- Home
- »
- Clinical Diagnostics
- »
-
At-Home STI Testing Market Size, Industry Report, 2033GVR Report cover
![At-Home STI Testing Market Size, Share & Trends Report]()
At-Home STI Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Infection (HIV, CT), By Test Type (Molecular/NAAT-based Kits, Multiplex Panel Testing), By Sample Type, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-863-3
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
At-Home STI Testing Market Summary
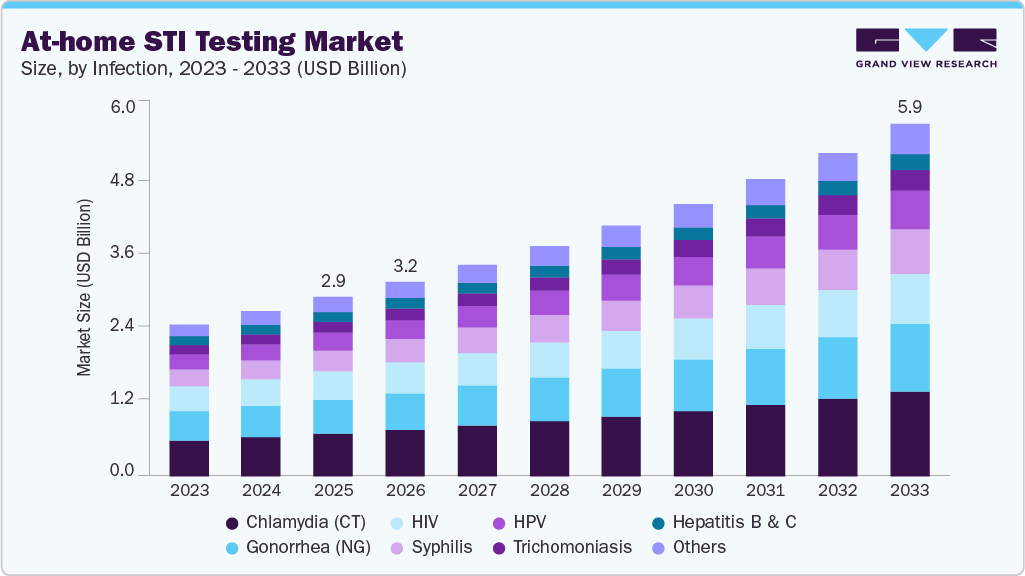
The global at-home STI testing market size was estimated at USD 2.98 billion in 2025 and is projected to reach USD 5.86 billion by 2033, growing at a CAGR of 8.85% from 2026 to 2033. The rising prevalence of sexually transmitted infections (STIs) such as chlamydia, gonorrhea, syphilis, and HIV, along with increasing awareness and demand for private, convenient testing solutions, is significantly driving market growth.
Key Market Trends & Insights
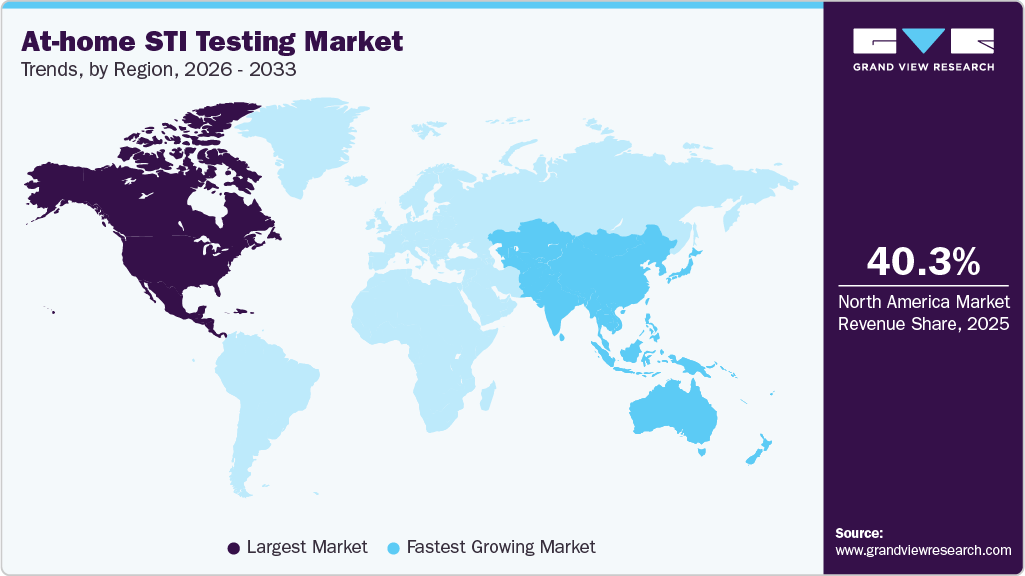
- North America at-home STI testing market dominated the global market and accounted for the largest revenue share of 40.27% in 2025.
