- Home
- »
- Medical Devices
- »
-
Exoskeleton Market Size And Share, Industry Report, 2033GVR Report cover
![Exoskeleton Market Size, Share, And Trend Report]()
Exoskeleton Market (2026 - 2033) Size, Share, And Trend Analysis Report, By Mobility (Mobile, Fixed/Stationary), By Technology (Powered, Non-powered), By Extremity (Upper Body, Lower Body), By End Use (Healthcare, Military), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-071-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Exoskeleton Market Summary
The global exoskeleton market size was estimated at USD 590.02 million in 2025 and is projected to reach USD 1.79 billion by 2033, growing at a CAGR of 14.48% from 2026 to 2033. Rapidly growing global geriatric population, rising adoption rates of medical devices in different industries such as automobile, military, defense, and construction, and rising incidence of stroke are some of the key driving forces responsible for the exoskeleton market growth.
Key Market Trends & Insights
- North America exoskeleton market dominated global market in 2025 with a revenue share of 44.10%.
- The U.S. exoskeleton market accounted for the largest share in North America in 2025.
- Based on mobility, the mobile segment held the largest share of 62.64% in 2025.
- Based on technology, the powered segment held the largest share in 2025.
- Based on extremity, the lower body segment held the largest share in 2025.
- Based on end use, the healthcare segment held the largest share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 590.02 Million
- 2033 Projected Market Size: USD 1.79 Billion
- CAGR (2026-2033): 14.48%
- North America: Largest market in 2025
- Asia Pacific: Fastest market in 2025
Furthermore, the growing incidence rate of spinal cord injuries (SCI) is expected to drive the demand across global markets. For instance, as per the National Spinal Cord Injury Statistical Center (NSCISC), in the U.S., 17,730 new SCIs are diagnosed annually, and approximately 291,000 live with SCIs.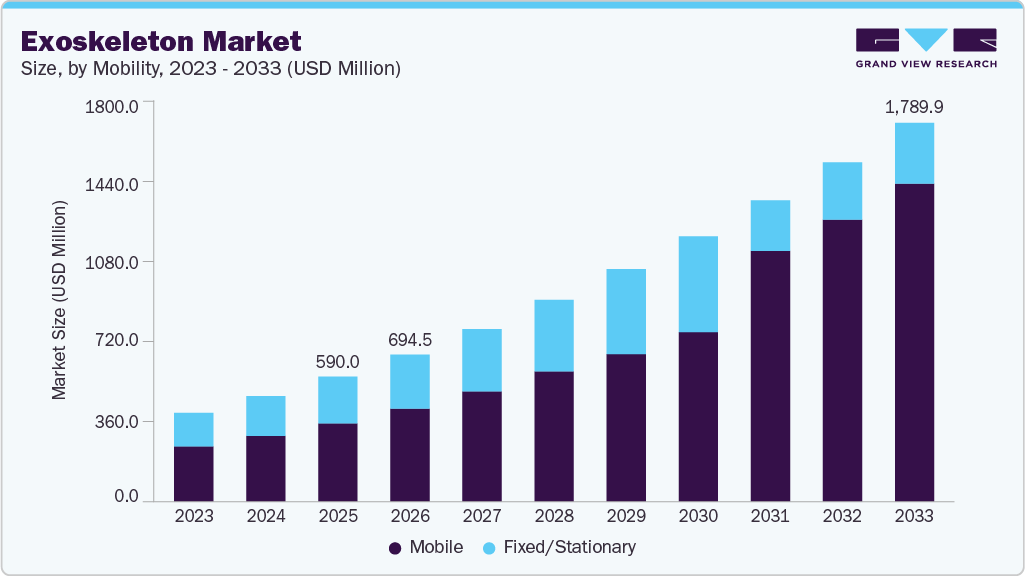
Exoskeleton solutions are widely adopted across industries to support employee health and boost productivity, positioning them as a critical segment within the broader wearable robot market. Growing demand has led to emergence of several startups expertizing in exoskeleton technology and providing solutions for rehabilitation. Furthermore, constantly evolving exoskeleton industry is driving market players to devise innovative product development strategies to boost adoption over the forecast period. In January 2026, RoboCT introduced its lightweight GoGo Exoskeleton series at CES 2026, marking the company’s expansion from clinical rehabilitation into consumer-focused daily mobility solutions. The 2.3 kg AI-powered wearable offers multi-mode, real-time adaptive support for activities such as walking, sitting, and balance assistance, targeting post-stroke recovery, neurodegenerative conditions, and everyday mobility needs.
