- Home
- »
- Clinical Diagnostics
- »
-
Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market 2033GVR Report cover
![Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Size, Share & Trends Report]()
Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market (2026 - 2033) Size, Share & Trends Analysis By Type (Amyloid-related Markers, Tau-related Markers, Neurodegeneration), By Technology (Next-generation Platforms), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-720-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Summary
The global blood-based biomarker for Alzheimer’s disease diagnostics market size was estimated at USD 169.22 million in 2025 and is projected to reach USD 529.90 million by 2033, growing at a CAGR of 17.37% from 2026 to 2033. A pivotal catalyst was the U.S. FDA’s May 16, 2025, marketing clearance of Fujirebio’s Lumipulse G pTau217/β-amyloid 1-42 plasma ratio-the first Alzheimer’s blood test cleared to aid diagnosis-quickly followed by Labcorp’s nationwide launch on Aug 18, 2025.
Key Market Trends & Insights
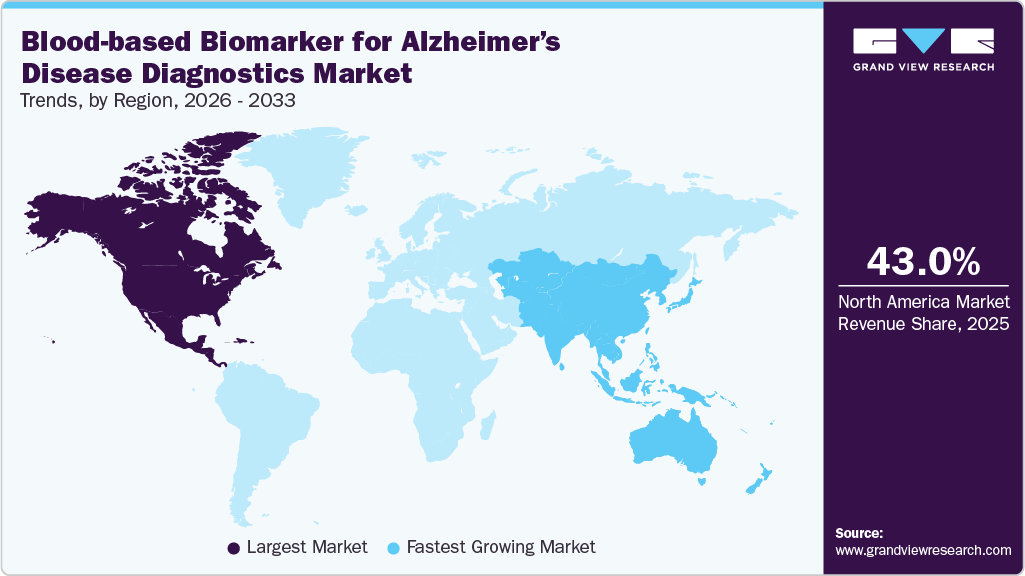
- The North America blood-based biomarker for Alzheimer’s disease diagnostics market accounted for the largest global revenue share of 43.03% in 2025.
- The U.S. blood-based biomarker for Alzheimer’s disease diagnostics industry led North America with the largest revenue share in 2025.
- Based on type, the tau-related markers segment dominated the global market with the largest revenue share of 45.70% in 2025.
- Based on technology, the immunoassays segment dominated the global market with the largest revenue share of 67.84% in 2025.
