cell Therapy PipelineRapid growth in the cell therapy pipeline is a key market driver, fueled by rising R&D investments, broader clinical applications, and advances in cell engineering. Expanding programs in oncology, autoimmune, and rare diseases are increasing demand for high-quality human raw materials such as cell culture media, cytokines, growth factors, and viral vectors needed for manufacturing and development.
Cell therapy product pipeline of Fate Therapeutics
Program
CAR / Antigen Target
Indication
Phase
Autoimmunity
FT819 (RMAT)
CD19
Systemic Lupus Erythematosus
Phase 1
Systemic Sclerosis (SSc)
Phase 1
ANCA-associated Vasculitis (AAV)
Phase 1
Idiopathic Inflammatory Myopathies (IIM)
Phase 1
FT839 (NxG)
CD19 / CD38 / CD20
Pan-Indication w/o LLC
Pre-clinical
FT522
CD19 / CD20
Pan-Indication w/o LLC
Phase 1
Oncology
FT825
HER2 / EGFR
Solid Tumor(s)
Phase 1
Undisclosed
Undisclosed
Solid Tumor(s)
Pre-clinical
FT836 (NxG)
MICA/B / EGFR / HER2
Pan-Indication (Solid/Heme) w/o LCC
Phase 1
FT839 (NxG)
CD19 / CD38 / BCMA / GPRC5D
Pan-Indication (Heme) w/o LCC
Pre-clinical
Source: Fate Therapeutics, Secondary Research, Primary Interviews, Grand View Research
The expansion of research targets, which now includes autoimmune diseases and regenerative medicine fields, has led to increased research activity across multiple cell therapy clinical trials worldwide. The development of clinical and commercial therapies requires standardized GMP-grade materials, which can be produced through scalable supply chains that are essential for market expansion.
Expansion Of Regenerative Medicine And Personalized Medicine
The cell therapy human raw materials market depends on the expansion of regenerative medicine and personalized medicine. The rising adoption of patient-specific therapies, which include autologous stem cell treatments and personalized immune cell therapies, drives the need for essential materials such as human serum, cytokines, growth factors, and specialized culture media required for effective cell production.
The development of precision medicine, together with biomarker-based treatment selection and gene-editing technologies, has created therapies that target specific needs in oncology, autoimmune disorders, and degenerative diseases. The healthcare industry is now moving toward personalized treatment methods, which require standardized, GMP-grade materials due to increased cell processing activities.
Market Concentration & Characteristics
The market growth stage is high, and the pace of the cell therapy human raw materials industry growth is accelerating. The market is characterized by a high degree of innovation owing to the rapid technological advancements driven by factors such as the use of CRISPR/Cas9 genome editing and the generation of genetically modified mouse.
M&A activity in the cell therapy human raw materials industry is rising as companies aim to strengthen supply chains, expand portfolios, and secure critical biological inputs. Acquisitions and partnerships focus on vertical integration, improving scalability, quality control, and regulatory compliance to support growing demand for GMP-grade materials from the expanding cell therapy pipeline.