Increasing prevalence of spinal cord injury is boosting the growth of the exoskeleton segment within the wearable robot market, as patients are increasingly recommended to use robotic wearable assistive devices to improve mobility and functional outcomes. RaThis has become major driver for adoption of exoskeleton by patients as well as healthcare providers. In September 2025, United Spinal Association reported that 250,000 to 390,000 people in the U.S. are living with spinal cord injury, with about 18,000 new cases each year, most often due to vehicle accidents and falls, and highlighted common secondary complications including depression, chronic pain, spasticity, pressure injuries, and bowel, bladder, and respiratory issues.
Some of the most common musculoskeletal injuries and disorders affecting the construction industry are occupational overuse syndrome (OOS), cumulative trauma disorders (CTD), and repetitive strain injury (RSI). For instance, as per WHO data estimates published in July 2022, approximately 1.71 billion individuals across the globe have musculoskeletal disorders that include neck pain, lower back pain, fractures, amputation, rheumatoid arthritis, and osteoarthritis. Similarly, in 2022, according to the U.S. Bureau of Labor Statistics, approximately 502,380 workers have occupation-related musculoskeletal disorders across several industries. These are some of the driving factors boosting adoption of exoskeleton solutions owing to benefits associated with exoskeleton technology, such as augmenting or assisting users’ physical activity or capability, improving users’ health & productivity, and reducing fatigue experienced by industrial workers.
Rapid technological advancements in the exoskeleton segment of the wearable robot market are expected to boost demand for robotic assistive devices across healthcare and industrial applications. Increasing popularity and growing adoption of exoskeletons by companies are expected to contribute to market growth. In addition, increasing adoption of exoskeletons in manufacturing sector, logistics, automotive, and construction is leading to a rise in penetration of exoskeletons, thereby fueling the market growth. In May 2025, Wandercraft initiated clinical trials of its AI powered Personal Exoskeleton, using NVIDIA AI and simulation tools to enable individuals with spinal cord injuries, stroke, and severe mobility impairments to stand and walk with real time adaptive support.
The trend of including exoskeletons in health insurance coverage is rising, making them more accessible to patients. This is particularly evident in countries such as Germany, where specific exoskeleton systems are recognized for insurance procurement. For instance, in November 2023, ReWalk Robotics Ltd. appreciated the Centers for Medicare & Medicaid Services (CMS) for officially publishing the Calendar Year 2024 Home Health Prospective Payment System Rule. This regulation, which was issued on November 1, 2023, and is set to be implemented starting from January 1, 2024, confirms the classification of exoskeletons under the Medicare brace benefit category.
Case Studies & Insights
Case Study Title: “Exoskeletons for Medical Rehabilitation - A Review of Recent Advances”
Source: Journal of Assistive Technology, Tandfonline, 2023
Overview
This case study examines the advancements in medical exoskeletons, specifically for patients with spinal cord injuries, stroke recovery, and other neurological conditions. It highlights the key challenges and recent technological improvements in the development of exoskeletons that aim to restore mobility and improve rehabilitation outcomes.
Key Findings
-
Technological Integration: The study emphasizes the integration of AI, robotics, and biomechanics to enhance the adaptive capabilities of exoskeletons. These technologies help to personalize therapy, allowing for a more tailored and effective rehabilitation process.
-
Patient Mobility: Significant progress has been made in creating lightweight exoskeletons that are easier to wear and provide natural, intuitive movement. This improves patient comfort and encourages longer usage, which is vital for rehabilitation.
-
Market Adoption: Despite technological advances, the study highlights that cost, regulatory hurdles, and the need for medical supervision limit wider adoption in rehabilitation centers.
-
Example: Ekso Bionics' EksoGT is a key example of an exoskeleton used for walking assistance in stroke and spinal injury patients. The device has shown positive results in improving patient mobility and rehabilitation.
