- Home
- »
- Medical Devices
- »
-
North America Bioregenerative Aesthetic Injectable Market Report, 2033GVR Report cover
![North America Bioregenerative Aesthetic Injectable Market Size, Share & Trends Report]()
North America Bioregenerative Aesthetic Injectable Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (PN, Peptide-based Injectables), By Application, By End Use (MedSpas, Aesthetic Surgery Centers), And Segment Forecasts
- Report ID: GVR-4-68040-850-1
- Number of Report Pages: 180
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2026
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
North America Bioregenerative Aesthetic Injectable Market Summary
The North America bioregenerative aesthetic injectable market size was estimated at USD 497.8 million in 2025 and is projected to reach USD 1,160.0 million by 2033, growing at a CAGR of 11.2% from 2026 to 2033. This growth is driven by rising consumer demand for natural-looking, minimally invasive treatments that promote long-term skin health.
Key Market Trends & Insights
- The U.S. bioregenerative aesthetic injectable market held the largest share of 90.9% in 2025.
- The bioregenerative aesthetic injectable industry in Canada is expected to grow significantly over the forecast period.
- By product, the biostimulatory injectables segment held the highest market share in 2025.
- By application, the wrinkle correction (for face) segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 497.8 Million
- 2033 Projected Market Size: USD 1,160.0 Million
- CAGR (2026-2033): 11.2%
Advances in regenerative technologies like platelet-rich plasma, polynucleotides, and exosomes are boosting product effectiveness. Increasing awareness, higher disposable incomes, and expanding medical aesthetics infrastructure worldwide are further driving adoption across key markets. In the U.S., consumer behavior toward bioregenerative aesthetic injectables is driven by a wellness focus and a strong preference for “natural” or autologous options that emphasize tissue repair rather than merely hiding imperfections. Adoption is highest among urban professionals in higher-income brackets who view aesthetic treatments as part of their self-care routine.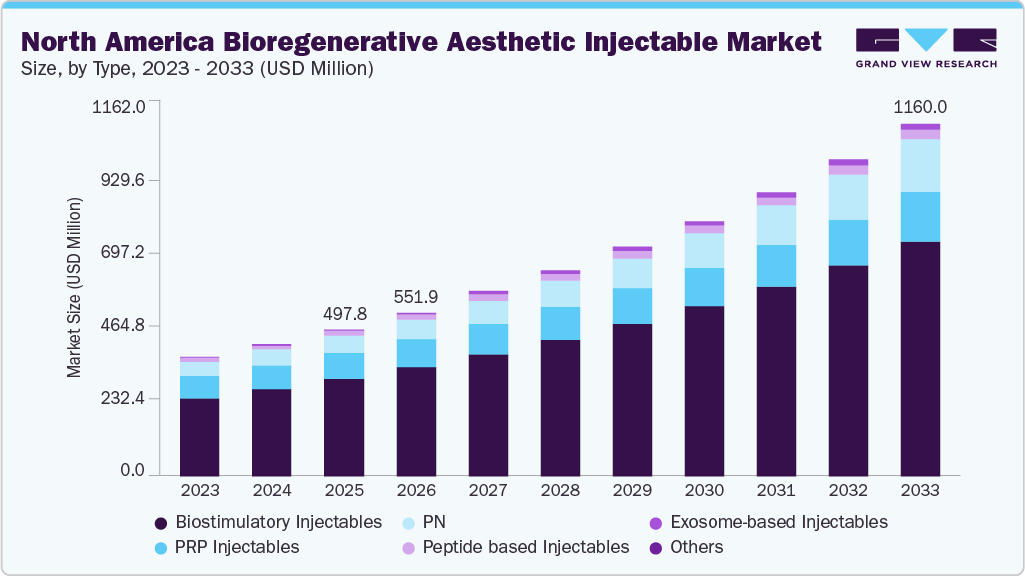
They prefer minimally invasive procedures that can be repeated, such as PRP, peptide-based boosters, and exosome boosters. Clinic results, patient reviews, and social media influencers shape their choices. Messaging highlights regeneration, healing, and biologics, resonates with those cautious about synthetic implants yet willing to spend on long-term skin health. The U.S. continues to see growth in minimally invasive injectables. Professional data show a slight increase in these treatments, and global surveys rank the U.S. as the leader in total aesthetic procedures, indicating a strong supply and patient familiarity.