- The U.S. led the North American market and held the largest revenue share in 2025.
- Based on infection, the chlamydia (CT) segment dominated the global market and accounted for the largest revenue share of 23.83% 2025.
- Based on test type, the rapid lateral flow immunoassays segment held the largest revenue share of 34.69% in 2025.
- Based on sample type, the urine segment held the largest revenue share of 31.37% in 2025.
- Based on distribution channel, the online pharmacies & D2C platforms segment held the largest revenue share of 45.89% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.98 Billion
- 2033 Projected Market Size: USD 5.86 Billion
- CAGR (2026-2033): 8.85%
- North America: Largest market in 2025
- Asia pacific: Fastest growing market
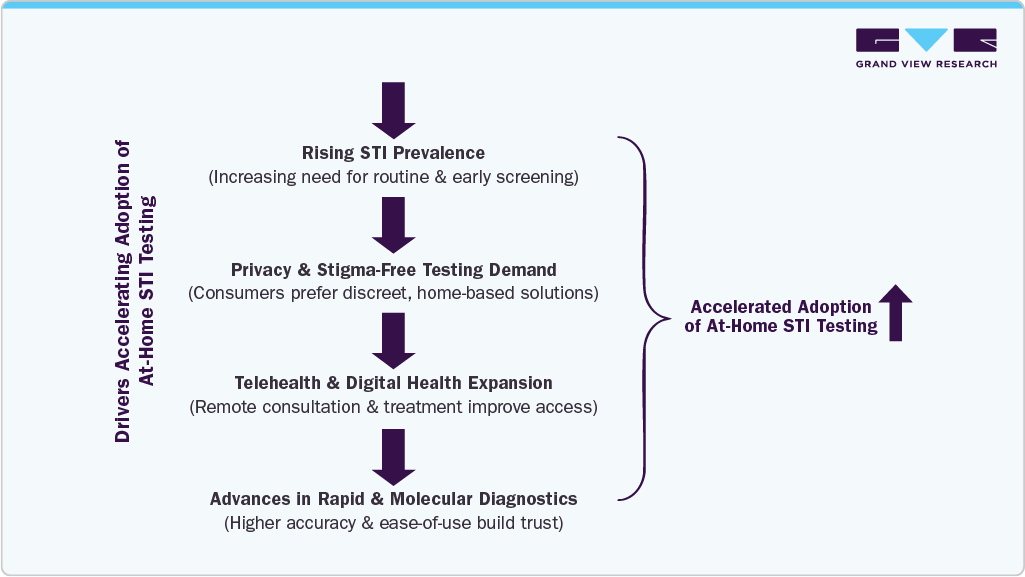
According to the World Health Organization, more than 1 million STIs are acquired every day worldwide, while the Centers for Disease Control and Prevention reports over 2.4 million cases of chlamydia, gonorrhea, and syphilis annually in the U.S. If left undiagnosed and untreated, STIs can lead to severe reproductive health complications, infertility, adverse pregnancy outcomes, and increased HIV transmission risk, underscoring the critical need for accessible screening solutions such as at-home STI testing.The increasing number of regulatory clearances for fully at-home and over-the-counter STI diagnostic solutions is significantly accelerating market adoption. In March 2025, the U.S. Food and Drug Administration granted marketing authorization to Visby Medical’s Women’s Sexual Health Test, the first fully at-home diagnostic capable of detecting chlamydia, gonorrhea, and trichomoniasis with results in approximately 30 minutes. This milestone marks a major shift toward consumer-operated STI diagnostics, enabling rapid detection without laboratory visits and expanding access to accurate home-based testing.
Advancements in rapid self-testing technologies are further improving accessibility and speed of diagnosis. In October 2025, the FDA approved the INSTI HIV Self-Test from bioLytical Laboratories, a finger-stick blood test delivering results in about one minute, significantly reducing testing barriers and enabling immediate status awareness. In addition, continued availability of oral-fluid rapid tests such as OraQuick supports broader HIV screening uptake through painless, user-friendly self-testing options. These innovations enhance convenience and privacy, further driving consumer adoption.