Conclusion
- Exoskeletons are promising tools for rehabilitation, but cost-efficiency, user comfort, and long-term effectiveness need continued refinement for broader market integration
Market Concentration & Characteristics
The wearable robots/exoskeleton market is characterized by a high degree of innovation due to the rapid technological advancements driven by factors such as advancements in robotics and artificial intelligence technology. Novel and innovative applications of exoskeletons are constantly emerging, leading to the creation of new opportunities for market players. In May 2025, German Bionic unveiled Exia, an AI powered exoskeleton trained on real world motion data that delivers adaptive lift assistance of up to 38 kg and adjusts support in real time, targeting reduced musculoskeletal injury risk and improved productivity across industries such as manufacturing, logistics, retail, and healthcare.
The exoskeleton market is characterized by a high level of merger and acquisition (M&A) activity by the leading players. This is due to several factors, including the desire to access new robotic technologies needed to consolidate in a rapidly growing market and the increasing adoption of these newly developed exoskeletons. In January 2026, EKSO Bionics announced a proposed merger with Applied Digital that could result in the separation or sale of its exoskeleton business, including the Indego Personal system, amid continued financial pressure despite Medicare coverage and ongoing sales in the complex rehab technology market.
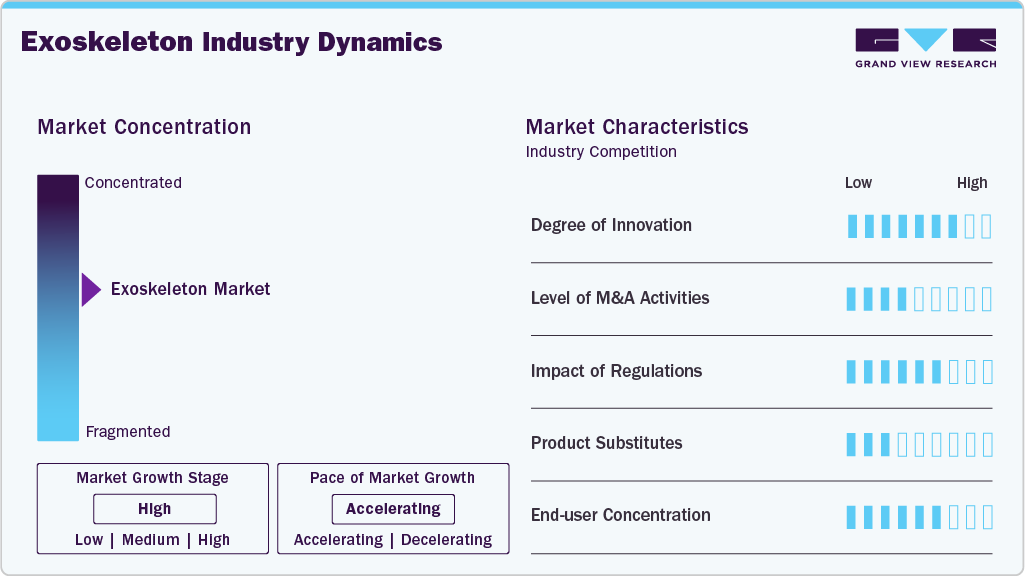
Well-developed regulatory framework and rising approval for the exoskeleton products from these regulatory bodies positively impact market growth. The certifications and standards provided by government authorities for exoskeletons in their manufacture, deployment, and use would enhance their adoption at workplace. In January 2026, Baltimore-based NextStep Robotics said it is seeking additional FDA authorization via a De Novo submission to expand use of its AMBLE lower-body exoskeleton beyond exercise therapy for stroke rehabilitation. The company cited NIH-funded clinical trial results showing sustained improvements in walking months after therapy, while advancing a subscription-based model to improve affordability for community clinics.
Traditional rehabilitation methods remain prevalent, and exoskeletons are emerging as valuable alternatives. They offer advantages such as enhanced mobility and independence for patients with spinal cord injuries or neurological disorders. The high cost and need for specialized training may limit their widespread adoption, positioning them as complementary rather than direct substitutes to conventional therapies.
Several market players are expanding their business by entering new geographical regions to strengthen their market position and expand their product portfolio. Rising research and development activities create more opportunities for market players to enter new regions. In January 2026, NAVEE announced its geographical expansion from Europe to the U.S., unveiling its global intelligent mobility strategy at a Silicon Valley event. The company introduced five AI-enabled products, including Exo-Fit, a full-terrain wearable exoskeleton for human mobility, alongside solutions for land, air, and water travel.