- Based on end-use, the clinical laboratories & hospital labs segment held the largest revenue share of 52.99% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 169.22 Million
- 2033 Projected Market Size: USD 529.90 Million
- CAGR (2025-2033): 17.37%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
These steps lower adoption friction (regulatory clarity, distribution) and are expected to accelerate payer uptake and clinician ordering, intensifying the growth curve for blood tests relative to imaging and CSF. Guidance presented at AAIC 2025 and published by the Alzheimer’s Association states that blood-based biomarker (BBM) tests with ≥90% sensitivity and ≥75-90% specificity can triage-and at higher performance, substitute for-amyloid PET or CSF testing in appropriate patients. P-tau217-containing panels consistently outperform earlier generations (Aβ42/40 or p-tau181 alone) for detecting amyloid and tau pathology. Real-world product data are converging: Quest’s 2025 AD-Detect™ reported >90% sensitivity and specificity for Alzheimer’s pathology in AAN-presented analyses, while clinical summaries highlight ~92% positive and ~97% negative agreement for the FDA-cleared Lumipulse test. As clinicians gain confidence in rule-in/rule-out accuracy, ordering shifts from specialty memory clinics to broader frontline settings, expanding test volumes, and normalizing BBMs as the first diagnostic step.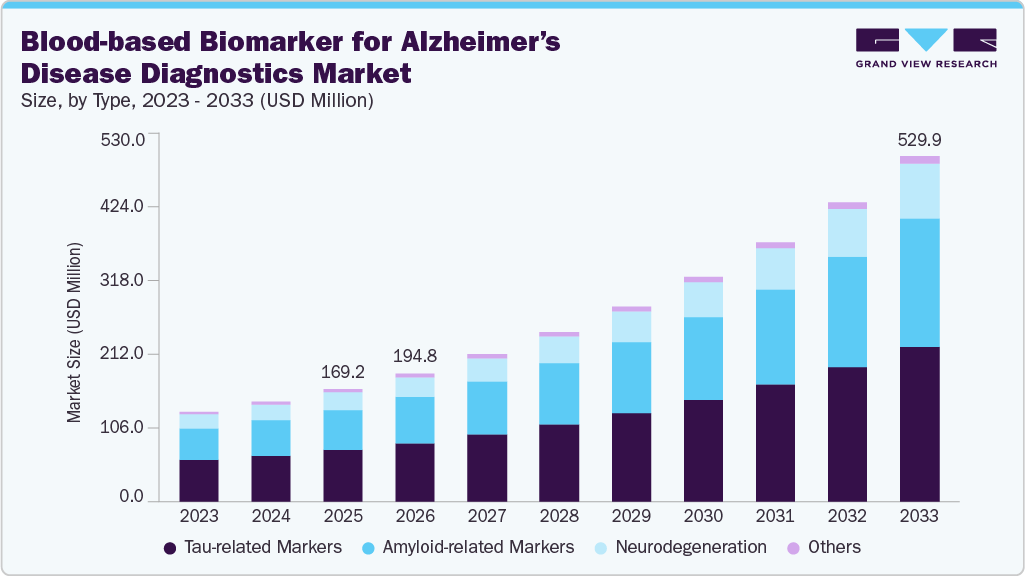
Technology maturation and clinical validation add momentum. Blood assays targeting phosphorylated tau (especially p-tau217) have received Breakthrough Device designations and now full FDA clearance, reflecting strong clinical validity and utility. Academic-industry consortia (e.g., UW-Madison, Johns Hopkins, Lund) supplied pivotal data underpinning the cleared IVD. Published reviews conclude that blood-based biomarker tests (BBMTs) bring value by reducing invasiveness and enabling earlier triage-crucial when disease-modifying therapies are most effective in prodromal/mild stages.
Market traction examples include reference lab launches and hospital adoption of memory-clinic pathways, where a positive p-tau217/Aβ ratio can expedite PET scheduling. At the same time, a negative result can avoid costly scans. As platforms scale, expect menu expansion (neurofilament light, GFAP, Aβ42/40), reflex algorithms, and bundled panels, improving sensitivity/specificity and supporting longitudinal monitoring-opening recurring-test revenue beyond one-time diagnosis.
Market Concentration & Characteristics
The Alzheimer’s blood-based biomarker market is highly innovative, driven by assays measuring p-tau217, Aβ42/40, and neurofilament light. Advances in multiplexing, AI-driven analytics, and early-stage detection are transforming diagnostics. FDA’s first clearance validates clinical reliability, spurring broader adoption and stimulating competitive R&D pipelines among diagnostics and biotech firms.