Notable & Recent Aesthetic Platform Transactions: U.S.
Target/Platform
Acquirer/Investor
Investment Date
Clinic Locations
Key States/Coverage
Formula Wellness
Trive Capital
March 2024
5
Texas
Aesthetic Partners
Norwest Venture Partners
November 2023
20
CA, FL, TX, VA
United Aesthetic Alliance
Varsity Healthcare Partners
September 2023
3+
Minnesota
Sona Dermatology & MedSpa
Levine Leichtman Capital Partners
June 2023
30+
Nationwide
Cosmetic Skin & Laser Center
Incline Equity Partners
May 2023
9
FL, MI
Alpha Aesthetics
The Jackson Group
April 2023
15
CA, NY, TX, VA
Advanced Aesthetic Partners
Raven Capital
October 202
N/A
U.S., UK
Elase Medical Spas
Imperial Capital
October 2022
13
FL, ID, UT
SkinSpirit
KKR
September 2022
41
Nationwide
Empower Aesthetics
Shore Capital
September 2022
12
NY, TN, TX
The prevalent working-age population significantly fuels the demand for PLLA fillers. According to Statistics Canada (September 2024), the median age in Canada is 40.3 years, with a substantial portion aged 20-65 actively seeking solutions to address wrinkles, sagging, and volume loss. PLLA fillers provide gradual, natural-looking improvements by stimulating endogenous collagen synthesis, making them a favorable option for this demographic. In parallel, the U.S. geriatric population, approximately 1 in 6 individuals above 65, as per the U.S. Census Bureau (May 2023), increasingly seeks regenerative aesthetic solutions to counteract age-related skin changes, further propelling market growth. The U.S. also has the highest concentration of plastic surgeons globally, creating widespread access to advanced injectables and reinforcing the market’s expansion potential.
Canadian patients tend to use bioregenerative injectables more cautiously than their U.S. counterparts. Their decisions are mainly guided by clinicians, such as dermatologists and plastic surgeons, recommended through referrals and influenced by stricter public perceptions of medical legitimacy. Since many Canadians rely on a universal health system for primary care, elective aesthetic procedures are largely discretionary, paid out-of-pocket, and mainly performed in urban areas like Toronto, Vancouver, and Montreal, where specialist clinics and dermatologists are concentrated.
Popular regenerative treatments like PRP are well-established, but they are regulated locally (Health Canada oversees autologous blood products), which increases the demand for clear safety and effectiveness evidence. This regulatory environment encourages patients to prefer clinic-led, evidence-supported treatments over social-media trends. As a result, the adoption of advanced regenerative injectables occurs more slowly but consistently, with patients emphasizing physician reputation, published evidence, and transparent safety measures when selecting exosome or peptide therapies.
Market Concentration & Characteristics
The North America bioregenerative aesthetic injectable industry exhibits moderate to high industry concentration, led by major players such as Galderma, AbbVie/Allergan, and Merz, supported by strong distribution networks and regulatory compliance capabilities. Market characteristics include high adoption of minimally invasive regenerative treatments, rapid innovation in collagen-stimulating injectables, and strong physician-led service delivery. Growth is supported by expanding med spa networks, rising consumer awareness of natural-looking aesthetic outcomes, and continuous professional training initiatives. Competition focuses on product efficacy, safety profiles, practitioner training, and brand positioning, with companies investing in clinical evidence and marketing to strengthen market share.