Mobility Insights
The mobile segment led the market in 2025 with the largest revenue share of 62.64% and is anticipated to witness fastest CAGR over the forecast period. Factors such as a rise in research and development activities and a surge in demand for motor-equipped robots aiding human body mechanics are attributed to market growth. In January 2026, Sumbu unveiled its Exo-S3 consumer exoskeleton lineup at CES 2026, introducing AI powered, lightweight leg assistance designed for everyday mobility, injury prevention, and outdoor use. The Exo-S3 series uses dual-vector drive technology and human-intent recognition AI to adapt support across varied terrain, targeting reduced fatigue, improved movement efficiency, and broader consumer adoption of wearable mobility devices.
The fixed/stationary segment is expected to grow significantly, driven by increasing demand for rehabilitation and assisted living solutions. These devices, designed to aid patients with limited mobility, provide vital support for standing, posture correction, and walking stationary. Growth is further supported by rising adoption in hospitals and rehabilitation centers, where controlled environments favor tethered systems for repetitive therapy, clinical monitoring, and long-duration use. For instance, the NX A3-M is a fixed-frame exoskeleton designed for gait rehabilitation, offering improvements over the earlier NX-A3 model.
Technology Insights
The powered segment held the largest revenue share with 71.40% in 2025 owing to rising implementation of powered exoskeleton solutions in several industries to enhance personal safety and boost productivity. In addition, the rise in innovative product launches and benefits associated with powered technology products are smoother lifting motion, reduced strain on worker’s body, increased strength, force multiplier, and enhanced productivity. For instance, in August 2024, Arc’teryx and Skip teamed up to introduce the world’s first powered pants, a wearable exoskeleton designed to improve human mobility by providing powered support.
Non-powered technology segment is anticipated to register the fastest growth rate over the forecast period, driven by demand for cost-effective, lightweight solutions that assist with lifting and mobility without relying on external power sources. These devices use mechanical systems, such as elastics or springs, to enhance movement and reduce fatigue, making them ideal for industrial and rehabilitation applications. Key drivers include the rising need for manual labor assistance in logistics, construction, and healthcare sectors, where workers handle heavy loads. For instance, the UPLIFT exoskeleton utilizes elastic mechanisms to support lifting tasks, highlighting the growing adoption of affordable, non-powered solutions in the workforce.
Extremity Insights
By extremity, market is segmented into lower body,upper body and full body. Lower body segment dominated the market in terms of revenue share with 40.72% in 2025. owing to rising investments, growing incidence of lower body disabilities, and increasing adoption rates of exoskeleton products by geriatric populations and paralyzed patients for weight-bearing capabilities and mobility. Initiatives such as, in April 2024, the ABLE Exoskeleton, a lightweight and affordable hip-knee-powered device by ABLE Human Motion, has received CE Mark approval under the new Medical Device Regulation, enabling its commercial sale for spinal cord injury rehabilitation in the EU.
The upper body segment is anticipated to register the fastest growth rate over the forecast period due to benefits provided by exoskeleton products in the upper extremities, such as supporting disabled communities and rehabilitation aid in post-stroke, neurological, or musculoskeletal impairments. Furthermore, growing prevalence of neurological disorders led to a rise in market growth. For instance, in May 2022, according to data published by the European Academy of Neurology, globally, a neurological disease affects 1 in 3 people at some point in their life.
End Use Insights
Healthcare segment dominated the market in terms of revenue share with 48.76% in 2025, owing to the rising incidence rate of spinal cord injuries (SCIs), widespread adoption of these products & solutions in rehabilitation centers, and significant surge in treatment numbers are some of the key driving factors. Moreover, a growing number of regulatory approvals and rising awareness of technologically advanced systems are driving the demand & adoption rate of exoskeleton products in the healthcare industry. For instance, in October 2023, Wandercraft SAS launched in the U.S. commercial operations of its exoskeleton, Atalante X. The solution is used for individuals with mobility impairments.