Technological innovation in molecular and multiplex STI testing is expanding the clinical reliability and scope of home diagnostics. New home-collected molecular tests enable simultaneous detection of multiple infections from a single specimen, improving screening efficiency and patient compliance. For example, multiplex STI testing solutions capable of detecting infections such as chlamydia, gonorrhea, trichomoniasis, and Mycoplasma genitalium from one sample are being deployed through telehealth-enabled care pathways, improving comprehensive sexual health screening and follow-up care. The expansion of multiplex testing is increasing diagnostic accuracy while enhancing the value proposition of at-home screening.
Public health initiatives and digital health distribution programs are also expanding access to home STI testing among high-risk and underserved populations. Programs such as the CDC-supported Together TakeMeHome initiative have distributed hundreds of thousands of free at-home HIV self-tests across the U.S., improving early detection and reaching first-time testers. In parallel, governments and healthcare systems increasingly recognize home testing as a critical strategy to improve screening rates, reduce stigma barriers, and support early treatment. These initiatives are strengthening the role of at-home STI testing in preventive healthcare and disease control strategies.
However, factors such as concerns regarding diagnostic accuracy, improper sample collection, and user interpretation errors can affect test reliability, particularly for self-administered rapid tests, which are anticipated to restrain market growth. Reimbursement limitations and out-of-pocket costs may restrict adoption in price-sensitive markets, especially where public health programs primarily support clinic-based screening. Privacy concerns related to digital result reporting and telehealth integration also continue to influence consumer trust. Furthermore, variability in regulatory frameworks and quality standards across countries creates barriers for manufacturers seeking global expansion.
Market Concentration & Characteristics
The degree of innovation in the at-home STI testing industry is high, driven by advancements in molecular diagnostics, rapid self-testing technologies, and digital health integration. In June 2025, Visby Medical received U.S. FDA authorization for the first fully at-home PCR test capable of detecting chlamydia, gonorrhea, and trichomoniasis, marking a major breakthrough in consumer-operated molecular diagnostics. In addition, improvements in self-collection kits and app-guided workflows are enhancing usability and result interpretation. The shift toward multiplex testing panels and smartphone-enabled diagnostics is further improving diagnostic accuracy and consumer confidence, accelerating the adoption of home-based sexual health screening.
Mergers, partnerships, and strategic collaborations in the at-home STI testing ecosystem are moderate to high, reflecting growing interest in integrated digital care and direct-to-consumer diagnostics. Between 2024 and 2025, telehealth platforms expanded collaborations with diagnostic laboratories to offer seamless testing, virtual consultations, and treatment pathways. These partnerships enable end-to-end care delivery, improve treatment follow-up rates, and strengthen consumer engagement. As digital health platforms scale globally, strategic alliances are expected to increase, further consolidating the market and enhancing service accessibility.
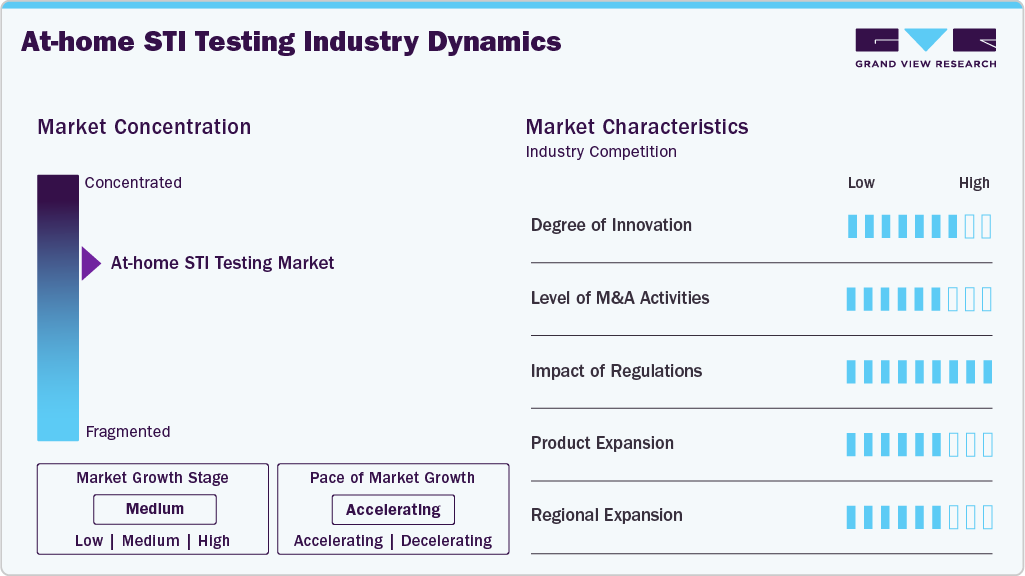
The impact of regulations on the at-home STI testing industry is high, as approvals and compliance standards directly influence product availability and consumer trust. In 2024-2025, regulatory agencies, including the U.S. Food and Drug Administration, expanded authorization pathways for over-the-counter and home-use STI diagnostics, enabling broader access to reliable self-testing solutions. Regulatory frameworks increasingly require usability validation and performance accuracy studies to ensure safe self-administration.