The North America bioregenerative aesthetic injectables industry demonstrates a high degree of innovation, driven by advances in collagen-stimulating formulations, improved injection techniques, and combination treatment protocols. Companies are developing next-generation injectables that promote tissue regeneration, longer-lasting results, and enhanced safety profiles. Innovation also extends to delivery methods, integrating treatments with energy-based devices, microneedling, and personalized aesthetic plans. Digital imaging and AI-based skin assessment tools further support customized treatment approaches. Continuous clinical research and practitioner training programs accelerate product adoption, enabling providers to offer regenerative, minimally invasive solutions that deliver natural, progressive, and long-term aesthetic outcomes.

The North America bioregenerative aesthetic injectables industry shows a moderate to high level of M&A activity as established aesthetic and pharmaceutical companies seek to expand regenerative portfolios and strengthen distribution networks. Large players frequently acquire innovative biotech firms and aesthetic brands to access new collagen-stimulating technologies and accelerate product commercialization. Strategic partnerships and investments also support expansion into fast-growing med spa and dermatology clinic networks. Consolidation trends aim to enhance market presence, broaden product offerings, and secure competitive advantages in a rapidly evolving minimally invasive aesthetics segment driven by increasing consumer demand for long-lasting regenerative treatments.
Regulation significantly influences the North America bioregenerative aesthetic injectables industry by ensuring product safety, quality, and clinical effectiveness. In the U.S., the FDA regulates injectable products through stringent approval pathways, manufacturing standards, and post-market surveillance, shaping product development timelines and market entry strategies. Compliance requirements increase development costs but strengthen physician and patient confidence in approved treatments. Regulations also govern practitioner qualifications and facility standards, affecting service delivery across clinics and med spas. While strict oversight can slow product launches, it ultimately supports sustainable market growth by promoting safe adoption of regenerative aesthetic treatments and maintaining high industry standards.
Product substitutes in the North America bioregenerative aesthetic injectables industry include traditional hyaluronic acid dermal fillers, botulinum toxin injections, energy-based skin tightening devices, and non-invasive cosmetic procedures such as laser resurfacing, chemical peels, and radiofrequency treatments. Surgical options like facelifts and fat grafting also serve as alternatives for patients seeking longer-lasting results. In addition, topical regenerative skincare products and collagen-stimulating therapies provide non-injectable options. The availability of multiple aesthetic solutions allows patients to choose treatments based on cost, invasiveness, recovery time, and desired outcomes, creating competitive pressure on bioregenerative injectable products in clinical and med spa settings.
Regional expansion in the North America bioregenerative aesthetic injectables industry is driven by growing adoption of minimally invasive regenerative treatments beyond major metropolitan areas into suburban and secondary cities. Increasing numbers of dermatology clinics, plastic surgery centers, and medical spas are expanding service availability across the U.S. and Canada. Cross-border aesthetic medical tourism between the two countries further supports market reach.
Product Insights
The biostimulatory injectables segment accounted for the largest revenue share of 78.6% in 2025, primarily due to its ability to deliver long-lasting, natural-looking aesthetic results by stimulating the body’s own collagen production. Unlike traditional fillers that provide immediate volume, biostimulatory injectables such as poly-L-lactic acid and calcium hydroxylapatite promote gradual tissue regeneration and structural improvement. Growing patient preference for minimally invasive treatments with durable outcomes, reduced maintenance frequency, and improved skin quality has driven adoption. Strong physician confidence, expanding clinical indications, and increasing use in combination therapies further support the segment’s dominant market position.
The exosome-based injectables segment is estimated to register the fastest growth rate from 2026 to 2033 due to increasing interest in advanced regenerative therapies that support cellular communication, tissue repair, and skin rejuvenation. Exosomes contain bioactive molecules such as growth factors, proteins, and RNA that enhance collagen synthesis, reduce inflammation, and accelerate healing. Rising demand for next-generation, minimally invasive aesthetic solutions, along with ongoing clinical research and product innovation, is driving adoption. In addition, expanding use in combination with microneedling and energy-based treatments, coupled with improving regulatory clarity and practitioner training, is expected to accelerate market growth.