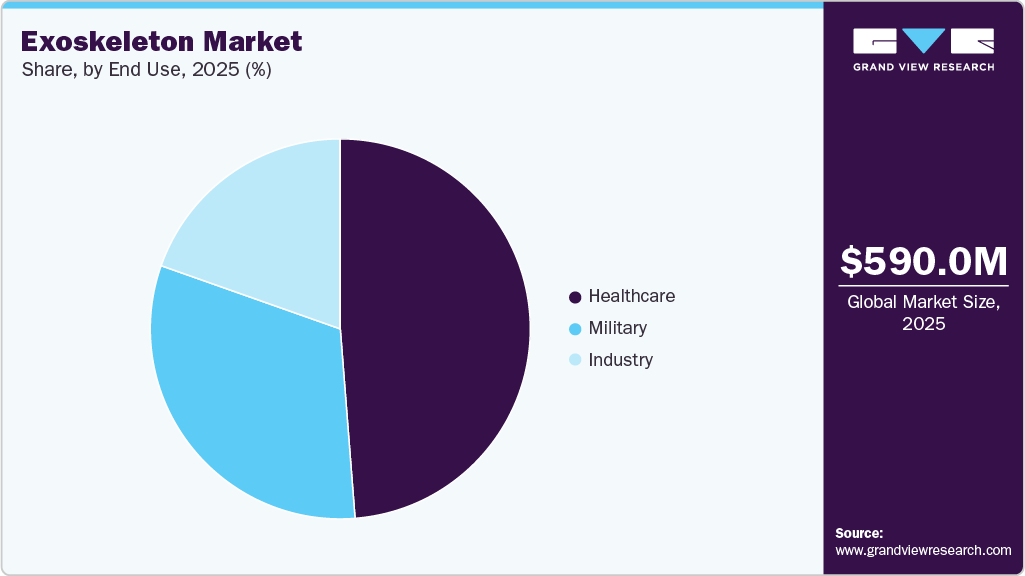
Industry segment is anticipated to register the fastest growth over the forecast period owing to rising awareness levels towards the benefits of exoskeleton technologies in several industry applications and the rapidly growing incidence rate of occupation-related injuries. Furthermore, the surge in demand for novel technologies and growing adoption of the exoskeleton in multiple sectors such as construction, manufacturing, and distribution is expected to accelerate growth of the industry segment. In August 2024, Festool announced the U.S. launch of its ExoActive powered exoskeleton for construction tradespeople, an industrial wearable designed to reduce shoulder and arm strain during overhead and front-facing tasks by up to 30%, providing up to 11 pounds of arm support to improve productivity and reduce work-related injuries in construction and trade environments.
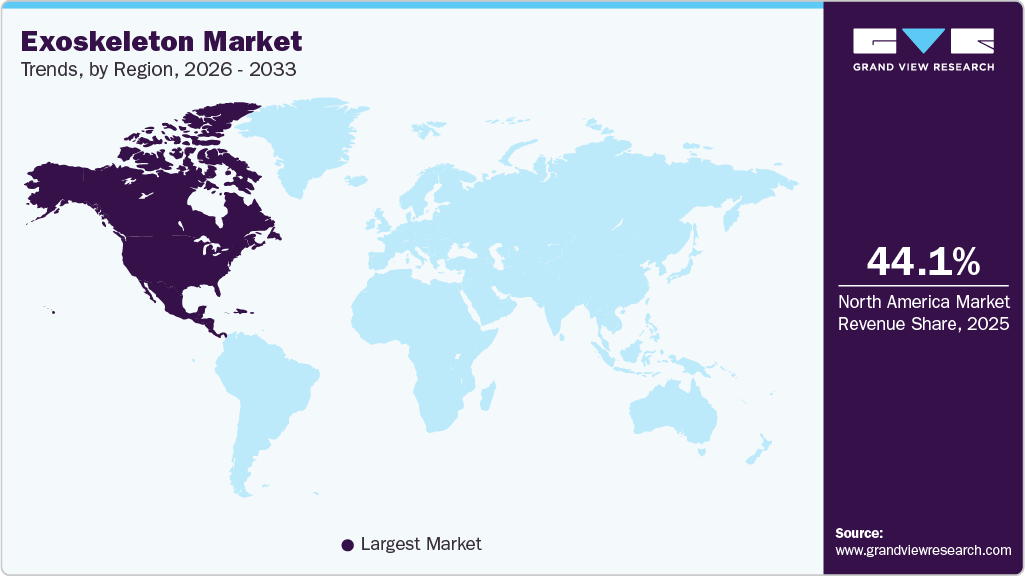
Regional Insights
North America Exoskeleton Market Trends
North America led the market in 2025, capturing 44.10% of the revenue share, driven by increasing investment in research, a growing disabled population, and expanding partnerships among key players, availability of advanced technologies, and strong public and private sector support. In June 2025, Wandercraft raised USD 75 million in Series D funding to launch its self-balancing personal exoskeleton, Eve, by 2026, supporting users with disabilities in daily mobility; the investment also expanded the Atalante X rehabilitation system, already FDA-cleared and deployed in over 100 U.S. hospitals and clinics, while enabling development of the Calvin‑40 humanoid robot.