Product expansion in the at-home STI testing industry is high, with manufacturers and digital health providers introducing comprehensive and multiplex screening solutions. In 2024 and 2025, providers expanded home testing panels to include infections such as chlamydia, gonorrhea, syphilis, HIV, trichomoniasis, and hepatitis, enabling consumers to screen for multiple infections from a single sample. Subscription-based screening services and bundled telehealth care pathways are also gaining traction, improving convenience and encouraging routine testing. These expanded offerings enhance clinical utility while increasing revenue per test.
Regional expansion of at-home STI testing is moderate, with strong growth in developed markets and emerging adoption in underserved regions. In 2024 - 2025, public health programs in North America and Europe expanded the distribution of free HIV self-tests and home sample collection kits targeting high-risk populations. Meanwhile, pilot programs in the Asia-Pacific and parts of Africa are exploring self-testing solutions to improve early detection and reduce stigma barriers. While adoption is accelerating globally, regulatory variability, affordability challenges, and awareness gaps continue to influence uptake in emerging markets.
Infection Insights
Chlamydia (CT) dominated the market and accounted for the largest revenue share of 23.83% in 2025. The segment’s dominance is primarily attributed to the high prevalence of chlamydia infections globally and strong screening recommendations for sexually active populations. Chlamydia remains one of the most commonly reported bacterial sexually transmitted infections worldwide, particularly among individuals aged 15-24, which necessitates routine screening and repeat testing. According to the WHO, hundreds of millions of new curable STI infections occur each year, with chlamydia representing a substantial share of reported cases. In addition, clinical guidelines in major markets recommend routine chlamydia screening for sexually active women and high-risk populations, contributing to high testing volumes. The increasing availability of home sample collection kits and multiplex STI panels that include chlamydia as a standard component is further supporting segment growth.
HPV is projected to experience the fastest growth rate of 10.45% during the forecast period. Increasing emphasis on cervical cancer prevention and the global shift toward self-collection screening methods are key factors driving growth in this segment. HPV is recognized as the primary cause of cervical cancer, prompting health authorities to expand screening coverage and improve early detection. Self-collected vaginal sampling has gained clinical acceptance as an effective alternative to clinician-collected samples and improves participation among women who avoid clinic-based screening. In November 2025, multiple European screening programs expanded HPV self-sampling initiatives to improve participation and early detection rates. Furthermore, the WHO continues to promote HPV screening as a central pillar of its cervical cancer elimination strategy, supporting increased adoption of home-based screening solutions.
Test Type Insights
In 2025, the rapid lateral flow immunoassays segment accounted for the largest market share of 34.69%. The segment’s growth is primarily driven by the widespread use of rapid self-tests for infections such as HIV and syphilis, where immediate results and ease of use are critical for early detection and timely care. Lateral flow tests offer advantages, including affordability, minimal user training, and rapid turnaround times, making them well-suited for home use and large-scale screening initiatives. Their accessibility has supported expanded testing among first-time users and high-risk populations. According to the CDC, increasing adoption of HIV self-testing has improved screening uptake among individuals who may not otherwise access traditional testing services. These factors continue to reinforce the segment’s dominant position.