Application Insights
The wrinkle correction (for face) segment accounted for the largest share of 20.6% in 2025. An aging population increasingly seeks non-surgical aesthetic solutions that can delay or diminish visible signs of aging without the recovery time or risk associated with surgical procedures. With populations over age 60 expanding worldwide, the prevalence of wrinkles and sagging skin has become a central driver of consumer demand for minimally invasive interventions.
The under eye-areas (for face) segment in the market is expected to witness the fastest CAGR during the forecast period. The rising demand is also influenced by lifestyle factors, including increased screen time and digital fatigue, which accentuate under-eye shadows and hollowness, especially among younger demographics who now view early preventative aesthetics as part of self-care.
End Use Insights
The medSpas segment held the largest share of revenue, more than 42.9%, in 2025. As more people seek holistic beauty and wellness solutions, medspas have expanded their offerings to include regenerative treatments such as PRP, exosomes, and collagen-stimulating injectables alongside traditional injectables like neurotoxins and dermal fillers. This shift is supported by growing interest in treatments that enhance the body’s own healing and rejuvenation processes, making bioregenerative options particularly appealing. In addition, the number of U.S. med spas rising from 1,600 in 2010 to nearly 10,500 in 2023 and projected to reach 13,000 by 2026, the sector represents a massive commercial channel for bioregenerative aesthetic injectables.
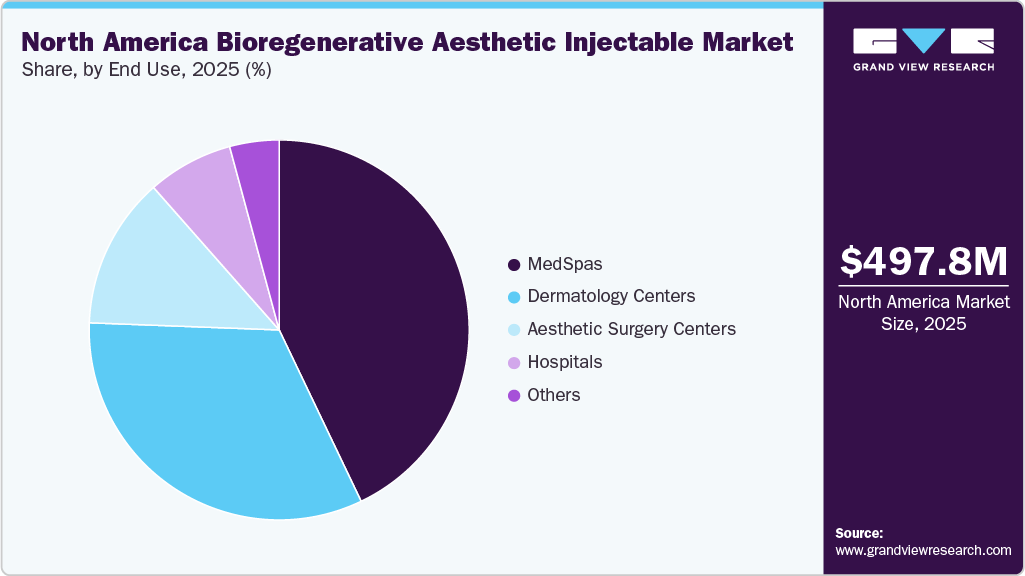
The dermatology centers in the market is driven by increasing patient demand for medically supervised, minimally invasive regenerative treatments. Patients increasingly prefer dermatologist-led procedures due to higher safety standards, clinical expertise, and access to FDA-compliant biostimulatory products. Rising awareness of collagen-stimulating injectables for skin aging, texture improvement, and scar management is further supporting demand. Dermatology centers are also expanding service offerings by integrating bioregenerative injectables with lasers, microneedling, and energy-based therapies. Ongoing physician training, advanced diagnostic tools, and strong referral networks continue to reinforce dermatology centers as key treatment hubs across North America.