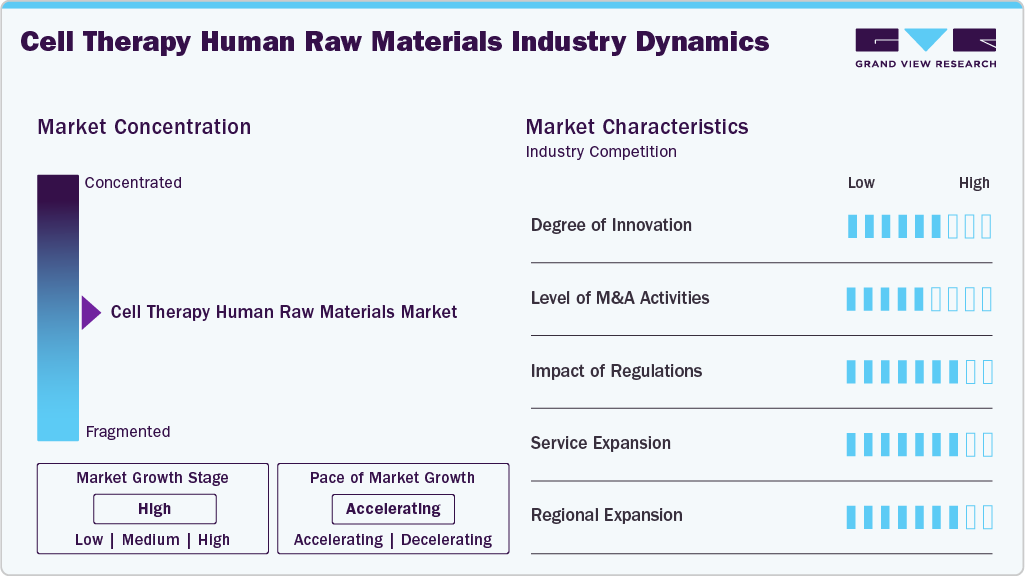
The cell therapy human raw materials industry is also subject to increasing regulatory scrutiny. Several regulatory and government authorities are focusing on establishing stringent guidelines for contamination control, biosafety, cell line authentication, and other factors. The growing focus on cell therapy is expected to continue.
Several companies are expanding their cell therapy -based products. Thus, product expansion in this industry is significant. Companies are including product launches specifically targeting MSCs that are anticipated to drive market growth. For instance, in July 2023, PromoCell GmbH, the company, introduced a new product, PromoExQ MSC Growth Medium XF, for cell therapy manufacturing.
Regional expansion is driving growth in the cell therapy human raw materials industry, supported by rising investments in biomanufacturing facilities, increasing clinical trial activity, and favorable regulatory support across North America, Europe, and the Asia Pacific. This expansion is accelerating demand for high-quality human-derived raw materials used in cell therapy development and manufacturing.
Product Insights
The cell culture supplements segment accounted for the largest share of 27.79% in the cell therapy human raw materials industry in 2025, driven by their critical role in supporting cell expansion, viability, differentiation, and consistent performance across clinical and commercial-scale cell therapy manufacturing workflows. Moreover, numerous market participants are actively exploring untapped opportunities in the market through initiatives such as the development of new products & business expansion.
The cell culture media segment is projected to grow at the fastest CAGR due to its critical role in providing nutrients and growth factors needed for cell survival, proliferation, and scalable manufacturing. Rising investments and product innovations, such as advanced media solutions, are further driving segment growth.
End Use Insights
Based on end use, the market is segmented into biotechnology & pharmaceutical companies, CROs & CMOs, and others. The biopharmaceutical & pharmaceutical companies segment dominated in 2025 with a share of 46.86%, due to rising R&D investments, growing demand for cell-based therapies, and strong technical capabilities. For example, in May 2023, NewBiologix secured USD 50 million to advance gene and cell therapy manufacturing technologies.
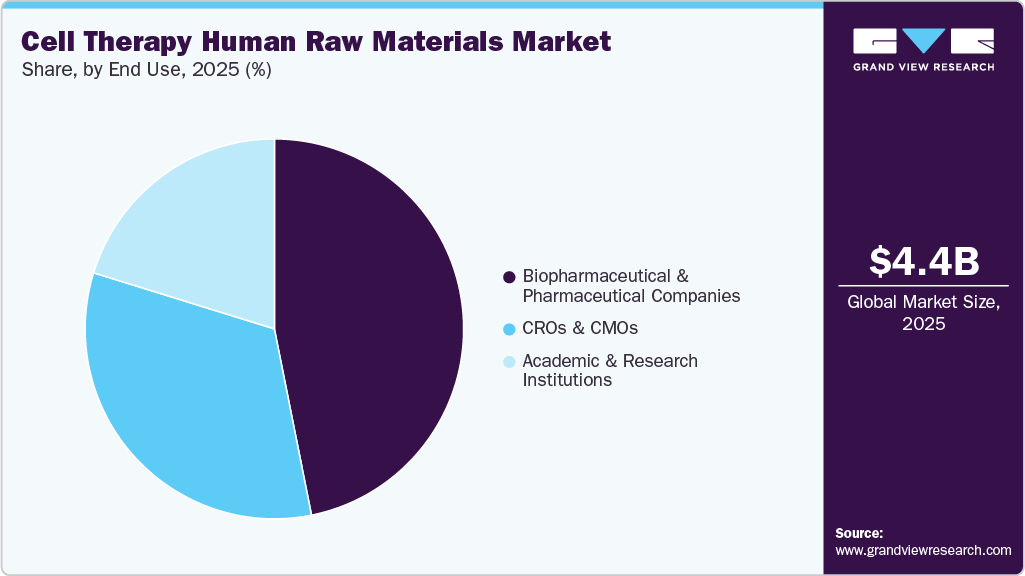
The CROs and CMOs segment is expected to register the fastest CAGR during the forecast period, driven by increasing outsourcing of cell therapy development and manufacturing. CROs support preclinical and clinical research through expertise in trial design, patient recruitment, and regulatory compliance, while CMOs provide cost-effective, specialized manufacturing capabilities for biopharmaceutical companies lacking in-house infrastructure, making them critical partners in the cell therapy value chain.
