- Home
- »
- Medical Devices
- »
-
CAUTI Prevention Urology Products Market Size Report 2033GVR Report cover
![CAUTI Prevention Urology Products Market Size, Share & Trends Report]()
CAUTI Prevention Urology Products Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Preventive Drainage Devices (Primary Prevention), Sterile Circuit & Maintenance Systems), By Application, By End-Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-849-8
- Number of Report Pages: 125
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
CAUTI Prevention Urology Products Market Summary
The global CAUTI prevention urology products market size was valued at USD 3.43 billion in 2025 and is projected to reach USD 6.05 billion by 2033, growing at a CAGR of 7.35% from 2026 to 2033. The market is driven by the rising incidence of catheter-associated urinary tract infections and increasing awareness of infection control in healthcare settings.
Key Market Trends & Insights
- North America dominated the CAUTI prevention urology products market with the largest revenue share of 37.76% in 2025.
- The CAUTI prevention urology products market in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product, the preventive drainage devices (Primary Prevention) segment led the market with the largest revenue share in 2025.
- By application, the urinary incontinence management segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.43 Billion
- 2033 Projected Market Size: USD 6.05 Billion
- CAGR (2026-2033): 7.35%
- North America: Largest market in 2025
Strict regulatory guidelines and hospital initiatives to reduce hospital-acquired infections are accelerating the adoption of antimicrobial and safety-enhanced urology products. Growing hospitalization rates, an aging population, and expanded use of urinary catheters in acute and long-term care further support market growth. The rising incidence of CAUTIs and other hospital-acquired infections is a key driver of the CAUTI prevention urology products market.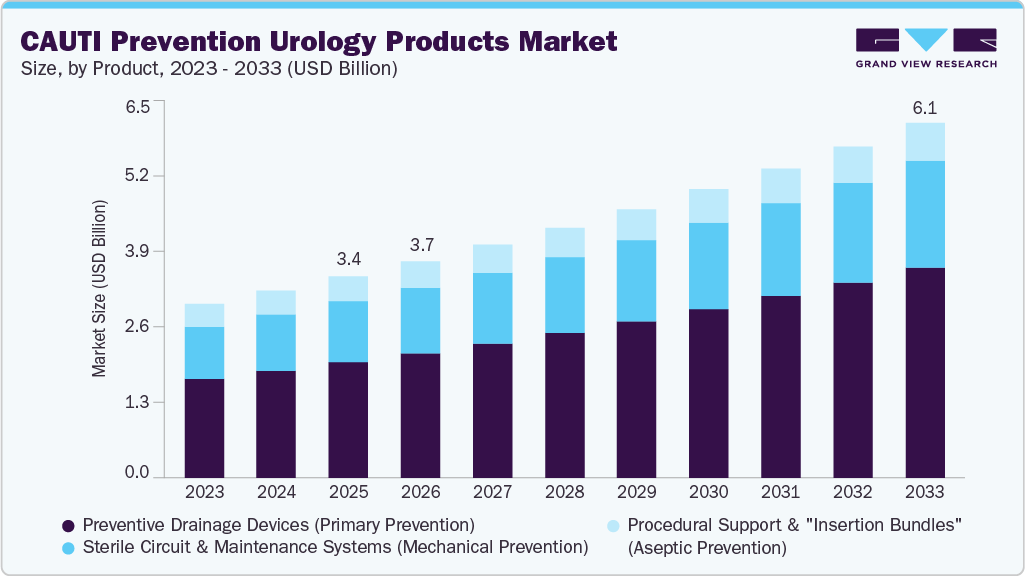
Increased awareness of patient safety, coupled with the clinical and financial impact of prolonged hospital stays, is pushing healthcare providers to prioritize infection prevention. This has accelerated demand for advanced urology products such as antimicrobial catheters, closed drainage systems, and catheter management solutions designed to reduce infection risk. According to the National Healthcare Safety Network (NHSN), about 12%-16% of adult hospital patients use an indwelling urinary catheter at some point during their hospital stay. Each additional day the catheter remains in place increases the patient’s risk of developing a catheter-associated urinary tract infection (CAUTI) by 3%-7%.