Multiplex panel testing is likely to grow with the fastest CAGR of 9.56% over the forecast period. The segment’s growth is driven by rising consumer preference for comprehensive sexual health screening and the clinical advantages of detecting multiple infections from a single specimen. Multiplex panels enable simultaneous testing for infections such as chlamydia, gonorrhea, trichomoniasis, HIV, and hepatitis, improving diagnostic efficiency and patient compliance. In 2024, the UK’s National Health Service expanded digital sexual health services that provide home-based multi-infection STI testing kits ordered online, reflecting growing demand for comprehensive screening solutions. As awareness of preventive sexual health increases and consumers seek convenient single-test options, multiplex panel testing is expected to experience sustained growth.
Sample Type Insights
In 2025, the urine segment led the market, accounting for 31.37% of the revenue share. The segment’s dominance is driven by the ease of self-collection, high user acceptability, and strong clinical suitability for detecting infections such as chlamydia and gonorrhea. Urine-based testing is particularly preferred for male screening and is widely included in home sample collection kits, improving participation among individuals reluctant to undergo swab-based testing. Public health screening programs and telehealth providers frequently recommend urine-based collection due to its non-invasive nature and reliability when used with molecular testing methods. The convenience and accuracy of urine sampling continue to support its widespread adoption and leading market position.
The vaginal swab segment is expected to be the fastest-growing segment over the forecast period, with a projected CAGR of 9.97%. Growth in this segment is driven by increasing adoption of self-collected vaginal sampling for detecting infections such as HPV, chlamydia, gonorrhea, and trichomoniasis. Clinical studies have demonstrated that self-collected vaginal swabs provide diagnostic accuracy comparable to clinician-collected samples, improving screening participation among women who may avoid clinic visits. In 2024, national screening programs in several European countries expanded the use of self-sampling kits to improve cervical cancer and STI screening coverage. As awareness increases and self-collection becomes more widely accepted, vaginal swab sampling is expected to witness sustained growth.
Distribution Channel Insights
The online pharmacies & D2C platforms segment led the market with a share of 45.89% in 2025 and is anticipated to grow at the fastest growth rate over the forecast period. The segment’s growth is driven by increasing consumer preference for discreet purchasing, home delivery convenience, and integrated telehealth services. Online platforms enable users to order test kits, receive digital results, and access virtual consultations and treatment options, reducing stigma and improving follow-up care. The rapid expansion of digital health ecosystems and subscription-based testing services has further strengthened direct-to-consumer distribution models. As privacy concerns and convenience remain key drivers, online and D2C platforms continue to be the primary growth engine of the market.
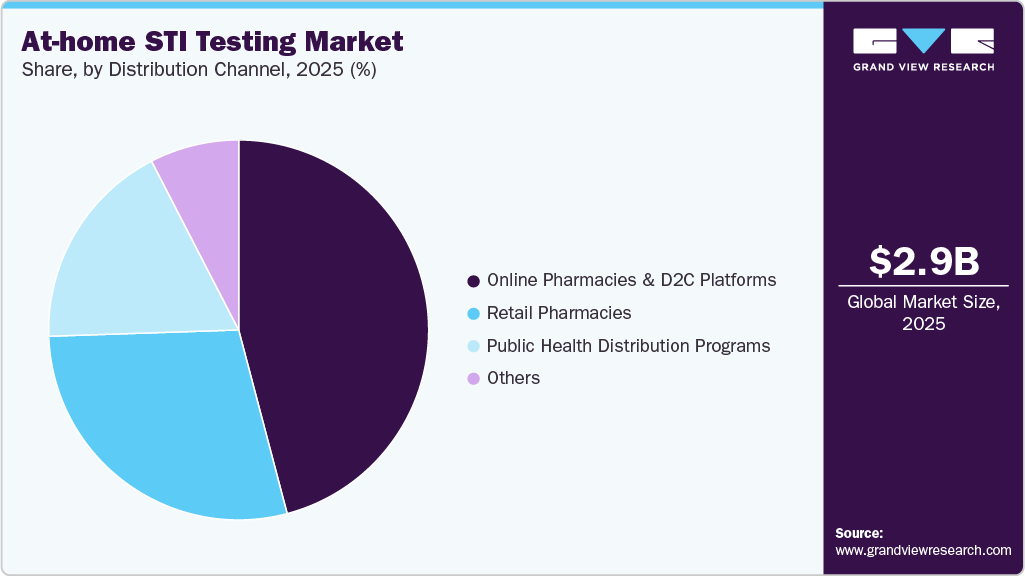
The retail pharmacies segment is expected to grow at a lucrative rate over the forecast period. Growth is supported by expanding over-the-counter availability of self-test kits and the trusted role of pharmacies as accessible healthcare touchpoints. Retail pharmacies provide immediate access to testing products without delivery wait times and often serve as the first point of care for consumers seeking discreet health solutions. In 2025, several major pharmacy chains in North America and Europe expanded shelf availability of HIV self-tests and home STI screening kits, reflecting rising consumer demand. As regulatory approvals increase OTC availability and pharmacies enhance private consultation services, the retail pharmacy channel is expected to experience steady growth.