Regional Insights
North America held the largest share of 45.98% in 2025, driven by supportive regulations, rising demand for advanced therapeutics, and strong initiatives by key players to develop cell-based treatments for chronic diseases. The region also hosts major manufacturers of cell therapy products, increasing demand for human raw materials. In addition, strategic collaborations are intensifying competition; for instance, in January 2023, Kite Pharma, a subsidiary of Gilead Sciences, partnered with Arcellx, Inc. to co-develop CART-ddBCMA for relapsed multiple myeloma.
U.S Cell Therapy Human Raw Materials Market Trends
The U.S. cell therapy human raw materials market is growing due to a strong pipeline of cell and gene therapies, increasing clinical trials, and rising regulatory approvals. Demand is rising for GMP-grade, chemically defined, and xeno-free materials, while investments in biomanufacturing capacity and partnerships between therapy developers and suppliers are further supporting market expansion.
Europe Cell Therapy Human Raw Materials Market Trends
Europe’s cell therapy human raw materials market is expanding steadily, supported by strong regulatory frameworks and increasing investment in advanced treatments. Growth is further driven by rising adoption of high-purity, standardized inputs and the scaling of manufacturing activities as more cell therapies progress toward commercialization.
The UK cell therapy human raw materials industry is driven by a well-established healthcare ecosystem, strong academic-industry collaboration, and expanding CDMO manufacturing capacity. Rising clinical trial activity and increasing investments in precision medicine are boosting demand for key inputs such as culture media, sera, cytokines, and reagents.
The Germany cell therapy human raw materials market is supported by advanced biomanufacturing infrastructure, strong engineering capabilities, and a high concentration of pharmaceutical production facilities. Demand for human raw materials is driven by large-scale CAR-T therapy production, and strict quality standards.
Asia Pacific Cell Therapy Human Raw Materials Market Trends
Asia Pacific is anticipated to witness the fastest CAGR of 18.90% in the market from 2026 to 2033. This is attributed to the increasing focus on biotechnology and the rising demand for advanced cell therapies. Moreover, the key factors contributing to market growth include a surge in R&D activities, strategic collaborations, and the establishment of manufacturing facilities by major players.
The China cell therapy human raw materials industry is a rapidly growing market, driven by strong government funding, expanding domestic manufacturing capacity, a large cell therapy pipeline, active CDMOs, and increasing localization of supply chains boosting demand for human raw materials.
The Japan cell therapy human raw materials market is driven by strong government support for regenerative medicine, particularly iPSC research, along with rising hospital-based manufacturing and clinical use, while longer regulatory timelines and cautious commercialization moderate growth.
MEA Cell Therapy Human Raw Materials Market Trends
The MEA market is emerging, driven by expanding precision medicine initiatives, rising investments in life science infrastructure, and the establishment of research hubs in key countries. Growing reliance on imported high-quality raw materials and international technology partnerships is shaping market development.