Regional Insights
North America held the largest market share, exceeding 33.15% in 2025, and is projected to experience strong growth in the coming years. Market expansion is fueled by rising demand for minimally invasive, durable, and biologically driven aesthetic treatments. Poly-L-lactic acid (PLLA) fillers are gaining popularity due to their ability to stimulate collagen formation, support skin regeneration, and enhance tissue repair rather than only restoring volume. Clinical adoption of regenerative aesthetic techniques is also expanding, with PLLA fillers increasingly used to address skin thinning, structural aging, and natural contour enhancement. Combination treatments involving microneedling, radiofrequency, and ultrasound-based collagen stimulation further demonstrate a growing preference for multimodal, regeneration-centered aesthetic solutions across North America.
Professional training trends reflect this shift, as major U.S. aesthetic education forums now emphasize regenerative injectables, focusing on tissue biology, collagen restoration, and healing mechanisms. In addition, cross-border medical tourism between the U.S. and Canada influences clinic marketing strategies, with providers increasingly promoting collagen regeneration and tissue-support benefits to attract international aesthetic patients.
U.S. Bioregenerative Aesthetic Injectable Market Trends
The bioregenerative aesthetic injectable market in the U.S. continues to play a central role in the North American market, supported by a growing shift toward regenerative therapies, particularly poly-L-lactic acid (PLLA). Patients increasingly prefer treatments that promote gradual, biologically driven skin rejuvenation rather than instant cosmetic correction. PLLA products, produced under FDA standards and distributed through certified dermatologists and plastic surgeons, stimulate natural collagen production, helping restore facial volume and skin elasticity, especially appealing to the aging population seeking subtle, long-term improvements. Medical spas have become an important channel for delivering bioregenerative injectables. Data from industry associations show rapid expansion in med spa facilities over the past decade, with many offerings bundled regenerative treatments such as skin boosters and scalp therapies delivered through scheduled treatment programs. Professional training programs for nurses and physician assistants are also expanding to ensure safe and standardized administration of PLLA procedures.
Meanwhile, clinics and distributors continue to broaden patient access by incorporating PLLA into comprehensive aesthetic treatment plans. Supported by regulatory oversight, technological advancements, and rising consumer interest in minimally invasive regenerative solutions, the U.S. market is positioned for continued expansion, with bioregenerative injectables increasingly shaping the future of aesthetic medicine through natural and long-lasting skin rejuvenation approaches.
Canada bioregenerative aesthetic injectable market is witnessing steady growth, driven by gaining traction among clinics and patients looking for subtle, long-lasting rejuvenation focused on collagen stimulation and tissue health rather than quick volumization. A key recent development in Canada’s regenerative injectable landscape is Health Canada’s expanded approval of regenerative biostimulators such as Radiesse, which now includes the décolleté area, signaling growing regulatory support for collagen-stimulating treatments beyond facial zones and reflecting strong demand for regenerative aesthetic options.
A major industry development in January 2026 underscores the expansion of advanced injectable-focused platforms across Canada. Victoria Park Medispa announced its acquisition of PHI Medical Aesthetics, marking its first entry into Alberta and Western Canada (PR Newswire, January 2026). Founded in Calgary, PHI is recognized for expert, injectables-focused clinical care and natural results, with established clinics in Marda Loop and Mahogany. This acquisition follows Victoria Park’s flagship Toronto clinic opening in October 2025 and brings its total footprint to 27 clinics nationwide, signaling accelerating consolidation and professionalization within Canada’s regenerative aesthetics ecosystem.
Key North America Bioregenerative Aesthetic Injectable Company Insights
Some of the key companies in the market include MD Skin Solutions, Mastelli s.r.l., Merz Aesthetics and among others. Most companies focus on R&D activities to develop technologically advanced products to gain a competitive edge. Companies are also adopting various strategies, such as mergers and acquisitions, joint ventures, and developing low-cost equipment, especially for developing economies.