Prevalence of CAUTI in 2023
Setting
Number of Facilities Reporting
Number of CAUTIs Reported
Acute care hospitals
3,774
17,370
Critical access hospitals
1,070
1,259
Inpatient rehabilitation facilities
1,218
1,415
Long-term acute care hospitals
390
1,481
Total
6,452
21,525
Source: Association for Professionals in Infection Control and Epidemiology
As CAUTI cases rise, healthcare facilities face higher patient morbidity, longer hospital stays, and increased treatment costs. To mitigate these risks, hospitals and care providers adopt infection-prevention urology products such as antimicrobial catheters, closed drainage systems, catheter securement devices, and alternative bladder management solutions. The growing need to reduce infection rates and comply with clinical guidelines directly fuels the demand and innovation in the CAUTI prevention market.
Urinary incontinence significantly drives the CAUTI prevention urology products market by increasing catheter use across hospitals, care homes, and home care settings, thereby increasing the risk of catheter-associated urinary tract infections. The resulting clinical complications, longer hospital stays, and increased healthcare costs are prompting providers to adopt CAUTI prevention strategies, such as antimicrobial catheters, closed drainage systems, and non-invasive bladder management alternatives, to improve outcomes and reduce infection rates. According to the Urology Foundation, an estimated 5 million people in England currently live with an overactive bladder, and this number is projected to increase to more than 7 million by 2035.
Prevalence and Impact of Urinary Incontinence in UK (2025)
Category
Key Statistic / Impact (UK)
Prevalence of bladder problems
1 in 5 people experience bladder problems (~14 million people)
Women affected
Around 1 in 3 women (34%) live with urinary incontinence
Men affected
Over 2 million men experience urinary incontinence
Cost to NHS
£233 million estimated annual cost
Hospital-acquired UTIs
Incontinence increases the risk of hospital-acquired UTIs
Impact on hospital stay
Hospital-acquired UTIs extend length of stay by ~6 days
Care home residents
Over 50% of care home residents have urinary incontinence
Source: The Urology Foundation
As a result of urinary incontinence, increased catheter use increases the risk of CAUTIs. This drives demand for urology products designed to prevent infections, such as antimicrobial catheters and closed drainage systems.
Benign prostatic hyperplasia (BPH) is another factor driving the industry, as it commonly causes urinary retention and voiding difficulties in older men, often necessitating catheterization. Increased catheter use elevates the risk of catheter-associated urinary tract infections, particularly in hospital and long-term care settings. This has driven demand for CAUTI prevention solutions, such as antimicrobial catheters, catheter management systems, and alternative bladder management products, to reduce infection risk and improve patient outcomes.
Incident Cases of BPH from 2022 to 2035
Age Group
2022
2035
Overall
13,918.71
18,109.76
40-59
3,546.70
4,165.56
60-79
9,769.99
12,977.31
80+
602.02
966.9
Source: Springer
(Note: Measured in Number of Cases × 1000)
Prevalent Cases of BPH from 2022 to 2035
Age Group
2022
2035
Overall
113,942.78
156,343.38
40-59
17,258.92
19,947.18
60-79
82,408.51
113,499.71
80+
14,275.35
22,896.50
Source: Springer
(Note: Measured in Number of Cases × 1000)
As a result of benign prostatic hyperplasia (BPH), many men experience urinary retention that often requires catheterization, increasing the risk of CAUTIs. This drives demand for urology products designed to prevent infections, such as antimicrobial catheters and advanced catheter management systems.
Market Concentration & Characteristics
The CAUTI prevention urology products market is characterized by a high degree of innovation focused on reducing infection risk and improving patient safety. Advancements include antimicrobial- and hydrophilic-coated catheters, closed drainage systems, and sensor-based monitoring technologies. Manufacturers are also developing single-use and ergonomically designed products to enhance ease of use and compliance with infection prevention protocols. For instance, in February 2025, the MDPI journal published a study on hydrogel-coated silicone urinary catheters featuring antibacterial and lubricating properties. These modified catheters demonstrated strong antibacterial activity, excellent biocompatibility, and reduced friction, highlighting their potential for widespread clinical use.