Mergers and acquisitions increasingly shape the blood-based biomarker for Alzheimer’s disease diagnostics industry, as large diagnostics players acquire startups with proprietary biomarker technologies. Strategic deals expand assay platforms, enhance neurodegenerative disease portfolios, and accelerate commercialization. Partnerships with pharmaceutical firms for companion diagnostics are rising, aligning biomarker testing with disease-modifying therapies and reinforcing end-to-end Alzheimer’s care pathways.
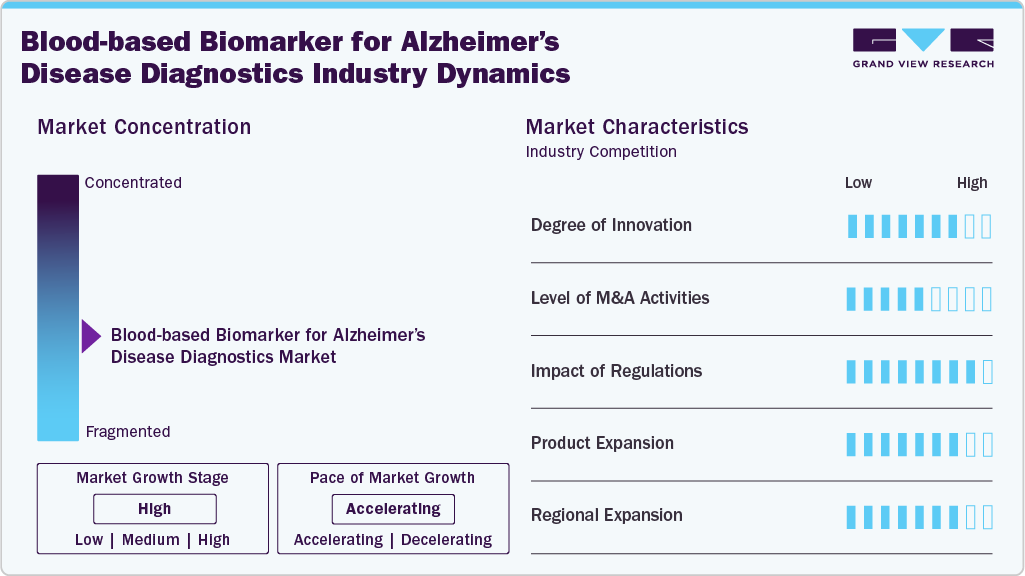
Regulation is pivotal, with FDA clearance of p-tau217/Aβ42 tests marking a breakthrough for clinical adoption. CMS’s conditional coverage and state-level mandates drive payer alignment. Europe’s evolving CE-IVD frameworks support wider market entry. These developments reduce barriers, strengthen physician confidence, and promote standardized diagnostic use in both research and clinical settings.
Companies are broadening test menus beyond amyloid and tau, incorporating markers like GFAP and neurofilament light to enhance diagnostic accuracy and disease staging. Launches include multiplex blood panels, lab-developed tests, and IVD kits. Integration with digital platforms enables longitudinal monitoring, expanding biomarker utility from early detection to therapy response assessment.
North America leads adoption, propelled by FDA approvals and payer policy shifts, while Europe advances via CE-marked tests. Asia-Pacific is projected to show the fastest growth, fueled by aging populations and infrastructure investments. Companies increasingly target China, Japan, and South Korea with localized strategies, clinical collaborations, and partnerships to scale access.