Key North America Bioregenerative Aesthetic Injectable Companies:
- PharmaResearch Products Co., Ltd.
- MD Skin Solutions
- Mastelli s.r.l.
- Merz Aesthetics
- Caregen Co. Ltd
- Croma Pharma
- PROMOITALIA
- Galderma
- Fillmed
- IBSA
- Bio-Formula
- BR Pharma
- Allergan (AbbVie)
- Merz
- Fidia
- VAIM
Recent Developments
-
In October 2025, Croma-Pharma GmbH announced a strategic distribution partnership with Renellence to strengthen its footprint in Canada. Renellence will distribute Croma’s MDR-approved Saypha hyaluronic acid fillers, while both companies collaborate on medical education, training, and long-term growth in the Canadian aesthetics market.
-
In June 2025, Galderma announced a new U.S. headquarters in Miami, aiming to accelerate innovation and growth in its largest market. Located in Brickell, the site will host about 150 employees by 2028, strengthening leadership in skincare and injectable aesthetics while maintaining key U.S. hubs nationwide.
North America Bioregenerative Aesthetic Injectable Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 551.9 million
Revenue forecast in 2033
USD 1,160.0 million
Growth rate
CAGR of 11.2% from 2026 to 2033
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, region
Regional scope
North America
Country scope
U.S.; Canada
Key companies profiled
PharmaResearch Products Co., Ltd.; MD Skin Solutions, Mastelli s.r.l., Merz Aesthetics, Caregen Co., Ltd, Croma Pharma, PROMOITALIA, Galderma, Fillmed, IBSA, Bio-Formula, BR Pharma, Allergan (AbbVie), Merz, Fidia, VAIM
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America Bioregenerative Aesthetic Injectable Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the North America bioregenerative aesthetic injectable market report based on product, application, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
PN
-
PDRN
-
PN Injectables
-
-
Peptide-based Injectables
-
Exosome-based Injectables
-
Biostimulatory Injectables
-
PLLA
-
CaHA
-
-
PRP Injectables
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Skin Hydration & Moisturization (For Face)
-
Elasticity & Firmness (For Face)
-
Under eye-areas (For Face)
-
Fine Lines (For Face)
-
Acne Scar (For Face)
-
Wrinkle Correction (For Face)
-
Body Application
-
Arms & Thighs
-
Décolletage
-
Intimate Areas
-
Knees
-
Scalp & Hair Rejuvenation
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
MedSpas
-
Aesthetic Surgery Centers
-
Hospitals
-
Dermatology Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
-
Frequently Asked Questions About This Report
b. The North America bioregenerative aesthetic injectable market size was estimated at 497.8 million in 2025 and is projected to reach 551.9 million by 2026.
b. The North America bioregenerative aesthetic injectable market size was estimated at 497.8 million in 2025 and is projected to reach 1,160.0 million by 2033, growing at a CAGR of 11.2% from 2026 to 2033.
b. The biostimulatory injectables segment accounted for the largest revenue share of 78.6% in 2025, primarily due to its ability to deliver long-lasting, natural-looking aesthetic results by stimulating the body’s own collagen production.
b. MD Skin Solutions, Mastelli s.r.l., Merz Aesthetics, Caregen Co. Ltd, Croma Pharma, PROMOITALIA, Galderma, Fillmed, IBSA, Bio-Formula, BR Pharma, Allergan (AbbVie), Merz, Fidia, VAIM are the key players in the North America Bioregenerative Aesthetic Injectable market.
b. This growth is driven by rising consumer demand for natural-looking, minimally invasive treatments that promote long-term skin health. Advances in regenerative technologies like platelet-rich plasma, polynucleotides, and exosomes are boosting product effectiveness. Increasing awareness, higher disposable incomes, and expanding medical aesthetics infrastructure worldwide are further driving adoption across key markets.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