The industry has seen a moderate level of mergers and acquisitions as companies seek to expand their product portfolios and strengthen their presence in infection prevention solutions. Strategic acquisitions are often aimed at gaining access to advanced catheter technologies and antimicrobial innovations. This consolidation trend helps companies enhance competitiveness, improve distribution networks, and accelerate product development.
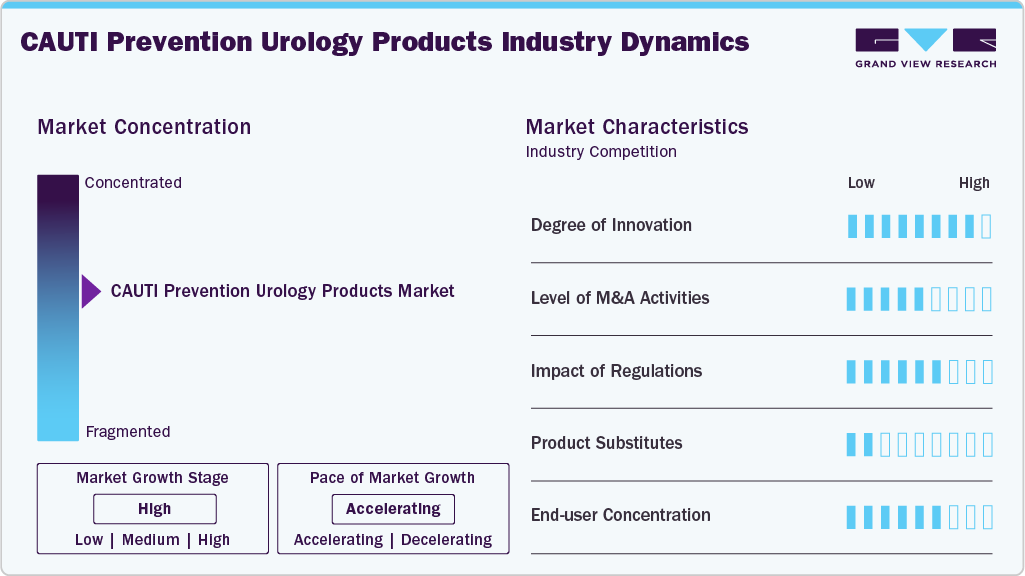
Regulations have a significant impact on the market, as strict guidelines focus on reducing hospital-acquired infections and improving patient safety. According to UCSF Health, 12% to 16% of hospitalized patients receive short-term indwelling urinary catheters (IUCs), and studies indicate that up to half of these catheters may be placed without a proper clinical reason. The risk of developing a urinary tract infection increases with the length of time a catheter remains in place, with a daily risk of 3% to 7% for bacteriuria. The most effective way to prevent these infections is to limit the number of patients receiving catheters and reduce the duration of catheter use. In addition to infections, IUCs can cause non-infectious complications, including urethral inflammation, strictures, mechanical trauma, and reduced mobility.
End-user concentration is primarily centered on hospitals, which account for the largest share due to high catheter usage and strict infection control protocols. Long-term care facilities and nursing homes are also significant end users, given the prolonged use of urinary catheters among elderly patients. Clinics and home healthcare settings are emerging as growing segments as awareness of CAUTI prevention increases.
Product Insights
Preventive drainage devices used for primary prevention dominated the CAUTI prevention urology products market in 2025 and are also expected to experience the fastest growth in the forecast period, driven by their critical role in minimizing infection risk at the point of catheter use. These devices help maintain closed drainage systems and prevent bacterial entry, making them widely adopted in hospitals and long-term care settings. Their ease of implementation and alignment with infection control guidelines further reinforce their leading market position.
Sterile circuit & maintenance systems (mechanical prevention) are growing rapidly as healthcare facilities focus on reducing contamination during catheter care. These systems help maintain sterility throughout catheter use, lowering the risk of infection associated with handling and maintenance. Increasing adherence to infection control protocols and rising awareness of mechanical prevention methods are driving strong adoption across hospitals and long-term care settings.