The Kuwait cell therapy human raw materials market is at an early stage, with growth mainly driven by increasing healthcare investments and gradual adoption of advanced treatments. Limited domestic manufacturing infrastructure results in high reliance on imports for key materials. However, ongoing efforts to strengthen research capabilities and regional collaborations are expected to support steady long-term market development.
Key Cell Therapy Human Raw Materials Company Insights
The cell therapy human raw materials market comprises global life-science leaders and specialized bioprocess suppliers competing through GMP-grade sourcing, integrated manufacturing, and application-focused portfolios. Major players like Thermo Fisher Scientific, Merck KGaA, Danaher, and Charles River Laboratories dominate due to strong distribution, regulatory expertise, and broad product offerings, while firms such as STEMCELL Technologies, ACROBiosystems, RoosterBio, and PromoCell focus on high-purity, cell-specific formulations for regenerative medicine applications.
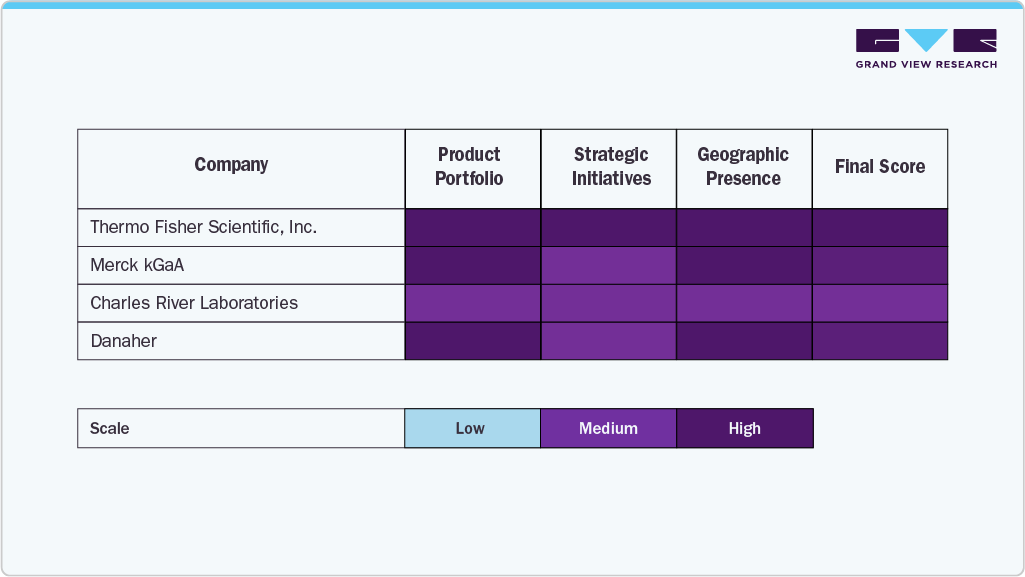
Emerging suppliers such as BioIVT, GeminiBio, Akron Biotech, AllCells, Actylis, Grifols, and CGT Global are enhancing competitiveness through investments in donor-derived materials, traceable supply chains, and scalable GMP production. Market growth is increasingly supported by portfolio diversification, capacity expansion, and strategic partnerships with cell therapy developers.
Key Cell Therapy Human Raw Materials Companies:
The following key companies have been profiled for this study on the cell therapy human raw materialsmarket.
- Thermo Fisher Scientific, Inc
- Merck KGaA
- Actylis
- ACROBiosystems
- STEMCELL Technologies
- Grifols, S.A.
- Charles River Laboratories
- RoosterBio, Inc.
- PromoCell GMBH
- Danaher
- BioIVT
- GeminiBio
- Akron Biotech
- AllCells
- CGT Global
Recent Developments
-
In February 2026, GeminiBio launched its AI-enabled aiMOS media optimization service in the U.S., strengthening cell therapy human raw materials by improving process efficiency, scalability, and critical quality attributes in manufacturing.
-
In August 2025, ProBio Inc. launched GMP AAV manufacturing services in the U.S., strengthening cell and gene therapy supply chains and supporting demand for high-quality raw materials and scalable vector production.