U.S. Exoskeleton Market Trends
The U.S. dominated the North America exoskeleton market due to its strong focus on technological innovation, high investment in healthcare and military sectors, and the widespread presence of leading exoskeleton manufacturers. For instance, in April 2025, Lifeward Ltd. announced the nationwide U.S. launch of the ReWalk 7 Personal Exoskeleton, representing the latest advancement in its technology.
“The ReWalk 7 was developed over several years, integrating advanced technological innovations with feedback from clinicians and patients to build upon the ReWalk’s world-class reputation for industry leadership. The result is a device that is optimized for real-world use, with an unmatched user experience and freedom of movement. We are thrilled to be able to provide paralyzed individuals across the country with a new option for integrating walking to everyday life.”
- Larry Jasinski, CEO of Lifeward
Asia Pacific Exoskeleton Market Trends
Asia Pacific is anticipated to register the fastest growth rate during the forecast period owing to a growing patient pool requiring rehabilitation support. Availability of government support & funding and developing & advancing healthcare infrastructure are driving the development & growth of the exoskeleton market in Asia Pacific. In February 2025, Kia and Hyundai unveiled the X-ble Shoulder, a lightweight carbon exoskeleton designed to reduce shoulder strain and improve industrial productivity, initially deployed across their South Korean facilities.
The exoskeleton market in China is rapidly advancing in technologies, focusing on enhancing mobility and strength. Chinese engineers are actively working on creating exoskeleton suits to assist astronauts, demonstrating the country's commitment to innovation in this field. In addition, in January 2025, the Chinese military showcased exoskeletons for logistics operations. The systems are intended to enhance soldier endurance and load-carrying capacity, highlighting growing defense-sector adoption of wearable robotic assistance.
Europe Exoskeleton Market Trends
Strong regulatory frameworks and a focus on medical and industrial applications drive the exoskeleton market in Europe. The European Medicines Agency (EMA) ensures that medical exoskeletons meet stringent safety and efficacy standards, facilitating their integration into healthcare systems. In June 2023, Comau, in collaboration with IUVO and Italy’s Esselunga, introduced the MATE-XB wearable lumbar exoskeleton designed to reduce lower back strain during lifting and repetitive tasks; the passive, battery-free device supports up to 25 kg, adapts to user movements, and was tested in Italy to ensure ergonomic benefits for logistics, fulfillment, and manual handling workers.
The exoskeleton market in Germany is growing in terms of technologies, and the country benefits from a well-established regulatory framework that supports the development and deployment of exoskeletons. The European Medicines Agency (EMA) is vital in ensuring the safety and effectiveness of medical exoskeletons, helping them integrate into healthcare systems. In December 2025, German Bionic expanded its Exia exoskeleton with new vest designs specifically for women, improving ergonomics, comfort, and long-term wearability for physically demanding jobs such as nursing and logistics.
Latin America Exoskeleton Market Trends
The exoskeleton market in Latin America is growing significantly. In November 2024, Einstein Hospital in São Paulo is conducting clinical trials to assess the effectiveness of exoskeletons in rehabilitation. It will be the first hospital in Latin America to use an exoskeleton that senses the patient’s movement intentions, amplifies the neurological micro-signals, and aids in executing the movement. This innovation enables patients to play a more active role in their rehabilitation.
Brazil is facilitating innovation in exoskeleton technology through local initiatives. Startups, including Exy Innovation, are developing wearable exoskeletons tailored to the Brazilian workforce to enhance productivity and reduce workplace injuries. In addition, research institutions are collaborating to create cost-effective and adaptable solutions suitable for tropical environments, addressing the region's unique needs. In April 2025, Triptyque and Architects Office completed AGE360, a 124-metre residential skyscraper in Curitiba, Brazil, featuring a load-bearing concrete exoskeleton that eliminates internal columns and enables flexible floor layouts, supporting apartments and shared wellness spaces while responding to structural and urban constraints.