Application Insights
Urinary incontinence management products dominated the market in 2025 due to the high prevalence of incontinence among elderly and hospitalized patients. Effective incontinence management reduces the need for prolonged catheterization, thereby lowering the risk of catheter-associated urinary tract infections. The growing adoption of non-invasive management solutions and the increased focus on patient comfort and infection prevention further support the dominance of this segment.
"Urinary incontinence is a common yet under-discussed condition that impacts 25 million Americans and can have profound effects on confidence, social engagement and quality of life, especially among those with mobility challenges," said Mike Cusack, worldwide president of Urology and Critical Care at BD. "With the launch of the PureWick Portable Collection System, we have an opportunity to help people regain their confidence and independence by delivering a discreet, accessible solution that empowers individuals to participate more fully in everyday activities."
Benign Prostatic Hyperplasia (BPH) and obstructive urology are expected to be the fastest-growing segments during the forecast period. The rising prevalence of BPH, particularly among the aging male population, is increasing the need for effective urinary management and catheterization solutions. Growing awareness of infection risks and the adoption of preventive urology products in the management of obstructive conditions are further driving rapid growth in this segment.
End Use Insights
Hospitals dominated the market for CAUTI prevention urology products in 2025 due to high patient admissions, frequent catheter use, and strict infection control protocols. The presence of advanced infrastructure and trained healthcare professionals supports the widespread adoption of preventive urology products. In addition, regulatory pressure to reduce hospital-acquired infections reinforces hospitals as the leading end-use segment.
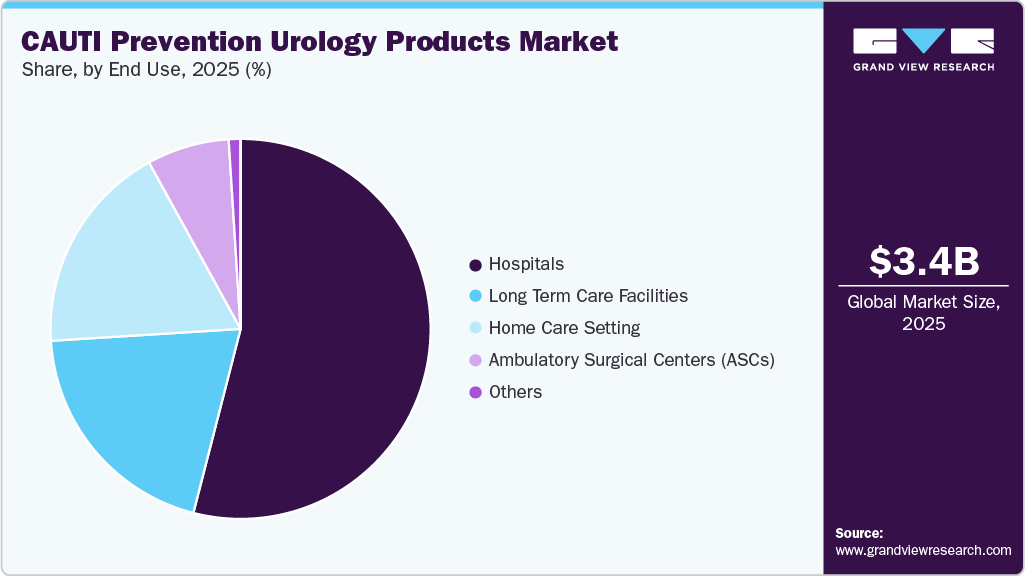
The home care setting is expected to be the fastest-growing segment during the forecast period. Increasing adoption of home healthcare services, rising awareness of infection prevention, and the growing elderly population requiring long-term catheter care are driving this growth. Convenience, patient comfort, and the availability of user-friendly urology products further support the rapid expansion of this segment.
Regional Insights
The CAUTI prevention urology products industry in North America held the largest share and accounted for 37.76% of global revenue in 2025. Driven by stringent healthcare regulations, rising awareness of hospital-acquired infections, and advanced infection control protocols. Increasing adoption of antimicrobial catheters, sterile drainage systems, and innovative mechanical prevention devices is shaping market trends. In addition, the expanding geriatric population and growing demand for home healthcare solutions are further fueling market development in the region.