Type Insights
The tau-related markers segment led the blood-based biomarker for Alzheimer’s disease diagnostics market, accounting for the largest share of 45.70% in 2025, and is expected to grow at the fastest CAGR during the forecast period. Phosphorylated tau isoforms-especially p-tau181, p-tau217, and p-tau231-have shown superior sensitivity and specificity in differentiating Alzheimer’s disease from other dementias. Among these, p-tau217 has demonstrated accuracy levels approaching 90%, making it a leading candidate for routine clinical use. In May 2025, the FDA cleared the first p-tau217/Aβ42 plasma ratio test, validating tau biomarkers as clinically reliable diagnostics and establishing a regulatory precedent. Market analysts project this segment to grow at a double-digit CAGR through 2030, driven by the clinical need for less invasive, cost-effective alternatives to PET scans and lumbar punctures. Tau-related assays are also being integrated into multiplex panels with amyloid and neurofilament markers, enhancing staging and monitoring. With ongoing payer discussions and pharma collaborations for therapy monitoring, tau biomarkers are poised to serve as the backbone of blood-based Alzheimer’s diagnostics.
The amyloid-related markers segment is experiencing rapid growth. While amyloid PET imaging remains the gold standard, blood-based amyloid tests are increasingly recognized as cost-effective, minimally invasive pre-screening tools. Recent FDA clearance of assays combining amyloid (Aβ42) with tau ratios underscores amyloid’s enduring diagnostic value. The segment is projected to grow steadily, supported by integration into triage strategies that prioritize patients for confirmatory imaging or lumbar puncture, reducing healthcare costs. Advances in assay sensitivity and standardization are enhancing reproducibility, enabling broader adoption in both clinical trials and early detection programs. As disease-modifying therapies such as anti-amyloid monoclonal antibodies expand, demand for blood-based amyloid testing will accelerate, positioning this segment as a critical component of Alzheimer’s patient identification and therapeutic eligibility pathways.
Technology Insights
Immunoassay held the largest share of 67.84% the blood-based biomarker for Alzheimer’s disease diagnostics industry in 2025, owing to its established use, scalability, and compatibility with clinical workflows. Techniques such as ELISA, chemiluminescent immunoassays, and fully automated immunoassay platforms are widely used to measure key biomarkers, including p-tau217, p-tau181, Aβ42/40, and GFAP, with high sensitivity. FDA clearance of the first Alzheimer’s blood test in 2025, based on an automated immunoassay platform, has validated the technology's reliability for routine diagnostic use. Market growth is fueled by rising adoption in hospital labs, reference laboratories, and pharmaceutical trials where immunoassays offer cost efficiency and throughput advantages over mass spectrometry. Analysts project this segment to expand significantly through 2030, driven by the need for early detection, therapy monitoring, and integration into treatment eligibility pathways. With continuous innovation in ultrasensitive immunoassay platforms, the segment will remain central to expanding accessibility of Alzheimer’s diagnostics worldwide.
The next-generation platforms segment in the blood-based biomarker for Alzheimer’s disease diagnostics market is anticipated to grow at the fastest CAGR over the forecast period. These technologies offer ultra-sensitive detection of low-abundance analytes, enabling precise quantification of p-tau isoforms, Aβ42/40 ratios, and neurofilament light. Mass spectrometry, in particular, is gaining traction as a reference method for assay validation and regulatory submissions. At the same time, Simoa has become a gold standard for research applications and early clinical use. Although currently costlier than immunoassays, next-generation platforms are projected to grow at the fastest CAGR through 2030, fueled by their role in advancing early detection and companion diagnostics for disease-modifying therapies. Pharma collaborations, academic validation studies, and advancements in automation are expanding adoption beyond research into specialized clinical settings. As standardization improves and costs decline, these platforms will transition from niche to mainstream, underpinning precision diagnostics in Alzheimer’s disease.
End-use Insights
Clinical laboratories & hospital labs dominated the market with largest revenue share of 52.99% in 2025, as these facilities are the primary hubs for high-throughput, standardized testing. The 2025 FDA clearance of a p-tau217/Aβ42 immunoassay for symptomatic adults has accelerated adoption in hospital labs, which are now integrating these tests into dementia care pathways as triage tools before PET scans or lumbar punctures. Clinical reference labs are expanding their menus to include Alzheimer’s blood tests, improving access for community physicians and supporting pharmaceutical trials. This segment’s growth is fueled by increasing demand for early diagnosis, therapeutic eligibility testing, and longitudinal monitoring of disease progression. Analysts forecast strong double-digit CAGR for clinical and hospital labs through 2030, supported by automation, reimbursement shifts, and collaborative networks between academic centers and commercial labs. Together, they form the backbone for scaling Alzheimer’s disease diagnostics worldwide.