Middle East & Africa Exoskeleton Market Trends
The exoskeleton market in the Middle East and Africa (MEA) region is experiencing rapid growth. Countries such as Saudi Arabia, the UAE, and South Africa are investing in healthcare infrastructure, including rehabilitation centers equipped with exoskeleton technologies. Public and private sector investments support this expansion to enhance mobility solutions for individuals with disabilities. In December 2025, Lifeward expanded ReWalk® Personal Exoskeleton access in the UAE via an exclusive distribution agreement with Verita Neuro, integrating the device into in-patient neurorehabilitation programs and reinforcing the UAE’s position as a regional hub for advanced rehabilitation technologies.
The exoskeleton market in South Africa is witnessing advancements, focusing on enhancing mobility and strength. Local initiatives are developing wearable exoskeletons tailored to the South African workforce to improve productivity and reduce workplace injuries. In addition, research institutions collaborate to create cost-effective and adaptable solutions suitable for the region's unique needs. In February 2024, Minerals Council South Africa announced the testing of wearable exoskeletons as part of its fall-of-ground prevention strategy to improve miner safety during high-risk underground barring activities, focusing on reducing physical strain, improving endurance, and limiting exposure to unstable rock, highlighting growing industrial adoption of exoskeletons in mining safety applications.
Key Exoskeleton Company Insights
Key players who focus on innovation, product development, and strategic partnerships to maintain their market positions dominate the exoskeleton market. For instance, in March 2023, Svaya Robotics developed quadruped robot and a wearable exoskeleton. The exoskeleton is specially designed to match the body structure of Indian soldiers, enhancing their physical strength to help them walk long distances without fatigue and carry heavy loads with reduced effort. The market share is concentrated among established players with extensive resources for research and development, while emerging players focus on cost-effective solutions and technological advancements.
Key Exoskeleton Companies:
The following key companies have been profiled for this study on the exoskeleton market.
- Ekso Bionics
- ARMASTEC PTE. LTD
- Hocoma
- Lockheed Martin Corporation
- Suit X
- Rex Bionics Plc.
- RB3D
- ReWalk Robotics
- Cyberdyne, Inc.
- ActiveLink (Panasonic Corporation)
- DIH Medical
Recent Developments
-
In January 2026, Ascentiz unveiled a modular exoskeleton at CES 2026, featuring swappable hip and knee modules optimized with distinct drive technologies for natural movement assistance. The system integrates AI trained on over 690,000 gait cycles to deliver rapid mode switching and significant reductions in leg effort, knee cartilage pressure, and energy loss during dynamic and load-bearing activities.
-
In January 2026, OYMotion Technology and Melexis formed a partnership to advance robot perception and control, combining OYMotion’s system integration with Melexis’s sensor technology. The collaboration targets commercialization of dexterous hands and rehabilitation exoskeletons, leveraging OYMotion’s EMG/EEG AI, lightweight designs, and global supply chain to grow in medical, industrial, and humanoid robotics.
-
In February 2025, Chulalongkorn University has introduced the “Exoskeleton Wheelchair,” a robotic suit designed to assist people with disabilities in sitting, standing, and walking.
“I wanted to create a wheelchair that could walk. An ordinary wheelchair can only move on a regular path, but it cannot get on the bus or go up and down the stairs. So my view is that if a wheelchair could be folded up and help the person sitting in it to get up and walk, it would make it easier for people with reduced mobility to walk.”
- Assoc. Prof. Dr. Ronnapee Chaichawarat, a professor from the International School of Engineering (ISE), Chulalongkorn University
-
In February 2025, Wandercraft launched a landmark clinical trial for the Personal Exoskeleton, the world’s first self-balancing device aimed at restoring natural standing, walking, bending, and reaching abilities for individuals with severe mobility impairments.
“The launch of this trial is a major milestone in our journey to redefine what mobility technology can achieve. We’ve seen the profound impact of our exoskeletons in rehabilitation centers, and now we are thrilled to explore how the Personal Exoskeleton could represent a significant leap forward in mobility and walking independence for people with severe walking impairment, starting with SCI, enabling them to walk hands-free without relying on crutches or walkers."
- Matthieu Masselin, CEO of Wandercraft
-
In January 2025, German Bionic unveiled the Apogee ULTRA, which offers 80 lbs (36 kg) of lifting support. It is designed to enhance worker safety and productivity in industries such as logistics, manufacturing, and healthcare.