U.S. CAUTI Prevention Urology Products Market Trends
The U.S. CAUTI prevention urology products industry is growing significantly, driven by stringent FDA regulations and hospital initiatives to reduce catheter-associated urinary tract infections. Adoption of advanced antimicrobial catheters, closed drainage systems, and mechanical prevention technologies is increasing across healthcare facilities. Rising awareness of infection control and the growing elderly population further support market expansion in the country.
Europe CAUTI Prevention Urology Products Market Trends
The CAUTI prevention urology products industry in Europe is expanding due to increasing emphasis on patient safety and stringent infection control guidelines across hospitals. Rising adoption of antimicrobial catheters, sterile drainage systems, and mechanical prevention devices is driving market growth. In addition, aging populations and growing awareness of CAUTI prevention in long-term care facilities are shaping key market trends in the region.
The UK CAUTI prevention urology products industry is witnessing growth driven by strict NHS infection control policies and initiatives to reduce hospital-acquired infections. Increasing use of antimicrobial catheters, sterile drainage systems, and mechanical prevention solutions is shaping market trends. In addition, rising awareness among healthcare providers and the growing elderly population are supporting market expansion in the country.
The CAUTI prevention urology products industry in France is experiencing significant growth, fueled by stringent national healthcare regulations and initiatives to minimize hospital-acquired infections. Adoption of advanced antimicrobial catheters, sterile drainage systems, and mechanical prevention devices is increasing across hospitals and long-term care facilities. In addition, rising awareness of infection control and an aging population are driving market demand in the country.
Asia Pacific CAUTI Prevention Urology Products Market Trends
The CAUTI prevention urology products industry in the Asia Pacific is witnessing rapid growth driven by expanding healthcare infrastructure, rising hospital admissions, and increasing awareness of infection prevention. Adoption of antimicrobial catheters, closed drainage systems, and mechanical prevention technologies is expanding across hospitals and long-term care facilities. In addition, the region’s aging population and increasing prevalence of chronic urinary conditions are driving demand for urology products.
The China CAUTI prevention urology products industry is growing rapidly, driven by rising healthcare expenditure, increasing hospital admissions, and enhanced focus on infection control. Adoption of antimicrobial catheters, sterile drainage systems, and mechanical prevention devices is accelerating in both hospitals and long-term care facilities. The aging population and growing prevalence of urinary disorders are boosting demand for effective CAUTI prevention solutions.
Latin America CAUTI Prevention Urology Products Market Trends
The CAUTI prevention urology products industry in Latin America is experiencing significant growth, supported by increasing awareness of hospital-acquired infections and government initiatives to improve healthcare quality. Rising adoption of antimicrobial catheters, sterile drainage systems, and mechanical prevention devices in hospitals and long-term care facilities is driving market expansion. In addition, growing hospital infrastructure and an aging population are contributing to increased demand in the region.
Middle East and Africa CAUTI Prevention Urology Products Market Trends
The CAUTI prevention urology products industry in the Middle East and Africa is witnessing gradual growth, driven by increasing healthcare investments and rising awareness of hospital-acquired infections. Adoption of antimicrobial catheters, sterile drainage systems, and mechanical prevention devices is growing in hospitals and long-term care facilities. Furthermore, expanding healthcare infrastructure and an aging population are driving market demand across the region.
Key CAUTI Prevention Urology Products Company Insights
The market is dominated by leading medical device companies with extensive product portfolios and strong distribution networks. Key players focus on advanced antimicrobial catheters, sterile drainage systems, and mechanical prevention solutions, capturing major market share in hospitals and long-term care settings. Both global and regional companies actively drive the market through innovation, partnerships, and strategic expansions.
Key CAUTI Prevention Urology Products Companies:
The following key companies have been profiled for this study on the CAUTI prevention urology products market.
- BD
- B. Braun SE
- Coloplast
- Teleflex Incorporated
- Cook Medical
- Medtronic
- Hollister Incorporated
- ConvaTec Group PLC
- Boston Scientific Corporation
- Cardinal Health
- Merit Medical
- Medline Industries, LP.