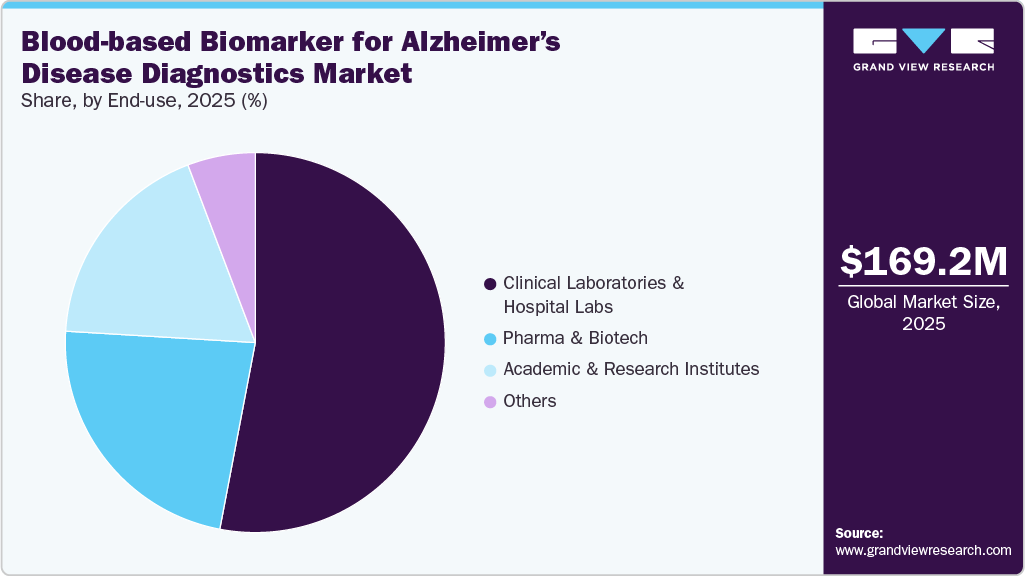
The other segment -comprising diagnostic centers, outpatient clinics, memory care centers, and direct-to-consumer (DTC) testing providers-is expected to grow at the fastest compound annual growth rate (CAGR) over the forecast period. Unlike hospital-based labs, these facilities focus on accessibility and early detection, particularly for at-risk or pre-symptomatic populations. Memory clinics are increasingly adopting blood tests as first-line screening tools, helping prioritize patients for confirmatory PET imaging or CSF analysis. Additionally, DTC companies are entering the space by offering Alzheimer’s risk panels bundled with genetic and wellness testing, appealing to health-conscious consumers. Diagnostic centers benefit from partnerships with pharma companies conducting decentralized clinical trials that require scalable biomarker testing. Although currently smaller in revenue share, this segment is projected to grow at a robust CAGR through 2030, driven by convenience, consumer awareness, and the shift toward community-based precision diagnostics outside traditional hospital settings.
Regional Insights
The North America blood-based biomarker for Alzheimer’s disease diagnostics market accounted for the largest global revenue share of 43.03% in 2025. The North America blood-based biomarker for Alzheimer’s disease diagnostics market is experiencing strong growth, due to early regulatory approvals, high healthcare spending, and widespread adoption of advanced diagnostics. The FDA’s 2025 clearance of a blood-based p-tau217/Aβ42 test has legitimized its clinical use and boosted adoption across labs. Strong pharmaceutical pipelines and ongoing clinical trials further support demand, as biomarker testing becomes critical for therapy eligibility. Growing Alzheimer’s prevalence, favorable reimbursement pathways, and integration into memory care programs add momentum.