“We know from extensive experience in both industrial and healthcare settings that there are moments in a worker’s day when tasks demand an extra level of strength - whether it’s lifting a particularly heavy package or mobilizing an incapacitated patient. That’s exactly why we created the Ultra-Mode, giving users that powerful boost they need to complete these challenging tasks more safely and with greater ease.”
- Norma Steller, Head of Development at German Bionic
-
In December 2024, RoboCT's UGO Series Rehab Exoskeleton received CE certification, officially recognizing it as a medical device.
-
In November 2024, Hippos Exoskeleton will release a knee brace featuring airbags designed to help prevent ACL injuries.
“The core technology we invented is not only on the hardware side, but also on the software side: the AI, the data processing capability and the ability to use our AI model, which we designed by ourselves, to personalize the triggering threshold for every single individual, athletes, soldiers, patients.”
- Kylin Shaw, Hippos CEO
-
In December 2022, Ekso Bionics announced the acquisition of Parker Hannifin Corporation’s Human Motion and Control business unit. This acquisition includes Parker’s Indego lower limb exoskeleton product line and the strategic development of robot-assisted prosthetic and orthotic devices.
-
In June 2022, CYBERDYNE Inc. formed a business and capital alliance with LIFESCAPES Inc. This collaboration highlighted the combined effort to further leverage the exoskeleton technology arena and improve rehabilitation options for patients with severe paralysis.
-
In June 2022, CYBERDYNE Inc. announced its collaboration with the Social Security Organization (SOCSO) in Malaysia. This initiative led to the expansion of Cybernics Treatment using hybrid assistive limb (HAL) for SOCSO-insured patients.
Exoskeleton Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 694.46 million
Revenue forecast in 2033
USD 1.79 billion
Growth rate
CAGR of 14.48% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue & Volume forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Mobility, technology, extremity, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; and MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; Spain; Italy; France; Denmark; Norway; Sweden; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Ekso Bionics; ARMASTEC PTE. LTD; Hocoma; Lockheed Martin Corporation; Suit X; Rex Bionics Plc.; RB3D; ReWalk Robotics; Cyberdyne, Inc.; ActiveLink (Panasonic Corporation); DIH Medical
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options Global Exoskeleton Market Report Segmentation
This report forecasts revenue and volume growth at global, regional, and country level and provides an analysis on industry trends in each of the sub segments from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the exoskeleton market report on the basis of mobility, technology, extremity, end use and region:
-
Mobility Outlook (Volume, 000' Units, Revenue USD Million 2021 - 2033)
-
Mobile
-
Fixed/Stationary
-
-
Technology Outlook (Volume, 000' Units, Revenue USD Million 2021 - 2033)
-
Powered
-
Upper Body
-
Lower Body
-
Full Body
-
-
Non-powered
-
Upper Body
-
Lower Body
-
Full Body
-
-
-
Extremity Outlook (Volume, 000' Units, Revenue USD Million 2021 - 2033)
-
Upper Body
-
Lower Body
-
Full Body
-
-
End Use Outlook (Volume, 000' Units, Revenue USD Million 2021 - 2033)
-
Healthcare
-
Military
-
Industry
-
-
Regional Outlook (Volume, 000' Units, Revenue USD Million 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
U.K.
-
Spain
-
Italy
-
France
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the exoskeleton market include Rex Bionics Plc.; Hocoma; Lockheed Martin Corporation; Suit X; ReWalk Robotics; RB3D; ReWalk Robotics Ltd; Cyberdyne, Inc.; and ActiveLink (Panasonic Corporation).
b. Key factors that are driving the exoskeleton market growth include the growing adoption of exoskeletons in healthcare and non-healthcare settings, technological advancements, and the increasing prevalence of spinal cord injuries (SCIs).
b. The global exoskeleton market size was valued at USD 590.02 million in 2025 and is expected to reach USD 694.46 million in 2026.
b. Mobile segment dominated the exoskeleton market with a revenue share of 62.64% in 2025. This is attributable to the introduction of innovative mobile exoskeletons by manufacturers for the aging and disabled population.
b. The global exoskeleton market is expected to grow at a compound annual growth rate of 14.48% from 2026 to 2033 to reach USD 1.79 billion by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