- Poiesis Medical LLC
Recent Developments
-
In November 2025, BD (Becton, Dickinson and Company), a global medical technology company, launched the PureWick Portable Collection System, a discreet, first-of-its-kind, battery-powered personal urine management device designed for wheelchair users to improve mobility at home and beyond.
"Urinary incontinence is a common yet under-discussed condition that impacts 25 million Americans and can have profound effects on confidence, social engagement and quality of life, especially among those with mobility challenges. With the launch of the PureWick Portable Collection System, we have an opportunity to help people regain their confidence and independence by delivering a discreet, accessible solution that empowers individuals to participate more fully in everyday activities." said Mike Cusack, worldwide president of Urology and Critical Care at BD.
-
In June 2025, Teleflex Incorporated, a global medical technology provider, announced results from a multinational study demonstrating the efficacy of Arrow Chlorhexidine-Impregnated Central Venous Catheters (CVCs). The prospective cohort study included over 6,670 patients from 12 intensive care units across eight hospitals in India, Malaysia, Papua New Guinea, Colombia, Egypt, and Turkey.
This new multinational study suggests that even with proper training and adherence to best practices-including catheter insertion, care, and maximal barrier precautions-unprotected CVCs may be an independent risk factor for CLABSI compared to chlorhexidine-impregnated CVCs, said Dr. Amy Bardin, Vice President of Clinical and Medical Affairs at Teleflex.
-
In January 2025, Teleflex Incorporated, a global medical technology provider, announced it was awarded a contract by Vizient, Inc., the largest provider-driven healthcare performance improvement company in the U.S., for the supply of Teleflex’s Central Venous Access Catheters and Arterial Catheters, effective January 1, 2025.
“As the market leader in both Central Venous Catheters (CVCs)1 and Arterials2, we are pleased to offer Vizient provider-customers one of the broadest portfolios of vascular access products. Arrow Vascular Access Products are designed to equally benefit clinicians and patients, help protect against vascular access related complications like infection, thrombosis, and tip malposition, and help clinicians follow independent third-party vascular access guidelines.” said Lisa Kudlacz, President and General Manager, Teleflex Vascular.
-
In May 2024, Coloplast expanded its Luja portfolio with a next-generation female intermittent catheter, designed to enable complete bladder emptying in a single free flow and help reduce the risk of urinary tract infections.
“So far, we have received great feedback on Luja for men in our product evaluations. Nearly all healthcare professionals would recommend the male catheter to their patients3, and seven out of ten users feel confident that it empties their bladder completely without needing to reposition the catheter4,” said Nicolai Buhl, Executive Vice President of Innovation, Coloplast.
-
In February 2023, Coloplast launched a male catheter specifically designed to reduce the risk of urinary tract infections.
-
In March 2023, Coloplast reported that its Luja catheter significantly improved bladder emptying compared to a competitor catheter.