U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Trends
The U.S. blood-based biomarker for Alzheimer’s disease diagnostics industry led North America with the largest revenue share in 2025. The U.S. Blood-based biomarker for Alzheimer’s disease diagnostics industry is growing, fueled by an advanced healthcare infrastructure and the highest adoption of FDA-approved Alzheimer’s diagnostics. Rising patient demand for non-invasive testing and CMS policies linking biomarker use with anti-amyloid therapy coverage are key growth drivers. Expanding clinical lab networks, payer discussions, and academic-industry collaborations also foster rapid scale-up.
Europe Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Trends
Europe is experiencing strong growth in the blood-based biomarker for Alzheimer’s disease diagnostics industry, driven by CE-IVD approvals, pan-European research consortia, and structured dementia screening programs. Governments emphasize early detection to reduce long-term healthcare costs. Adoption is accelerating in specialized clinics and research hubs, particularly as biomarkers are integrated into clinical trial protocols. Harmonization efforts across the EU further promote consistent implementation.
The UK blood-based biomarker for Alzheimer’s disease diagnostics market is growing rapidly. The UK market benefits from the NHS’s focus on early dementia diagnosis, combined with academic leadership in Alzheimer’s biomarker research. National dementia strategies encourage integration of innovative diagnostics. Collaborations between universities, biotech firms, and memory clinics foster adoption. Government funding and patient advocacy groups also support broader implementation and accessibility.
Germany’s blood-based biomarker for Alzheimer’s disease diagnostics market is witnessing progressive growth, supported by a strong diagnostics industry, advanced lab infrastructure, and public health emphasis on early dementia care. High adoption in hospital-based labs and memory clinics drives usage. Robust collaborations among academia, pharma, and diagnostics firms, along with the availability of CE-marked assays, strengthen clinical acceptance and scalability.
Asia Pacific Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Trends
The Asia Pacific blood-based biomarker for Alzheimer’s disease diagnostics industry is growing rapidly, fueled by rapidly aging populations, rising dementia incidence, and increasing healthcare investments. Governments in Japan, China, and South Korea are prioritizing early detection programs. Expanding lab infrastructure, growing awareness, and multinational clinical trial activity accelerate adoption. Local manufacturing and partnerships aid cost-effective test deployment.
The blood-based biomarker for Alzheimer’s disease diagnostics market in Japan is expected to grow during the forecast period. Japan is gradually embracing Blood-based biomarkers for Alzheimer’s disease diagnostics, driven by its aging society and the government's commitment to dementia care innovation. Strong academic research, integration of biomarkers into dementia guidelines, and widespread adoption of advanced diagnostics fuel growth. Collaborations with global firms ensure rapid technology transfer, supporting early clinical use nationwide.
China’s blood-based biomarker for Alzheimer’s disease diagnostics market is expanding rapidly, propelled by government-backed aging initiatives, the rapid expansion of diagnostic labs, and rising Alzheimer’s prevalence. Domestic biotech companies are investing in amyloid and tau biomarker platforms, while global firms partner locally to enter the market. Affordability-focused innovation and large patient pools provide substantial opportunities for scaling adoption.
Latin America Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Trends
Latin America is experiencing moderate to high growth, driven by rising awareness, urbanization, and increasing dementia prevalence. Countries like Brazil and Mexico are improving their healthcare infrastructure and participating in multinational Alzheimer’s trials. Private diagnostic centers and memory clinics drive adoption, while international partnerships help bridge technological and cost barriers.