Regional Insights
North America at-home STI testing market dominated the overall global market and accounted for 40.27% of the revenue share in 2025. Growth in the region is supported by rising STI incidence rates, strong public health screening initiatives, and high adoption of digital health solutions. Well-established healthcare infrastructure and widespread telehealth integration have accelerated consumer acceptance of home-based testing. The presence of major diagnostic manufacturers and direct-to-consumer testing providers further strengthens regional expansion.
U.S. At-home STI Testing Market Trends
The at-home STI testing market in the U.S. held the largest share in the North America market in 2025, driven by a large target population, strong consumer awareness, and the presence of key diagnostic and digital health companies. The country continues to invest heavily in preventive healthcare and digital health innovation. In 2025, the National Institutes of Health supported research initiatives focused on improving access to decentralized diagnostic technologies, including home-based infectious disease testing. In addition, increasing telehealth adoption and growing consumer preference for discreet testing solutions are expected to support sustained market growth.
Europe At-home STI Testing Market Trends
The at-home STI testing market in Europe is experiencing significant growth due to rising STI incidence, expanding preventive screening programs, and increasing acceptance of self-collection diagnostics. Many European countries are strengthening sexual health screening initiatives to improve early detection and reduce transmission rates. Government-backed digital health services and online ordering systems are improving access to home STI testing kits. Growing emphasis on preventive healthcare and privacy-focused testing solutions continues to drive adoption across the region.
The UK at-home STI testing market is expanding due to strong public health support and the widespread availability of digital sexual health services. The National Health Service (NHS) has integrated online STI testing services that allow individuals to order home sample collection kits and receive results remotely, improving screening accessibility. In 2024, NHS-supported digital sexual health programs expanded coverage to reach underserved populations and improve testing participation. Increasing awareness, convenience, and confidentiality are key factors driving growth in the UK.
The at-home STI testing market in Germany is growing steadily, supported by its advanced healthcare infrastructure and increasing adoption of preventive screening practices. The country’s strong laboratory network and digital health initiatives facilitate the integration of home sample collection services with clinical diagnostic workflows. Growing awareness of sexual health screening and increased acceptance of self-collection methods are contributing to market expansion.
Asia Pacific At-home STI Testing Market Trends
The at-home STI testing market in the Asia Pacific is projected to witness the fastest growth, driven by improving healthcare infrastructure, expanding digital health access, and increasing awareness of sexual health screening. Rising STI prevalence in densely populated countries and growing smartphone penetration are enabling wider adoption of telehealth-enabled testing services. Governments and health organizations in the region are exploring self-testing solutions to improve screening coverage and reduce stigma-related barriers.
China at-home STI testing market is growing due to rapid advancements in biotechnology, expanding digital healthcare platforms, and increasing consumer adoption of home diagnostics. Significant investments in healthcare modernization and diagnostic technologies are improving access to preventive screening services. The integration of online healthcare platforms with diagnostic services is enabling broader access to discreet STI testing solutions.
Latin America At-home STI Testing Market Trends
The at-home STI testing market in Latin America is experiencing steady growth due to increasing awareness of sexual health, expanding telehealth services, and improving access to diagnostic solutions. Countries such as Brazil and Argentina are strengthening preventive healthcare initiatives and digital health adoption, supporting broader access to STI screening. Growing demand for discreet and convenient testing options is further supporting regional market growth.
Middle East And Africa At-home STI Testing Market Trends
The at-home STI testing market in the MEA is expanding due to rising healthcare investments and increasing awareness of preventive health screening. Improvements in healthcare infrastructure and digital health adoption are supporting access to diagnostic services. Public health initiatives aimed at improving HIV screening and reducing transmission are contributing to the adoption of self-testing solutions across several countries in the region.