Cell Therapy Human Raw Materials Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 5.14 billion
Revenue forecast in 2033
USD 16.79 billion
Growth rate
CAGR of 18.44% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Products, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
Thermo Fisher Scientific, Inc; Merck KGaA; Actylis; ACROBiosystems; STEMCELL Technologies; Grifols, S.A.; Charles River Laboratories; RoosterBio, Inc.; PromoCell GMBH; Danaher; BioIVT; GeminiBio; Akron Biotech; AllCells; CGT Global
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Cell Therapy Human Raw Materials Market Report Segmentation
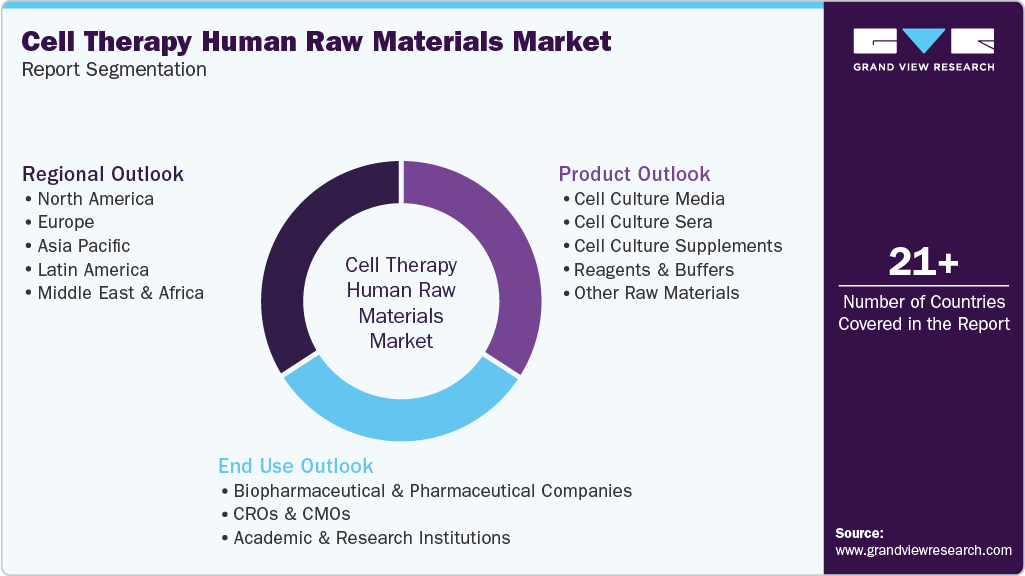
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the cell therapy human raw materials market on the basis of products, end use, and region.
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Cell Culture Media
-
Cell Culture Sera
-
Cell Culture Supplements
-
Proteins
-
Growth Factors
-
Nucleotides
-
Other Supplements
-
-
Reagents & Buffers
-
Other Raw Materials
-
-
End Use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Biopharmaceutical & Pharmaceutical Companies
-
CROs & CMOs
-
Academic & Research Institutions
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
U.K.
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Key factors that are driving the cell therapy human raw materials market growth include increasing investments in cell therapy research and development, growing number of clinical trials, and rising adoption of regenerative and personalized medicines. Furthermore, the growing investments in research & development activities as well as growing number of vaccines & drug development are further projected to impel the growth of the market over the forecast period.
b. The global cell therapy human raw materials market size was estimated at USD 4.37 billion in 2025 and is expected to reach USD 5.14 billion in 2026.
b. The global cell therapy human raw materials market is expected to grow at a compound annual growth rate of 18.44% from 2026 to 2033 to reach USD 16.79 billion by 2033.
b. North America dominated the cell therapy human raw materials market with a share of 45.98% in 2025. This is attributable to the presence of major pharmaceutical & biotechnology companies and growing spending on R&D activities for various biopharmaceutical development.
b. Some of the key players operating in the cell therapy human raw materials market include Danaher Corporation, Thermo Fisher Scientific, Merck KGaA, Grifols, S.A., Charles River Laboratories, and PromoCell GmbH among others.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