Middle East and Africa Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Trends
Middle East and Africa shows strong potential for long-term growth of blood-based biomarkers for Alzheimer’s disease diagnostics. Rising life expectancy and a growing elderly population are increasing the prevalence of Alzheimer’s across the region. Governments, particularly in Gulf countries like Saudi Arabia and the UAE, are investing heavily in healthcare modernization and digital health infrastructure, creating opportunities for the adoption of advanced diagnostic tools. International collaborations with European and U.S. research institutions are fostering early clinical use, while multinational diagnostic companies are entering the region through partnerships. Although limited awareness, affordability issues, and infrastructure gaps remain barriers in parts of Africa, expanding private healthcare, memory clinics, and telemedicine platforms are expected to gradually drive the biomarker uptake and market expansion.
Key Blood-based Biomarker For Alzheimer’s Disease Diagnostics Company Insights
The blood-based biomarker for Alzheimer’s disease diagnostics market features several key players driving innovation and adoption. Leading companies include Fujirebio, Roche Diagnostics, Labcorp, Quest Diagnostics, C2N Diagnostics, Quanterix, Siemens Healthineers, Johnson & Johnson, Abbott Laboratories, BioArctic, Araclon Biotech, and Diadem. These firms are investing heavily in immunoassays, multiplex platforms, and next-generation biomarker technologies. Strategic collaborations, regulatory clearances, and clinical trial support strengthen their market presence, while expanding test portfolios and regional footprints enable them to meet rising demand for non-invasive, early Alzheimer’s diagnostics.
Key Blood-based Biomarker For Alzheimer’s Disease Diagnostics Companies:
The following key companies have been profiled for this study on the blood-based biomarker for Alzheimer’s disease diagnostics market.
- F. Hoffmann-La Roche Ltd
- Quanterix
- Fujirebio
- C2N Diagnostics
- Labcorp
- Quest Diagnostics Incorporated
- Siemens Healthineers
- Lilly USA, LLC
- Abbott
- ALZpath
Recent Developments
-
In January 2026, Roche Diagnostics and BGI Genomics rolled out blood-based Alzheimer’s diagnostic tests, including Roche’s pTau181 assay, in China, with initial clinical use at a hospital in Hainan and efforts underway for broader availability.
-
In December 2025, Siemens Healthineers signed a licensing agreement with ALZpath to use ALZpath’s proprietary pTau217 antibody in the development of a pTau217 in vitro diagnostic assay for use on Siemens’ Atellica immunoassay systems, to expand global access to blood-based Alzheimer’s testing.
-
In May 2025, Fujirebio announced that the U.S. Food and Drug Administration (FDA) had granted 510(k) clearance for its Lumipulse G pTau 217/β-Amyloid 1-42 Plasma Ratio in-vitro diagnostic (IVD) test. Designed to assess amyloid pathology in individuals being evaluated for Alzheimer’s disease or other causes of cognitive decline, the test had previously received Breakthrough Device Designation from the FDA. It now becomes the first FDA-cleared blood-based IVD test in the U.S. to support the identification of patients with amyloid pathology linked to Alzheimer’s disease (AD).
-
In July 2025, at the Alzheimer’s Association International Conference (AAIC) in Toronto, the Alzheimer’s Association unveiled its first clinical practice guideline (CPG) for the diagnosis, treatment, and care of Alzheimer’s disease and related dementias. This initial guideline emphasizes the use of blood-based biomarker (BBM) tests by specialists to evaluate Alzheimer’s pathology in individuals with cognitive impairment. Developed as part of the Association’s ALZPro initiative-a centralized hub for dementia professionals-the guideline provides evidence-based resources to support earlier and more accurate diagnosis, ensuring timely access to appropriate treatment.
-
In May 2025, researchers at the Keck School of Medicine of USC introduced a new blood test capable of detecting early indicators of Alzheimer’s disease by measuring proteins associated with the condition. Called the Penta-Plex Alzheimer’s Disease Capture Sandwich Immunoassay (5ADCSI), the test simultaneously measures five biomarkers-more than current blood tests-and is compatible with laboratory equipment already widely in use. A proof-of-concept study, supported by the National Institutes of Health, was recently published in the Journal of Alzheimer’s Disease
-
In April 2024, Labcorp, a global leader in innovative and comprehensive laboratory services, announced the nationwide rollout of its pTau-217/Beta Amyloid 42 Ratio test. This advanced blood-based biomarker assay is designed to support the diagnosis of Alzheimer’s disease and further strengthens Labcorp’s expanding portfolio of blood-based diagnostics for Alzheimer’s and other dementias.
Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 194.78 million
Revenue forecast in 2033
USD 529.90 million
Growth rate
CAGR of 17.37% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, technology, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd; Quanterix; Fujirebio; C2N Diagnostics; Labcorp; Quest Diagnostics Incorporated; Siemens Healthineers; Lilly USA, LLC; Abbott; ALZpath
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
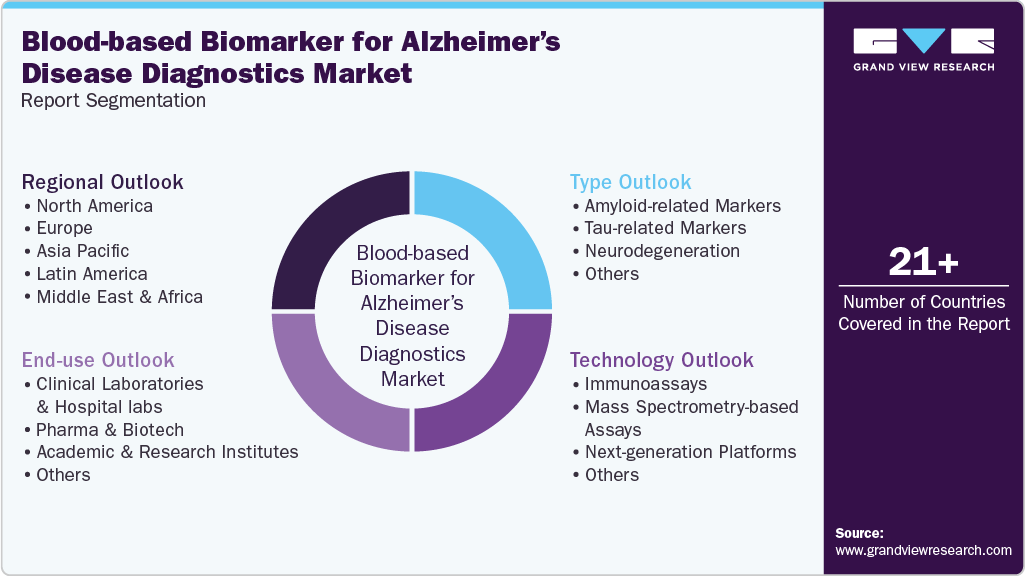
Global Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global blood-based biomarker for Alzheimer’s disease diagnostics market report based on type, technology, end-use, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Amyloid-related markers
-
Tau-related markers
-
Neurodegeneration
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Immunoassays
-
Mass spectrometry-based assays
-
Next-generation platforms
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical laboratories & hospital labs
-
Pharma & biotech
-
Academic & research institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the blood-based biomarker for Alzheimer’s disease diagnostics market includeF. Hoffmann-La Roche Ltd, Quanterix, Fujirebio, C2N Diagnostics, Labcorp, Quest Diagnostics Incorporated, Siemens Healthineers, Lilly USA, LLC, Abbott, and ALZpath
b. Key factors that are driving the market growth include technology maturation and clinical validation and early regulatory approvals, high healthcare spending, and widespread adoption of advanced diagnostics.
b. The global blood-based biomarker for Alzheimer’s disease diagnostics market size was estimated at USD 169.22 million in 2025 and is expected to reach USD 194.78 million in 2026.
b. The global blood-based biomarker for Alzheimer’s disease diagnostics market is expected to grow at a compound annual growth rate of 17.37% from 2026 to 2033 to reach USD 529.90 million by 2033.
b. On the basis of type, tau-related markers accounted for the largest market share of45.70% in 2025 and expected fastest growth in forecast year.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