Saudi Arabia at-home STI testing market is expanding due to increased healthcare investment and ongoing modernization initiatives under Vision 2030. The government’s focus on preventive healthcare and digital health transformation is improving access to diagnostic services. Expansion of telehealth platforms and growing awareness of preventive screening are expected to support increased adoption of discreet home-based STI testing solutions.
Key At-home STI Testing Company Insights
The competitive landscape of the market is highly competitive, with established diagnostics manufacturers and emerging direct-to-consumer (D2C) health platforms competing to expand market share. Key players such as OraSure Technologies, Abbott Laboratories, bioLytical Laboratories, Chembio Diagnostics, Becton Dickinson, Hologic, LetsGetChecked, Everlywell, and Nurx hold significant positions in the market. Leading companies are focusing on expanding their customer base through product innovation, telehealth integration, and strategic partnerships that enable end-to-end testing, consultation, and treatment services.
Key At-home STI Testing Companies:
The following key companies have been profiled for this study on the at-home sti testing market.
- OraSure Technologies Inc.
- Abbott
- bioLytical Laboratories Inc.
- Chembio Diagnostics, Inc.
- BD (Becton Dickinson)
- LetsGetChecked
- Everlywell
- NURX Inc.
- Hologic, Inc.
Recent Developments
-
In January 2026, OraSure Technologies announced the expansion of its OraQuick HIV Self-Test distribution network across select Asia-Pacific markets, strengthening access to consumer-operated HIV testing and supporting regional self-testing initiatives.
-
In February 2026, Hologic, Inc. received U.S. FDA approval for its Aptima HPV Assay as a primary screening option, expanding cervical cancer screening capabilities and strengthening its women’s health diagnostics portfolio. The mRNA-based assay detects high-risk HPV infections most likely to progress to cervical cancer and supports guideline-recommended screening approaches, including HPV primary testing, co-testing, and cytology. This approval enhances screening flexibility for providers and reinforces the shift toward HPV-based primary screening strategies.
-
In October 2025, Becton Dickinson introduced an HPV self-collection solution for use outside the U.S., designed to simplify at-home sample collection and expand access to cervical cancer screening. The IVDR-certified dry swab technology enables safe, non-invasive self-collection and convenient mailing to laboratories, while automated processing through the BD COR system enhances workflow efficiency and result reliability. This innovation aims to reduce logistical barriers and improve screening participation, particularly among underscreened populations.
At-home STI Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.24 billion
Revenue forecast in 2033
USD 5.86 billion
Growth rate
CAGR of 8.85% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Infection, test type, sample type, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
OraSure Technologies Inc.; Abbott; bioLytical Laboratories Inc.; Chembio Diagnostics, Inc.; BD (Becton Dickinson); LetsGetChecked; Everlywell; NURX Inc.; Hologic, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global At-home STI Testing Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global at-home STI testing market report based on infection, test type, sample type, distribution channel, and region:
-
Infection Outlook (Revenue, USD Million, 2021 - 2033)
-
HIV
-
Chlamydia (CT)
-
Gonorrhea (NG)
-
Syphilis
-
Trichomoniasis
-
HPV
-
Hepatitis B & C
-
Others
-
-
Test Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Rapid Lateral Flow Immunoassays
-
Molecular / NAAT-based Kits
-
Multiplex Panel Testing
-
Others
-
-
Sample Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood
-
Urine
-
Oral Fluid/Saliva
-
Vaginal Swab
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Online Pharmacies & D2C Platforms
-
Retail Pharmacies
-
Public Health Distribution Programs
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global at-home STI testing market size was estimated at USD 2.98 billion in 2025 and is expected to reach USD 3.24 billion in 2026.
b. The global at-home STI testing market is expected to grow at a compound annual growth rate of 8.85% from 2026 to 2033 to reach USD 5.86 billion by 2033.
b. North America dominated the at-home STI testing market with a share of 40.27% in 2025. This is attributable to rising STI incidence rates, strong public health screening initiatives, and high adoption of digital health solutions.
b. Some key players operating in the at-home STI testing market include OraSure Technologies Inc., Abbott, bioLytical Laboratories Inc., Chembio Diagnostics, Inc., BD (Becton Dickinson), LetsGetChecked, Everlywell, NURX Inc., Hologic, Inc.
b. Key factors that are driving the market growth include increasing prevalence of sexually transmitted infections (STIs) such as chlamydia, gonorrhea, syphilis, and HIV, along with increasing awareness and demand for private, convenient testing solutions, is significantly driving market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