CAUTI Prevention Urology Products Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.69 billion
Revenue forecast in 2033
USD 6.05 billion
Growth rate
CAGR of 7.35% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East and Africa
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Norway; Denmark; Sweden; Spain; Italy; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; KSA; UAE; South Africa; Kuwait
Key companies profiled
BD; B. Braun SE; Coloplast; Teleflex Incorporated; Cook Medical; Medtronic; Hollister Incorporated; ConvaTec Group PLC; Boston Scientific Corporation; Cardinal Health; Merit Medical; Medline Industries, LP.; Poiesis Medical LLC
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global CAUTI Prevention Urology Products Market Report Segmentation
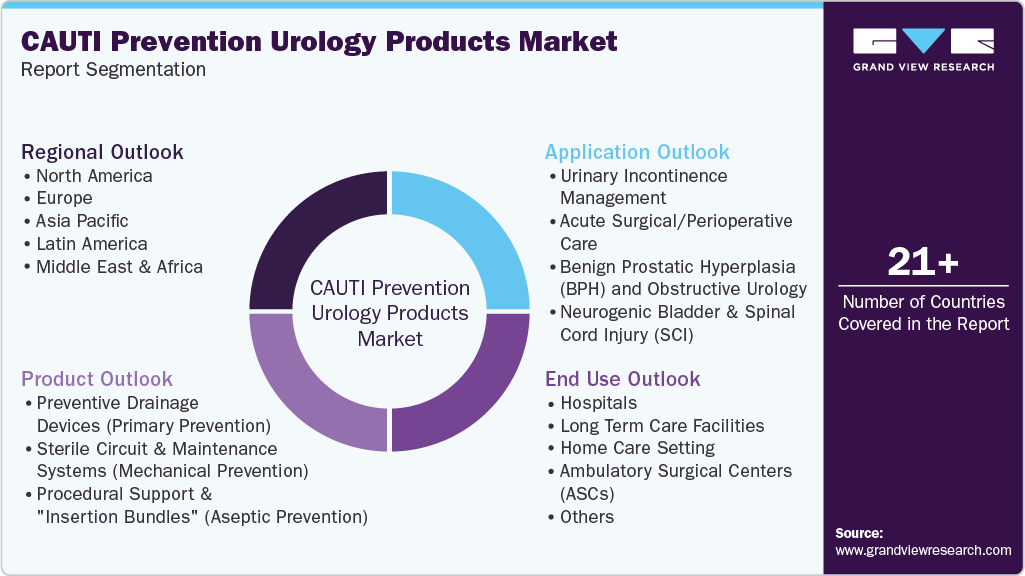
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global CAUTI prevention urology products market report on the basis of product, application, end use and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Preventive Drainage Devices (Primary Prevention)
-
Intermittent (Single-Use) Catheters
-
Antibacterial/Antimicrobial-Coated
-
Compact/Travel Intermittent
-
-
External (Non-Invasive) Collection Systems
-
Indwelling (Foley) Catheters
-
Silver-Alloy Coated Foley
-
Silicone-Only Foley
-
Drug-Eluting Catheters
-
-
-
Sterile Circuit & Maintenance Systems (Mechanical Prevention)
-
Closed Drainage Bags
-
Anti-Reflux Systems
-
Urine Meters
-
-
Catheter Stabilization/Securement Devices
-
Needleless Sampling Ports
-
-
Procedural Support & "Insertion Bundles" (Aseptic Prevention)
-
Pre-Packaged Aseptic Trays
-
Bladder Scanners (Portable Ultrasound)
-
Sterile Lubricant & Cleansing Solutions
-
-
-
Application Outlook (Revenue, USD Million; 2021 - 2033)
-
Urinary Incontinence Management
-
Acute Surgical/Perioperative Care
-
Benign Prostatic Hyperplasia (BPH) and Obstructive Urology
-
Neurogenic Bladder & Spinal Cord Injury (SCI)
-
-
End Use Outlook (Revenue, USD Million; 2021 - 2033)
-
Hospitals
-
Long Term Care Facilities
-
Home Care Setting
-
Ambulatory Surgical Centers (ASCs)
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global CAUTI prevention urology products market size was estimated at USD 3.43 billion in 2025.
b. The global CAUTI prevention urology products market is expected to grow at a compound annual growth rate of 7.35% from 2026 to 2033 to reach USD 6.05 billion by 2033.
b. North America dominated the CAUTI prevention urology products market, accounting for 37.76% in 2025. This is attributable to the high prevalence of hospital-acquired infections, advanced healthcare infrastructure, and high adoption of advanced infection prevention technologies across hospitals and long-term care facilities.
b. Key companies operating in the CAUTI prevention urology products market include BD, B. Braun SE, Coloplast, Teleflex Incorporated, Cook Medical, Medtronic, Hollister Incorporated, ConvaTec Group PLC, Boston Scientific Corporation, Cardinal Health, Merit Medical, Medline Industries, LP., and Poiesis Medical LLC.
b. The CAUTI prevention urology products market is primarily driven by the rising incidence of hospital-acquired infections and the increasing use of urinary catheters in hospitals, ICUs, and long-term care settings. Growing aging populations and chronic disease prevalence are increasing long-term catheterization, which raises infection risk and drives demand for prevention products.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










